The global single use system in biopharma manufacturing market was valued at US$ 7,030.0 Mn in 2021 and is forecast to reach value of US$ 26,450.3 Mn by 2028 at a CAGR of 20.8% between 2022 and 2028. The global single use system in biopharma manufacturing market is experiencing strong growth due to the increase in disease burden and increase in demand for drugs/vaccines across the globe. Moreover, rise in demand for biopharmaceutical products is expected to boost the growth of the market. However, factors such as high-end manufacturing requirements and high cost of manufacturing are expected to hamper the growth of the global single use system in biopharma manufacturing market.
Global Single Use System in Biopharma Manufacturing Market: Regional Insights
Based on geography, the global single use system in biopharma manufacturing market is segmented into North America, Latin America, Europe, Asia Pacific, and Middle East & Africa.
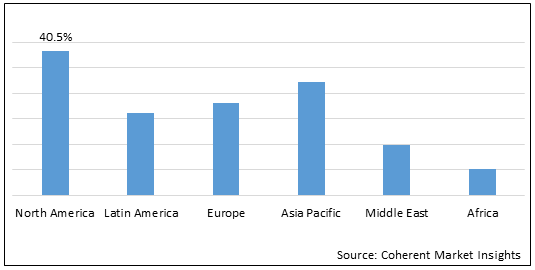
Among regions, North America is expected to gain highest share in the market over the forecast period owing to the expanding biopharmaceutical R&D coupled with the presence of large-scale biopharmaceutical manufacturing facilities in the region, especially in the United States. For instance, in January 2021, Fujifilm planned to invest over 200 Billion yen (US$ 2 Billion) to establish new manufacturing site for biopharmaceuticals in the U.S., as well as to accelerate the growth of its biopharmaceutical contract development and manufacturing business.
Moreover, Asia Pacific is expected to witness robust growth in the global single use system in biopharma manufacturing market due to growing biopharmaceutical manufacturing industry. For instance, in recent years, China and India have emerged as a potential market for single-use technology with growing biopharmaceutical manufacturing industry. In November 2021, WuXi Biologics announced it has successfully launched the GMP operation of its new drug product facility located in Wuxi, China. This facility serves as an example of WuXi Biologics’ implementation of single-use technology.
Figure 1. Global Single Use System in Biopharma Manufacturing Market Share (%), by Region, 2021
To learn more about this report, Download Free Sample
Global Single Use System in Biopharma Manufacturing Market Drivers:
Increasing demand for biopharmaceuticals across the globe is expected to augment growth of the global single use system in biopharma manufacturing market over the forecast period. The huge demand for biopharmaceuticals has been facilitated by increasingly focused R&D related investments. For instance, according to the United Nations Educational, Scientific and Cultural Organization (UNESCO) Institute, the global spending on research and development (R&D) reached a record high of ~USD 1.7 trillion in 2020.
Increasing demand for safe and effective drugs and vaccines worldwide is expected to boost the growth of the global single use system in biopharma manufacturing market over the forecast period. The ability of biopharmaceutical products to address previously untreatable conditions has paved the way for the introduction of innovative drugs into the market. In August 2022, Philanthropist donated US$ 172 million to speed up drug development for the next pandemic. With Funds, research centre in Melbourne will develop technologies to quickly create antiviral treatments such as monoclonal antibodies when new infectious diseases emerge.
Global Single Use System in Biopharma Manufacturing Market Opportunities:
Growing shift towards biopharmaceutical contract manufacturing is expected to offer lucrative growth opportunities for players in the global single use system in biopharma manufacturing market. For instance, in 2020, Pfizer and Gilead Sciences signed an agreement to manufacture and supply ‘Remdesivir’ for the treatment of COVID-19. The agreement includes delivering contract manufacturing services at Pfizer’s McPherson, Kansas facility.
Rising government support towards biosimilar & biological drugs is expected to offer lucrative growth opportunities for players in the global single use system in biopharma manufacturing market. For instance, India has a thriving biosimilar ecosystem in comparison to other countries and because of that Indian pharmaceutical companies have risen as the global market leaders in biosimilars. There are more than 100 biopharmaceutical companies in India.
Single Use System in Biopharma Manufacturing Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2021 | Market Size in 2021: | US$ 7,030.0 Mn |
| Historical Data for: | 2017 to 2020 | Forecast Period: | 2022 to 2028 |
| Forecast Period 2022 to 2028 CAGR: | 20.8% | 2028 Value Projection: | US$ 26,450.3 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Getinge AB, Rentschler Biopharma SE, Kühner AG, Danaher Corporation, Merck KgaA, Lonza, PBS Biotech Inc., ThermoFisher Scientific, Corning Incorporated, Eppendorf, Pall Corporation, Entegris, and Sartorius AG (Sartorius Stedim Biotech), among others |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Global Single Use System in Biopharma Manufacturing Market Trends:
Increase in demand for single use systems around the world is expected to propel growth of the global single use system in biopharma manufacturing market. For instance, in March 2022, Central Research Laboratories launched a new Single-Use Beta Bag product line. Single-use bags are indispensable components in ready-to-use, component-transfer, and waste-handling applications during the manufacturing of Life Science products.
Increase in development of single-use solutions is expected to drive growth of the global single use system in biopharma manufacturing market. For instance, in April 2022, High Purity New England announced the launch of a new manufacturing facility. It will serve as the company's Speciality Manufacturing and Distribution Center and increases its manufacturing capacity to develop single-use systems/solutions.
Global Single Use System in Biopharma Manufacturing Market Restraints:
High-end manufacturing requirements are expected to hamper growth of the global single use system in biopharma manufacturing market. For instance, biopharmaceutical manufacturing is characterized by the use of advanced technologies, harnessing of new scientific advances, and driven by a highly complex research and development (R&D) enterprise.
High cost of manufacturing is expected to hinder growth of the single use system in biopharma manufacturing market. For instance, large-scale biotech-manufacturing facilities require $ 200 million to $ 500 million or more to build, compared with similar-scale small-molecule facilities that may cost just $ 30 million to $ 100 million, and they can take four to five years to build.
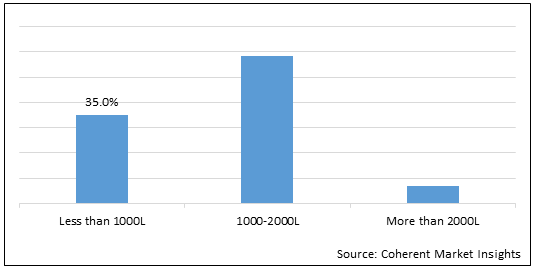
Figure 2. Global Single Use System in Biopharma Manufacturing Market Share (%) in Terms of Value, by Bioreactor, 2021
To learn more about this report, Download Free Sample
Global Single Use System in Biopharma Manufacturing Market Segmentation:
The global single use system in biopharma manufacturing market report is segmented into Bioreactors, Application, Bioprocessing, Product, Modality, Component, and Region.
Based on Bioreactors, the market is segmented into less than 1000L, 1000-2000L, and More than 2000L. Out of which, All Segments are expected to dominate the global single use system in biopharma manufacturing market over the forecast period and this is attributed to the rise in demand for single use biopharmaceuticals across the globe.
Based on Application, the market is segmented into Filtration, Storage (Buffer & Media Storage, Freeze & Thaw), Cell Culture, Mixing, Purification, and Aseptic Filling. Out of which, Filtration Segment is expected to dominate the single use system in biopharma manufacturing market over the forecast period and this is attributed to the rise in usage of single-use filtration assemblies during biopharma manufacturing.
Cell Culture Segment is also expected to witness significant growth in the near future and this is owing to the ability of biopharmaceuticals to treat untreatable diseases. Cell culture is one of the major tools used in cellular and molecular biology, providing excellent model systems for studying the normal physiology and biochemistry of cells, effects of drugs & toxic compounds on the cells, and mutagenesis and carcinogenesis.
Based on Bioprocessing, the global single use system in biopharma manufacturing market is segmented into Small-scale, Mid-scale, and Large-scale. Of which, All Segment are expected to dominate the market over the forecast period and this is attributed to the increase in demand for biopharmaceutical products. Single-use bioprocessing is a rapidly evolving technology used in the development of disposable bioprocessing equipment and accessories to manufacture biopharmaceutical products.
Based on Product, the single use system in biopharma manufacturing market is segmented into Sampling Systems, Bioprocess Containers, Bioreactors, Mixers, Membrane Absorbers, Bottles, Equipment, Transfer Units, and Others (Disposable filter Cartridges (DFC), Depth Filters, Clamps, etc. Out of which, All Segments are expected to dominate the global single use system in biopharma manufacturing market over the forecast period and this is attributed to the increase in focus on the biopharma manufacturing.
Based on Modality, the market is segmented into Protein & Monoclonal Antibody (Mab), Cell Therapy, Gene Therapy, Conventional Vaccine, & mRNA. Of which, Protein & Monoclonal Antibody (Mab) Segment is expected to dominate the global single use system in biopharma manufacturing market over the forecast period and this is attributed to the rise in prevalence of chronic diseases. Monoclonal antibody drugs are treatments that enlist body's germ-fighting immune system against diseases, including cancer.
Gene Therapy Segment is also expected to witness significant growth in the near future and this is owing to the increase in disease burden. Gene therapy replaces the faulty gene or adds a new gene in an attempt to cure disease or improve the body's ability to fight disease.
Based on Component, the market is segmented into Drug Substance and Drug Product. Out of which, Both Segments are expected to dominate the global single use system in biopharma manufacturing market over the forecast period and this is attributed to the increase in demand for safe and effective drugs across the world. Single Use System offers a complete range of solutions for producing, scaling, and transporting drug substances or products.
Global Single Use System in Biopharma Manufacturing Market: Key Developments
In October 2021, Waters Corporation and Sartorius announced partnered to provide bioprocess experts with direct access to high-quality mass spectrometry (MS) data to accelerate the speed and improve the accuracy of biopharmaceutical process development.
In June 2021, Entegris, Inc. announced an investment of around US$ 30 for the expansion & development of manufacturing capability for the production of life sciences. The facilities will develop & manufacture bioprocessing assemblies, such as its Aramus critical fluid handling product line currently used to produce & deliver vaccines worldwide.
In December 2020, Sartorius acquired WaterSep BioSeparations. WaterSep's innovative bio-processing products fit nicely into Sartorius' current gene and cell therapy applications, vaccine production, and intensified bioprocessing solutions.
Global Single Use System in Biopharma Manufacturing Market: Key Companies Insights
The global single use system in biopharma manufacturing market is highly competitive. This is attributed to the rise demand for biopharmaceuticals across the globe, as a result, players in the market are focusing on launching novel products in the market.
Some of the key players in the global single use system in biopharma manufacturing market are Getinge AB, Rentschler Biopharma SE, Kühner AG, Danaher Corporation, Merck KgaA, Lonza, PBS Biotech Inc., ThermoFisher Scientific, Corning Incorporated, Eppendorf, Pall Corporation, Entegris, and Sartorius AG (Sartorius Stedim Biotech), among others.
*Definition: Single use system (SUS) refers to a biopharmaceutical manufacturing equipment designed to be used once (or for a single manufacturing campaign) and then discarded. SUS represent the future in biopharmaceutical processing of therapeutic drugs with significant advantages over traditional reusable stainless-steel systems and partly disposable systems.
Share
Share
Missing comfort of reading report in your local language? Find your preferred language :
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
Select a License Type
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients