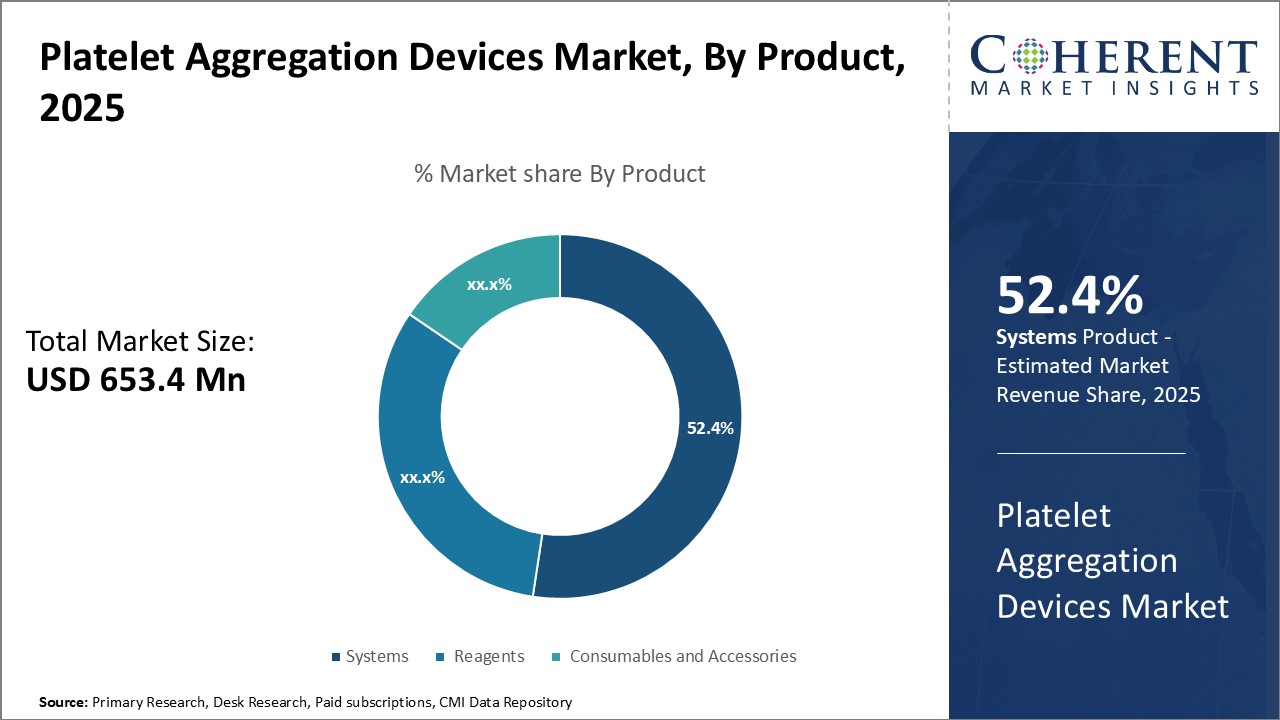
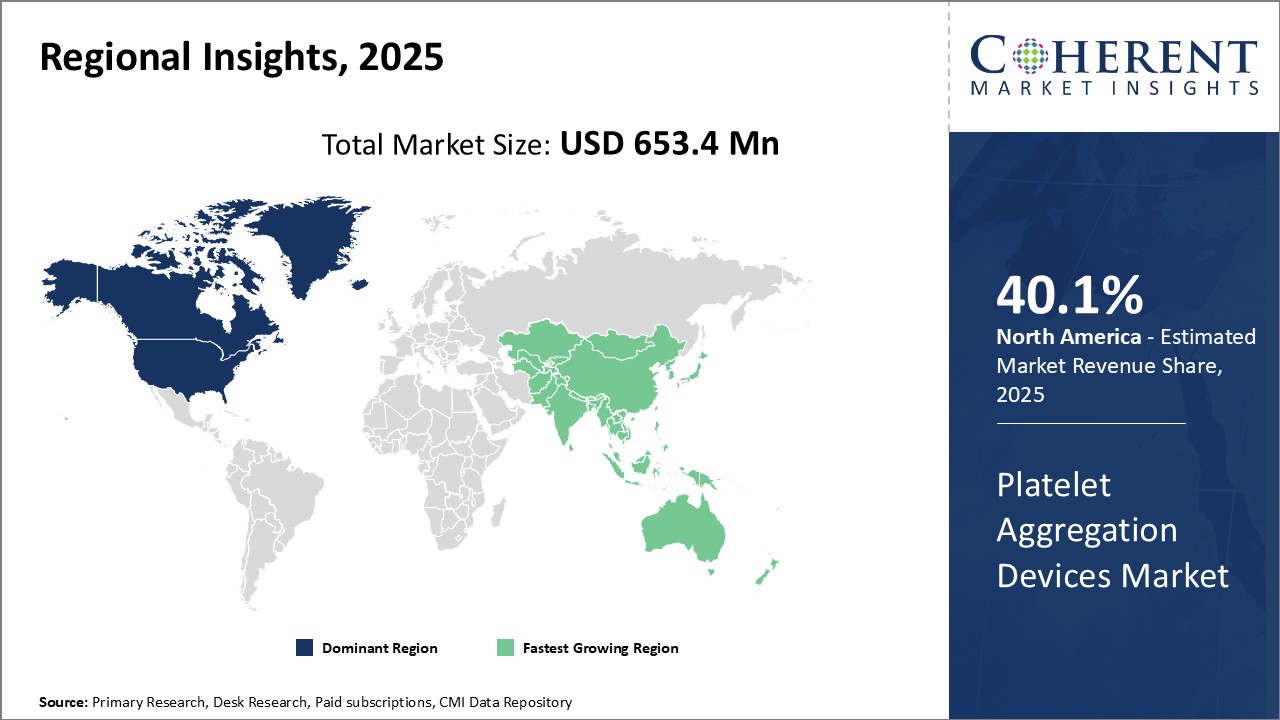
Platelet aggregation devices market is estimated to be valued at USD 653.4 Mn in 2025 and is expected to reach USD 1,036.2 Mn by 2032, exhibiting a compound annual growth rate (CAGR) of 6.8% from 2025 to 2032.
Discover market dynamics shaping the industry: Download Free Sample
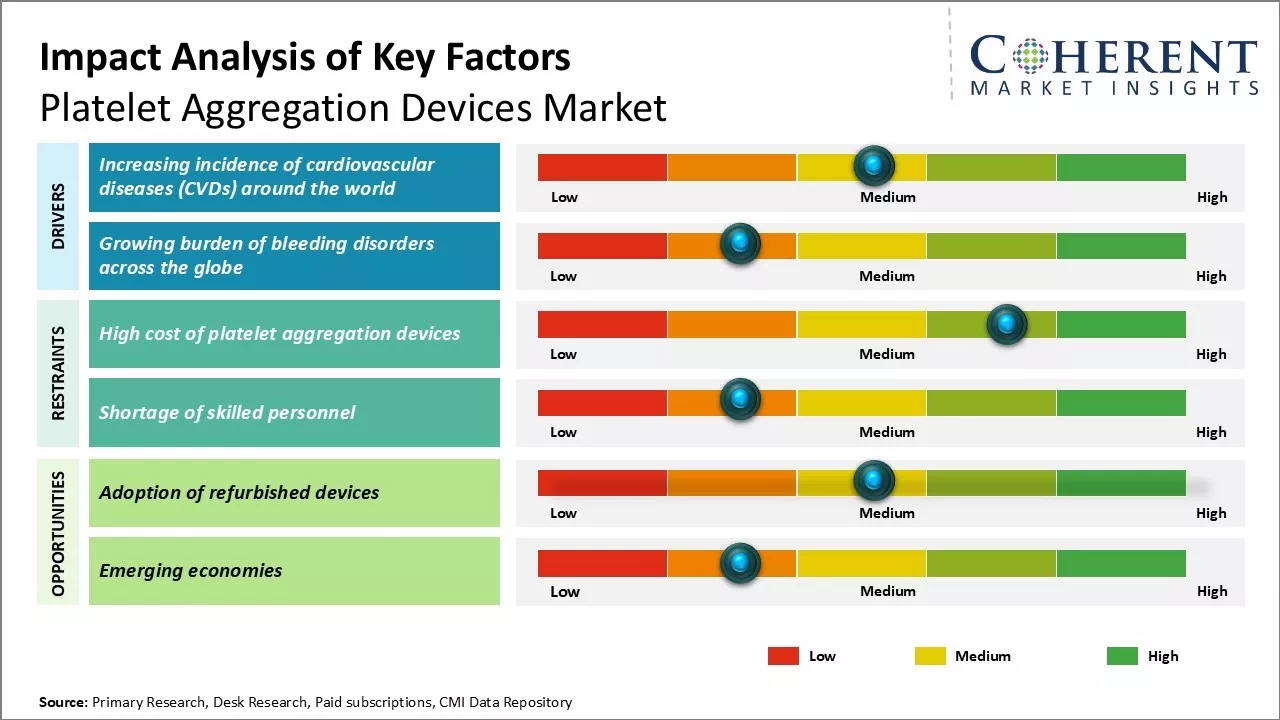
The global platelet aggregation devices market is projected to grow significantly, driven by the increasing prevalence of chronic diseases, technological advancements, and heightened awareness of platelet-related disorders. These devices are crucial for diagnosing conditions related to hemostasis and thrombosis, enabling healthcare professionals to identify defects in platelet function. However, market growth may be restrained by high costs of devices and a shortage of skilled professionals.
Discover high revenue pocket segments and roadmap to it: Download Free Sample
Insights By Product- Systems segment dominates due to their versatility and advanced functionality
The systems segment of the platelet aggregation devices market is projected to hold the largest market share of 52.4% in 2025, attributed to their versatility and advanced functionality. These fully integrated devices automate the entire testing process, from preparation to analysis, providing reproducible results with minimal manual intervention. With features like onboard protocol storage, automated quality control, and customizable reporting options, they streamline workflows and save time for operators.
Insights By Application- The critical need for testing in diagnosis and treatment monitoring drives clinical segment growth
In terms of application, clinical segment is estimated to contribute the highest market share of 63.9% in 2025, primarily due to critical need for platelet function testing in diagnosis and treatment monitoring. This testing is vital for diagnosing bleeding disorders, assessing the efficacy of anti-platelet therapies, and managing surgical risks, making it an essential tool for clinicians treating patients on anti-platelet medications or undergoing invasive procedures.
Insights By End User- Availability of advanced testing facilities and expertise drives the hospitals segment growth
In terms of end user, hospitals segment is estimated to contribute the highest market share of 48.7% in 2025, owing to availability of advanced testing facilities and expertise. Most hospitals have specialized hemostasis and thrombosis laboratories equipped with advanced platelet function analyzers to provide efficacious diagnostic evaluation and management of bleeding disorders as well as cardiovascular patients. These also have expertise in standardized test protocols and interpretation of results.
Need a Different Region or Segment? Download Free Sample
Dominating Region- North America
North America is expected to dominate the market, with an estimated market share of 40.1% in 2025, due to well-established healthcare infrastructure, high healthcare expenditure, and presence of leading market players. The region has witnessed significant technological advancements and adoption of advanced devices.
Fastest-Growing Region- Asia Pacific
Asia Pacific region exhibits the fastest growth, accounting for 22.9% share in 2025, owing to increasing patient population, rising medical tourism, and growing healthcare spending across developing countries. Emerging economies in the region provide lucrative opportunities for market expansion.
Platelet Aggregation Devices Market Outlook for Key Countries
Presence of key market players in the U.S.
The U.S. platelet aggregation devices industry is influenced by the presence of global players. Companies like Aggredyne Inc., a medical diagnostic company in the U.S., plays a crucial role in driving the market growth through continuous innovations. Its advancements in technology and product offerings enhance diagnostic capabilities, addressing the rising demand for effective platelet function testing in clinical settings and contributing to improved patient outcomes.
China’s favorable government initiatives
China platelet aggregation devices industry growth is driven by favorable government initiatives aimed at modernizing the healthcare system. These reforms enhance access to advanced medical technologies and improve overall healthcare delivery. Local manufacturers have solidified their position by leveraging these initiatives, contributing to the increased adoption of platelet aggregation testing and improving patient outcomes across the country.
Availability of low-cost products in India
India is emerging as a leading market for platelet aggregation devices due to the availability of low-cost products that enhance access to essential diagnostic tools. This affordability allows more healthcare facilities to implement platelet function testing, crucial for managing bleeding disorders and cardiovascular diseases. As awareness about these conditions grows, there will be huge demand for effective platelet aggregation devices, further solidifying India's position in the market.
Rising awareness about platelet disorders in Germany
Germany platelet aggregation devices industry is characterized by rising awareness about platelet disorders and the availability of reimbursement options, which significantly enhance access to diagnostic testing. Increased public and professional education about the importance of platelet function testing has led to more diagnoses and treatments. Favorable reimbursement policies encourage healthcare providers to adopt advanced platelet aggregation devices, ultimately improving patient care and outcomes in managing bleeding disorders.

Get actionable strategies to beat competition: Download Free Sample
Top Strategies Followed by Platelet Aggregation Devices Market Players
Emerging Startups in the Platelet Aggregation Devices Market
Key Takeaways from Analyst
Platelet Aggregation Devices Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2024 | Market Size in 2025: | USD 653.4 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2025 To 2032 |
| Forecast Period 2025 to 2032 CAGR: | 6.8% | 2032 Value Projection: | USD 1,036.2 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Sysmex Corporation, WerfenLife SA, Siemens Healthineers, Sentinel Ch. SpA, Haemonetics Corporation, Chrono-Log Corporation, Bio/Data Corporation, Aggredyne Inc., F. Hoffmann-La Roche Ltd., and Helena Laboratories Corporation |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Market Driver- Increasing incidence of cardiovascular diseases (CVDs) around the world
The rising burden of cardiovascular diseases over the past few decades can drive the market growth. According to the WHO, in 2021, cardiovascular diseases (CVDs) are the leading cause of death globally. An estimated 17.9 million people died from CVDs in 2019, representing 32% of all global deaths responsible for over 17 million deaths annually, with 75% occurring in low- and middle-income countries. Rising prevalence of CVDs boosts demand for platelet aggregation devices from healthcare providers.
Market Challenge- High cost of platelet aggregation devices
Platelet aggregation devices market growth can be hampered due to high cost of these devices. This expense makes it difficult for small laboratories, research institutes, and hospitals in developing countries to acquire these devices. The significant capital needed for setup and maintenance hinders the widespread adoption of platelet function testing. Manufacturers must focus on value engineering and innovative financing options to make these devices more accessible.
Market Opportunity- Adoption of refurbished devices
High cost of new platelet aggregation devices creates an opportunity for refurbished or pre-owned models in the market. Hospitals and laboratories upgrading to newer models can sell their well-maintained devices at significantly reduced prices, enabling other users to conduct platelet function testing affordably, as increased acceptance can expand the customer base and boost the adoption of testing methods.
Share
Share
About Author
Komal Dighe is a Management Consultant with over 8 years of experience in market research and consulting. She excels in managing and delivering high-quality insights and solutions in Health-tech Consulting reports. Her expertise encompasses conducting both primary and secondary research, effectively addressing client requirements, and excelling in market estimation and forecast. Her comprehensive approach ensures that clients receive thorough and accurate analyses, enabling them to make informed decisions and capitalize on market opportunities.
Missing comfort of reading report in your local language? Find your preferred language :
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients