Sciatica is a term which is used to describe the symptoms such as leg pain, tingling, numbness, or weakness observed in lower back and travel through sciatic nerve in the back of the leg in individuals. Sciatica most commonly occurs in patients suffering from herniated disk, bone spur on the spine or narrowing of the spine (spinal stenosis), which leads to inflammation, pain, and numbness in the affected leg. Nonsurgical and surgical options for sciatica treatment are available. Nonsurgical techniques are usually attempted initially but when the underlying cause is severe and/or progressive neurological impairments, such as limb weakness, emerge, surgery may be recommended. First line treatments of sciatica typically include some combination of physical therapy, medications, chiropractic therapy, massage therapy, therapeutic injections (epidural steroid injections, selective nerve root blocks injection), and alternative therapies.
The global sciatica market is estimated to be valued at US$ 6,302.9 million in 2021 and is expected to increase to US$ 8,776.2 Mn by 2028, witnessing a CAGR of 4.8% over the forecast period (2021-2028).
Figure 1. Global Sciatica Market Share (%), By Region, 2021
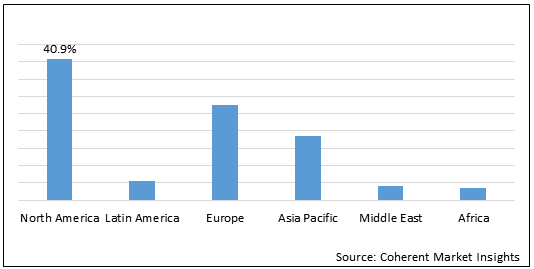
To learn more about this report, Download Free Sample
Robust product pipeline for the treatment of sciatica which are estimated to launch over the forecast period is expected to drive the growth of the global sciatica market.
The key players operating in the global sciatica market are focusing on vigorous product pipeline for the treatment of sciatica which are estimated to launch over the forecast period, is expected to drive the growth of the global sciatica market. For instance, on September 4, 2019, SpineThera Australia Pty Ltd, a wholly owned subsidiary of SpineThera, Inc., initiated a phase I/II clinical trial study to assess the safety and efficacy of two doses of SX600 administered by lumbosacral transforaminal epidural injection in patients with radicular pain secondary to lumbar intervertebral disc herniation. The study is estimated to complete by September 2022.
Sciatica Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2020 | Market Size in 2021: | US$ 6,302.9 Mn |
| Historical Data for: | 2017 to 2020 | Forecast Period: | 2021 to 2028 |
| Forecast Period 2021 to 2028 CAGR: | 4.8% | 2028 Value Projection: | US$ 8,776.2 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Sorrento Therapeutics, Inc., Teva Pharmaceutical Industries Ltd., Aurobindo Pharma Ltd., Zydus Cadila, Glenmark Pharmaceuticals Ltd., Amneal Pharmaceuticals LLC, Jubilant Life Sciences Ltd., Hikma Pharmaceuticals PLC., Mylan N.V., Horizon Therapeutics Plc., Sun Pharmaceutical Industries Ltd., Alkem Labs, SCILEX Pharmaceuticals, Inc., Seikagaku Corporation, Kolon Life Science, Inc., Teijin Limited, and SpineThera, Inc |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Figure 2. Global Sciatica Market Share (%), by Disease Type, 2021
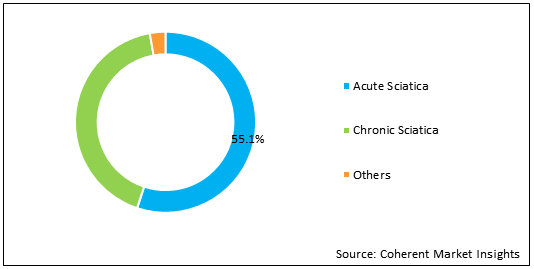
To learn more about this report, Download Free Sample
Global Sciatica Market – Impact of Coronavirus (COVID-19) Pandemic
Since the COVID-19 virus outbreak in December 2019, the disease has spread to over 100 countries across the globe. The Coronavirus (COVID 19) pandemic and lockdown in various countries across the globe have impacted the financial status of businesses in all sectors. Private healthcare sector is one of the sector, which is majorly impacted by the COVID-19 pandemic. The COVID-19 pandemic caused major disruptions to clinical trial executions in the U.S., impacting key stakeholders across the industry. Investigative site capabilities experienced disturbances, driven by staff furloughs, social-distancing protocols, financial losses, and concerns over patient safety. Sponsors, contract research organizations, and other organizations that support drug development shifted to remote working environments. Recently, the U.S. Food and Drug Administration (FDA) guidance (March 2020 and updated on July 2020) acknowledged that the impact of COVID-19 may require companies conducting clinical trials to consider virtual patient visits or include novel ways for conducting clinical trials in place of current protocols. Therefore, there are now more opportunities for using remote healthcare including conducting virtual or decentralized trials, site-less clinical trials, and use of other non-traditional approaches that do not involve in-person visits.
Global Sciatica Market: Restraint
Potential side effects of drugs used in the treatment of sciatica and low patient compliance to the drugs such as anti-depressants and anticonvulsants due to its side effects is expected to restrain the market growth over the forecast period. Adverse events associated with epidural steroid injections include infection, bleeding, dural puncture nerve damage, cardiovascular system (heart) complications, risk associated with local anesthetics, and risk associated with steroids.
Key Players
Major players operating in the global sciatica market include Sorrento Therapeutics, Inc., Teva Pharmaceutical Industries Ltd., Aurobindo Pharma Ltd., Zydus Cadila, Glenmark Pharmaceuticals Ltd., Amneal Pharmaceuticals LLC, Jubilant Life Sciences Ltd., Hikma Pharmaceuticals PLC., Mylan N.V., Horizon Therapeutics Plc., Sun Pharmaceutical Industries Ltd., Alkem Labs, SCILEX Pharmaceuticals, Inc., Seikagaku Corporation, Kolon Life Science, Inc., Teijin Limited, and SpineThera, Inc.
Share
Share
Missing comfort of reading report in your local language? Find your preferred language :
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
Select a License Type
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients