Plasmid DNA Manufacturing Market is estimated to be valued at USD 1,353.0 Mn in 2025 and is expected to reach USD 5,600.8 Mn in 2032, exhibiting a compound annual growth rate (CAGR) of 22.5% from 2025 to 2032.
Plasmids are small circular molecules of DNA found in bacteria and other microscopic organisms. Plasmids are physically separated from the chromosomal DNA and replicate independently. They usually have a small number of genes, especially those associated with antibiotic resistance, that are passed from one cell to another. Plasmid DNA can be used directly as therapeutic agents, such as gene therapy and vaccine antigen generation, or indirectly for a variety of research applications such as the use of plasmid DNA as a key starting material for transient transfection.
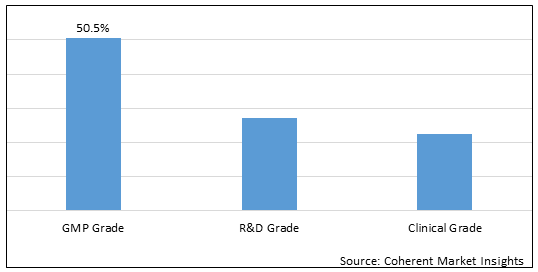
Figure 1.Global Plasmid DNA Manufacturing Market Share (%), by Grade, 2025
To learn more about this report, Download Free Sample
Global Plasmid DNA Manufacturing Market- Driver
The increasing demand for plasmid (pDNA) DNA and increasing research and development activities by key players in the market is expected to drive the market growth over the forecast period. For instance, in October 2022, Forge Biologics, a contract development and manufacturing organization, announced the addition of plasmid DNA manufacturing to its Columbus, Ohio facility. The plasmid DNA manufacturing services encompass three grades which includes research grade, for discovery, research and development, etc.
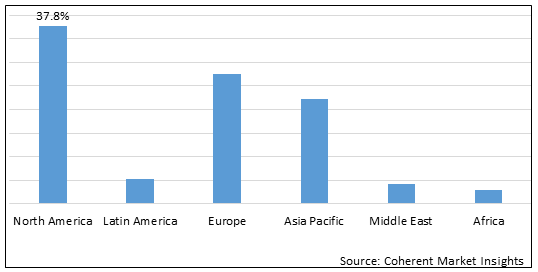
Figure 2. Global Plasmid DNA Manufacturing Market Share (%), by Region, 2025
To learn more about this report, Download Free Sample
The increasing Inorganic strategies such as collaboration by key players in the market to expand product portfolio is expected to drive the market growth over the forecast period. For instance, in September 2022, Lonza, a global manufacturing partner to the pharma, biotech, and nutrition industries, announced its collaboration with Touchlight, a biotechnology company pioneering enzymatic DNA production, to enable the genetic medicine revolution. This collaboration will further help Lonza to integrate an additional, differentiated source of DNA in its end-to-end offering to customers developing messenger RNA therapeutics and vaccines.
Global Plasmid DNA Manufacturing Market– Impact of Coronavirus (COVID-19) Pandemic
Coronavirus disease 2019 (COVID-19) is a highly contagious infectious disease caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), according to World Health Organization (WHO) as of October 10, 2022, 44,614,437 people are affected worldwide.
The key players operating in the market are focusing on development of DNA vaccines for the treatment of Covid -19. For instance, in May 2020, AGC Biologics, a global contract development and manufacturing organization (CDMO) for biopharmaceuticals, partnered with Takara Bio, the biomedical business unit of Takara Shuzo Co., Ltd., to develop vaccine for COVID-19. As a result of this partnership, AGC Biologics is manufacturing the plasmid DNA intermediate vaccine for treatment of COVID-19 with Takara Bio.
Global Plasmid DNA Manufacturing Market: Key Developments
On October 10, 2022, Ray Therapeutics, a biotechnology company developing optogenetic gene therapies for patients with retinal degenerative conditions, and Forge Biologics, a gene therapy-focused contract development and manufacturing organization, announced further collaboration for their manufacturing partnership to include clinical stage plasmid DNA production to support Ray Therapeutics’ lead optogenetics gene therapy program, RTx-015, in clinical trials for patients with retinitis Pigmentosa. By adding clinical grade plasmid production to their existing suite of associated viral vector (AAV) manufacturing capabilities, Forge is easing the scope of production and accelerating the development of lead therapeutic.
On August 26, 2021, INOVIO, a biotechnology company announced that it had received regulatory authorization from Brazil's ANVISA (Agência Nacional de Vigilância Sanitária), the national health regulatory agency of Brazil, to initiate the global Phase 3 segment of its Phase 2/3 trial, INNOVATE (INOVIO INO-4800 Vaccine Trial for Efficacy), for INO-4800, its DNA vaccine candidate for COVID-19. INOVIO plans to conduct the global INNOVATE Phase 3 segment in multiple countries, including Brazil, with partner Advaccine Biopharmaceuticals Suzhou Co., Ltd. (Advaccine).
Plasmid DNA Manufacturing Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2024 | Market Size in 2025: | USD 1,353.0 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2025 To 2032 |
| Forecast Period 2025 to 2032 CAGR: | 22.5% | 2032 Value Projection: | USD 5,600.8 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Cobra Biologics and Pharmaceutical Services (Charles River Laboratories), VGXI, Inc., Aldevron (Danaher), Kaneka Corporation, Nature Technology, PlasmidFactory GmbH & Co. KG, Cell and Gene Therapy Catapult, LakePharma, Inc., MeiraGTx Limited, Eurofins Genomics, Vigene Biosciences, Luminous BioSciences (LBS), LLC, GenScript, GENEWIZ, Creative Biogene, Akron Biotech, Biomay, JAFRAL Ltd., WuXi Biologics, GeneImmune Biotechnology Corp., Lonza, Greenpak Biotech Ltd., Luina Bio (AcuraBio), Ajinomoto Bio-Pharma, Synbio Technologies, Genopis Inc., Altogen Biosystems, Puresyn, Inc., Cepham Life Sciences, Catalent, Inc., Biomiga, Waisman Biomanufacturing |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Global Plasmid DNA Manufacturing Market: Restraint
The high cost of gene therapies is adversely affecting the market growth. For instance, according to an article published by PubMed in May 2021, an upfront price of US$ 2.125 million, the one-time gene therapy onasemnogene abeparvovec for spinal muscular atrophy, a rare neuromuscular disorder that is usually fatal by 2 years of age if untreated, has been called the "most expensive drug ever”. Therefore, it is essential to lower the prices quoted by companies in order to increase the market reach.
Global Plasmid DNA Manufacturing Market: Key Players
Major players operating in the global plasmid DNA manufacturing market include Cobra Biologics and Pharmaceutical Services (Charles River Laboratories), VGXI, Inc., Aldevron (Danaher), Kaneka Corporation, Nature Technology, PlasmidFactory GmbH & Co. KG, Cell and Gene Therapy Catapult, LakePharma, Inc., MeiraGTx Limited, Eurofins Genomics, Vigene Biosciences, Luminous BioSciences (LBS), LLC, GenScript, GENEWIZ, Creative Biogene, Akron Biotech, Biomay, JAFRAL Ltd., WuXi Biologics, GeneImmune Biotechnology Corp., Lonza, Greenpak Biotech Ltd., Luina Bio (AcuraBio), Ajinomoto Bio-Pharma, Synbio Technologies, Genopis Inc., Altogen Biosystems, Puresyn, Inc., Cepham Life Sciences, Catalent, Inc., Biomiga, Waisman Biomanufacturing
Share
Share
About Author
Abhijeet Kale is a results-driven management consultant with five years of specialized experience in the biotech and clinical diagnostics sectors. With a strong background in scientific research and business strategy, Abhijeet helps organizations identify potential revenue pockets, and in turn helping clients with market entry strategies. He assists clients in developing robust strategies for navigating FDA and EMA requirements.
Missing comfort of reading report in your local language? Find your preferred language :
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
Select a License Type
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients