Tauopathies Treatment Market is estimated to be valued at USD 1,144.7 Mn in 2025 and is expected to reach USD 2,230.7 Mn in 2032, exhibiting a compound annual growth rate (CAGR) of 10.0% from 2025 to 2032.
Analysts’ views on the global tauopathies treatment market:
Increasing prevalence of neurodegenerative disorders, new product launches, and strategies like mergers, acquisitions, and collaborations are expected to drive the global tauopathies treatment market growth over the forecast period. For instance, according to data published by the World Health Organization, uptil March 15, 2023, more than 55 million people had dementia worldwide, over 60% of whom live in low-and middle-income countries. Every year, there are nearly 10 million new cases. Alzheimer’s disease is the most common form of dementia and may contribute to 60-70% of cases. Dementia is currently the seventh leading cause of death and one of the major causes of disability and dependency among older people globally. With such high prevalence of the diseases related to tauopathies, the demand for treatment will also increase, thus driving the market growth.
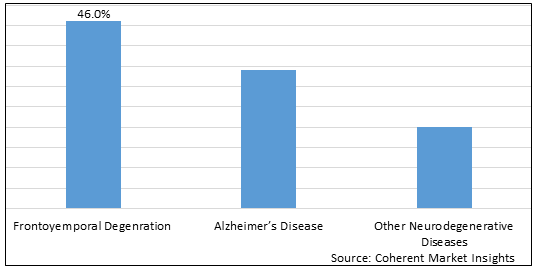
Figure 1. Global Tauopathies Treatment Market Share (%), By Disease, 2025
To learn more about this report, Download Free Sample
Global Tauopathies Treatment Market - Drivers
Increasing research and development activities in tauopathies treatment
Increasing research and development in the field of tauopathies treatment is expected to drive the global tauopathies treatment market growth over the forecast period. For instance, according to article published by the journal Annals of Neurology on March 10, 2023, a class of drugs called Dual Orexin Receptor Antagonists (DORAs) developed to help treat insomnia may play a role in the development of Alzheimer’s disease. In this study, 38 volunteers were participated. All participants were between the ages of 45 to 65 and cognitively healthy. The researchers randomly assigned them to receive either two higher doses of a DORA called suvorexant over 36 hours, two lower doses of suvorexant, or a placebo. The team then measured changes in levels of amyloid-β and a form of tau called phosphorylated tau in the cerebrospinal fluid. People receiving suvorexant did not show an increase in sleep time or quality over the night of the study. But those who received the higher dose of the drug showed a 10-15% drop in phosphorylation at a site on tau that contributes to tau tangles. By 12 hours after receiving high-dose suvorexant, participants had amyloid-β levels 10-20% lower than those in the placebo group. These levels increased over time, then dropped again after a second dose of the drug.
Increasing launch of new products for the treatment of tauopathies
Increasing launch of new products for the efficient treatment of tauopathies is expected to drive the global tauopathies treatment market growth. For instance, on January 6, 2023, Eisai Co., Ltd., a Japan-based pharmaceutical company, along with Biogen Inc., a U.S.-based biotechnology company, announced that the U.S. Food and Drug Administration (FDA) has approved LEQEMBI, lecanemab-irmb 100 mg/mL injection for intravenous use, a humanized immunoglobulin gamma 1 (IgG1) monoclonal antibody directed against aggregated soluble and insoluble forms of amyloid beta (Aβ) for the treatment of Alzheimer's Disease (AD). The approval is based on Phase 2 data that demonstrated that LEQEMBI reduced the accumulation of Aβ plaque in the brain, a defining feature of AD. Using the recently published data from the large global confirmatory Phase 3 clinical trial, Clarity AD, Eisai Co., Ltd., will work quickly to file a Supplemental Biologics License Application (sBLA) to the FDA for approval under the traditional pathway.
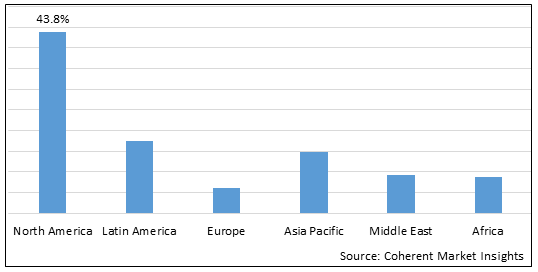
Figure 2. Global Tauopathies Treatment Market Share (%), By Region, 2025
To learn more about this report, Download Free Sample
Global Tauopathies Treatment Market - Regional Analysis
Among region, North America is estimated to hold a dominant position in the global tauopathies treatment market over the forecast period, owing to increasing launches of products. For instance, on May 3, 2025, Eli Lilly and Company, a U.S.-based pharmaceutical company announced positive results of the TRAILBLAZER-ALZ 2 Phase 3 study showing that donanemab significantly slowed cognitive and functional decline in people with early symptomatic Alzheimer's disease. The primary endpoint was of change from baseline until 18 months on the integrated Alzheimer's Disease Rating Scale (iADRS) while secondary endpoints of cognitive and functional decline were also met and showed highly statistically significant clinical benefits with similar magnitude. In this study, the primary endpoint (iADRS) showed 35% slowing of decline, and an important key secondary endpoint (Clinical Dementia Rating-Sum of Boxes, or CDR-SB) showed 36% slowing of decline (p<0.0001) over 18 months.
Global Tauopathies Treatment Market - Impact of Coronavirus (COVID-19) Pandemic
Since the COVID-19 virus outbreak in December 2019, the disease has spread to over 100 countries across the globe, and the World Health Organization declared it a public health emergency on January 30, 2020.
COVID-19 affected the economy in three main ways: by directly affecting the production and demand of drugs, by creating disruptions in distribution channels, and through its financial impact on firms and financial markets. Due to nationwide lockdowns, several countries, such as China, India, Saudi Arabia, the U.A.E., Egypt, and others, faced problems regarding transportation of drugs from one place to another.
However, the COVID-19 pandemic had negative impact on the global tauopathies treatment market, due to reduced patients visits and follow-ups of treatment during COVID 19. For instance, according to an article published by International Journal of Nursing Studies, visiting restrictions had several consequences, mainly negative, for the patient's health, the health and well-being of family members and the provision of care. Among physical health consequences, reduced nutrition intake, decreased activities of daily living and increased physical pain and symptoms were reported. Among mental health consequences for the patient, loneliness, depressive symptoms, agitation, aggression, reduced cognitive ability, and overall dissatisfaction were observed. For family members, worry, anxiety and uncertainty occurred, and they reported an increased need for information from care providers. For care providers, visiting restrictions added the burdens of ethical dilemmas, learning new technical means to enable social interaction and an increased demand for communication with families and providing social support to both family members and patients.
Tauopathies Treatment Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2024 | Market Size in 2025: | USD 1,144.7 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2025 To 2032 |
| Forecast Period 2025 to 2032 CAGR: | 10.0% | 2032 Value Projection: | USD 2,230.7 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Avanir Pharmaceuticals, Inc., Otsuka Pharmaceutical Co., Ltd., AB Science, Cortice Biosciences, REGENXBIO Inc., Aquinnah Pharmaceuticals, Neurimmune, Oligomerix, Inc., Biogen, TauRx, Sellas, Catalent Inc., Selvita S.A, Bristol-Myers Squibb Company, Chronos Therapeutics Limited, and SK Biopharmaceuticals Co., Ltd. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Global Tauopathies Treatment Market Segmentation:
The global tauopathies treatment market report is segmented into disease, distribution channel, and region.
Based on disease, the global tauopathies treatment market is segmented into frontoyemporal degeneration, Alzheimer’s disease, and other neurodegenerative diseases. Out of which, the frontoyemporal degeneration segment is expected to dominate the market due to increasing research and development activities in this region.
Based on distribution channel, the global tauopathies treatment market is segmented into hospital pharmacy, retail pharmacy, and online sales. Among these, the hospital pharmacy segment is expected to dominate the market over the forecast period due to increasing numbers of hospital pharmacies for tauopathies treatment.
Based on region, the global tauopathies treatment market is segmented into North America, Latin America, Europe, Asia Pacific, Middle East, and Africa. Among these, North America is expected to dominate the market over the forecast period due to increasing launch of products in the region.
Among all segmentation, the disease segment has the highest potential due to increasing clinical trials by market players. For instance, on June 27, 2022, Otsuka Pharmaceutical Co., Ltd., a Japan-based pharmaceutical company announced positive results of the Phase 3 clinical trial of brexpiprazole in the treatment of agitation in patients with Alzheimer's dementia. The analysis concluded that there is a statistically significant difference (p=0.0026) in the mean change from baseline to week 12 in the Cohen-Mansfield Agitation Inventory (CMAI) total score between brexpiprazole and placebo.
Global Tauopathies Treatment Market Cross Sectional Analysis:
Introduction of newer products and technologies in cell culture by key market players in Europe is expected to drive the growth of the disease segment in the region. For instance, on June 28, 2022, the European Commission approved Xenpozyme (olipudase alfa), a product of Sanofi, a France-based multinational pharmaceutical and healthcare company, as the first enzyme replacement therapy for the treatment of non-central nervous system manifestation of acid sphingomyelinase deficiency historically known as Niemann-Pick disease types A, A/B, and B in pediatric and adult patients. The approval is based on positive data from the ASCEND and ASCEND-Peds clinical trials. Given the urgent unmet medical needs of the ASMD community, the European Medicines Agency (EMA) granted Xenpozyme PRIority MEdicines (PRIME) designation. Xenpozyme has also received special breakthrough designations from several other regulatory agencies around the world.
Global Tauopathies Treatment Market: Key Developments
On February 12 2020, Oligomerix, Inc., a U.S.-based biotechnology company focused on developing disease-modifying and treatment therapeutics for neurodegenerative diseases, presented efficacy data in two pre-clinical models of tauopathy. The findings were presented by the company at the TAU2020 Global Conference. According to Oligomerix’s findings, their lead compound decreased soluble levels of tau self-association and caused a dose-dependent drop of insoluble as well as phosphorylated insoluble tau aggregates in the tau mice brains, which represents tauopathy in Alzheimer’s Disease (AD).
On January 13, 2020, Biogen Inc., a U.S.-based biotechnology company announced the acquisition of Alzheimers and Parkinson’s disease assets from Pfizer Inc., a U.S.-based multinational pharmaceutical and biotechnology corporation for US$ 710 million. Biogen is buying novel CNS penetrate inhibitor of casein kinase 1 (CK1) to develop for patients with behavioral and neurological symptoms in various psychiatric and neurologic diseases. Biogen Inc. indicates its initial focus will be to develop the Phase I compound for Sundowning in Alzheimer’s disease (AD) and Irregular Sleep Wake Rhythm Disorder (ISWRD) in Parkinson’s Disease (PD). Under the terms of the deal, Biogen Inc. is paying Pfizer Inc. US$ 75 million upfront with up to US$ 635 million in various milestones, as well as tiered royalties in the high single digits to sub-teens.
On April 13, 2023, Walgreen Co., a U.S.-based pharmacy store chain, announced a collaboration with Prothena, an Ireland-based biotechnology company to accelerate patient identification and recruitment for Prothena’s ongoing ASCENT-2 multiple ascending dose clinical trial evaluating the safety and tolerability of PRX012, a potential best-in-class anti-amyloid beta antibody under development for the treatment of Alzheimer’s disease. As part of the collaboration, Walgreens Co. will leverage its national footprint, portfolio of industry-leading healthcare companies and compliance framework to match patient populations to this Prothena clinical trial for PRX012, which has been granted Fast Track designation by the U.S. Food and Drug Administration.
On June 7, 2021, U.S. Food and Drug Administration approved Aduhelm (aducanumab) for the treatment of Alzheimer’s disease. Researchers evaluated Aduhelm’s efficacy in three separate studies representing a total of 3,482 patients. The patients receiving treatment has significant dose and time dependent reduction of amyloid beta plaque, while patients in the control arm of the studies had no reduction of amyloid beta plaque. Amyloid beta plaque was quantified using Positron Emission Tomography (PET) imaging to estimate the brain levels of amyloid beta plaque in a composite of brain regions expected to be widely affected by Alzheimer’s disease.
Global Tauopathies Treatment Market: Key Trends
Partnerships between organizations to spread awareness
Partnerships between organizations to spread awareness can drive the market growth. For instance, on October 31, 2022, on occasion of National Alzheimer’s Disease Awareness Month, 2022 U.S. government launched the transformational Advanced Research Projects Agency for Health (ARPA-H) at the National Institutes of Health,U.S. government organization which is investing a billion dollars in cutting-edge research to prevent, treat, and cure Alzheimer’s and other deadly diseases. Modeled on the Pentagon program that brought game-changing technologies like the Internet and GPS, ARPA-H will support bold ideas that neither traditional research nor the private sector is willing to pursue, driving new biomedical breakthroughs. At the same time, the Department of Health and Human Services is investing in research and technology that can keep Alzheimer’s patients living longer in their own homes; training caregivers to support them; and educating Americans about early warning signs of Alzheimer’s, dementia risks, and brain health generally. U.S. Government also announced the Inflation Reduction Act, which will protect Alzheimer’s patients from high bills at the pharmacy by capping what they pay at US$ 2,000 per year.
Funding to advance neurodegenerative diseases treatment research
Funding to advance neurodegenerative diseases treatment research can drive the growth of the market. On March 28, 2023, Mayo Clinic, a U.S.-based non-profit academic medical center focused on integrated health care, education, and research received US$ 41 million in new federal funding for three multiethnic projects to identify targets for the treatment of Alzheimer's disease. The new research program seeks to identify the next generation of precision medicine biomarkers and potential novel therapeutic targets of Alzheimer's disease and related dementias in multiethnic populations. The program, called Centrally-Linked Longitudinal Peripheral Biomarkers of AD in Multiethnic Populations (CLEAR-AD), will be funded by a five-year grant from the National Institute on Aging, division of the U.S. National Institutes of Health. The grant is expected to total US$ 41 million.
Global Tauopathies Treatment Market: Restraints
Failure of clinical trials associated with treatment drugs
The failure of clinical trials associated with treatment drugs is expected to hamper the global tauopathies treatment market growth. For instance, on December 21, 2022, new experimental antibody that appears to slow cognitive decline in some Alzheimer’s patients caused death of 79-year-old Florida woman participating in an ongoing trial of the antibody after experiencing extensive brain swelling and bleeding as well as seizures. The death is believed to be likely caused by the antibody, lecanemab. It was concluded that brain swelling and microhemorrhages could be a serious side effect of the study medication by trial investigator.
To counterbalance this restraint, more research should be done to introduce and develop effective drug against neurodegenerative disorders.
Costly treatment development for neurodegenerative diseases
High cost of treatment development of neurodegenerative diseases is expected to hamper the global tauopathies treatment market growth. For instance, according to an article published in Journal Alzheimer's & Dementia, on September 28, 2021, the cumulative private expenditures on clinical stage AD R&D were nearly US$ 42.5 billion, with the greatest costs US$ 24,065 million incurred during Phase 3. Out of 235 agents analyzed, 112 remain in active clinical development, six reached commercialization and 117 had negative outcomes equating 95% failure rate. The information was collected from AD clinical trials (n = 1099; phases 1–4) conducted between January 1, 1995 and June 21, 2021 from various databases.
To counterbalance this restrain, more funding by governments for research activities should be allocated.
Global Tauopathies Treatment Market - Key Players
The major players operating in the global tauopathies treatment market include Avanir Pharmaceuticals, Inc., Otsuka Pharmaceutical Co., Ltd., AB Science, Cortice Biosciences, REGENXBIO Inc., Aquinnah Pharmaceuticals, Neurimmune, Oligomerix, Inc., Biogen, TauRx, Sellas, Catalent Inc., Selvita S.A, Bristol-Myers Squibb Company, Chronos Therapeutics Limited, and SK Biopharmaceuticals Co., Ltd.
*Definition: Tauopathies are clinically, morphologically, and biochemically heterogeneous neurodegenerative diseases that are characterized by the tau protein aggregation in the brain. Currently, there are no approved treatments for tauopathies. The most common disease associated with tauopathy is Alzheimer’s disease. Characteristic symptoms and signs of tauopathies include changes in social behavior, personality, difficulty in making decisions, speaking, poor coordination and balance, dementia, and psychiatric symptoms.
Share
Share
About Author
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Missing comfort of reading report in your local language? Find your preferred language :
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
Select a License Type
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients