Global Peripheral Vascular Devices Market size is estimated to be valued at USD 13.51 Bn in 2025 and is expected to reach USD 21.00 Bn by 2032, exhibiting a compound annual growth rate (CAGR) of 6.5% from 2025 to 2032.
To learn more about this report, Download Free Sample
Global Peripheral Vascular Devices Market is expected to witness positive growth over the forecast period due to rising geriatric population globally which are more prone to developing peripheral artery diseases (PAD). Moreover, increasing prevalence of lifestyle diseases such as diabetes and obesity are also contributing to the high risk of peripheral vascular disorders. Growing adoption of minimally invasive endovascular procedures over complex open surgeries boosts demand for peripheral vascular devices. However, high costs associated with peripheral vascular devices may hamper market growth to a certain extent over the coming years.
|
Event |
Description and Impact |
|
Technological Advancements and Product Launches |
|
|
Geopolitical Trade Policy Shifts |
|
|
Clinical Trial Advancements |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
The peripheral vascular devices pipeline is advancing rapidly, focusing on drug-eluting technologies, bioresorbable scaffolds, and next-gen delivery systems. In late-stage trials, Becton Dickinson's Lutonix 035 DCB and Medtronic’s IN.PACT Admiral DCB target complex and below-the-knee lesions. Boston Scientific's Eluvia Stent and Cook Medical’s Zilver PTX are undergoing extended follow-up studies.
In Phase II, Abbott’s Absorb BVS 1.1 and Biotronik’s DREAMS 3G lead bioresorbable scaffold trials, while Boston Scientific’s Ranger DCB and QT Vascular’s Chocolate Touch DCB explore broader use cases. Bentley’s BeGraft Stent Graft targets complex disease.
Early-stage devices include Selution SLR and MagicTouch PTA (sirolimus DCBs), Magnitude and Fantom Encore bioresorbable stents, and Boston Scientific’s Jetstream Navitus. Avinger’s Avenger Catheter focuses on real-time imaging and lesion crossing.
Preclinical innovations involve nano-coated balloons, gene-eluting stents, and AI-guided navigation tools. Key players like Medtronic, Boston Scientific, Abbott, and BD are driving this innovation, with strong R&D pipelines aimed at improving outcomes in complex PAD cases.
The peripheral vascular devices market exhibits a dynamic and competitive patent landscape, driven by continuous innovation in drug delivery, stent design, and scaffold technology. As of 2025, over 300 active patents globally relate to drug-coated balloons (DCBs), drug-eluting stents (DES), and bioresorbable scaffolds. Leading companies such as Boston Scientific, Medtronic, Abbott, and Becton Dickinson have significantly expanded their intellectual property portfolios, focusing on polymer coatings, controlled drug release mechanisms, and flexible scaffold structures.
Notably, Boston Scientific has secured recent patents for advanced balloon-expandable delivery systems, while Medtronic holds a strong position in drug-layering technologies. Patent filings from Asia-Pacific innovators—particularly in China and India—are rising, reflecting growing R&D capabilities and market ambitions in these regions.
Several first-generation stent patents are approaching expiry, encouraging the emergence of generic device manufacturers, particularly in cost-sensitive markets. Meanwhile, academic institutions and medtech startups are exploring novel IP areas such as gene-eluting stents and AI-integrated intervention systems.
Overall, the patent ecosystem is shifting toward next-generation platforms that emphasize biocompatibility, durability, and procedural precision, shaping long-term market differentiation and competitive advantage.
The global reimbursement landscape for peripheral vascular devices is diverse and deeply influences market access, pricing strategies, and device adoption. In established markets such as the United States, Europe, and Japan, reimbursement frameworks are well-defined, while emerging markets continue to evolve their policies.
In the U.S., reimbursement is driven by CPT and HCPCS codes covering angioplasty (e.g., 37220–37235), stenting (37236–37239), and balloon catheters (C2617). Procedures are reimbursed under Diagnosis-Related Groups (DRGs) and Medicare, which supports around 64.3 million beneficiaries. Medicare covers ~80% of eligible procedures, while Medicare Advantage plans offer broader coverage (85–95%) but often require prior authorization.
Europe follows country-specific systems under the EU MDR 2017/745. Agencies like NICE (UK), HAS (France), and G-BA (Germany) evaluate both clinical and economic aspects for device approval and reimbursement. HTAs play a pivotal role in market entry and pricing negotiations.
In Japan, the PMDA regulates reimbursement under its National Health Insurance, with fast-track programs like Sakigake for novel devices. Australia manages reimbursement through the Prostheses List and Medicare Benefits Schedule.
Globally, the push for value-based care and cost containment is prompting stricter coverage evaluations, highlighting the importance of real-world evidence and clinical value.
Prescriber choices for peripheral vascular devices are increasingly shaped by disease severity, anatomical complexity, and the demand for minimally invasive solutions. The Rutherford classification system is widely used to guide treatment decisions. For early-stage PAD (Categories 0–1), conservative therapy and pharmacological management dominate. Device interventions become more common in moderate to severe claudication (Categories 2–3), often initiated with diagnostic angiography. In critical limb ischemia (Categories 4–6), 78% of clinicians prefer an endovascular-first approach, emphasizing revascularization.
Drug-coated balloons (DCBs) are highly favored for femoropopliteal and below-the-knee lesions. Top choices include Medtronic IN.PACT Admiral™, BD Lutonix™ 035, and Boston Scientific Ranger™. For stenting, Abbott Supera™ leads for complex lesions, Boston Scientific Eluvia™ for long segments, and Medtronic Complete SE™ for iliac arteries.
Atherectomy devices are used selectively. Boston Scientific SilverHawk™ and Medtronic HawkOne™ are preferred for directional plaque removal, while Diamondback 360°® is used for heavily calcified lesions.
When endovascular therapy fails, bypass surgery becomes the second-line option, particularly for long occlusions or unsuitable vessel anatomy. Overall, prescribers prioritize patency, procedural safety, and limb salvage outcomes in device selection.
To learn more about this report, Download Free Sample
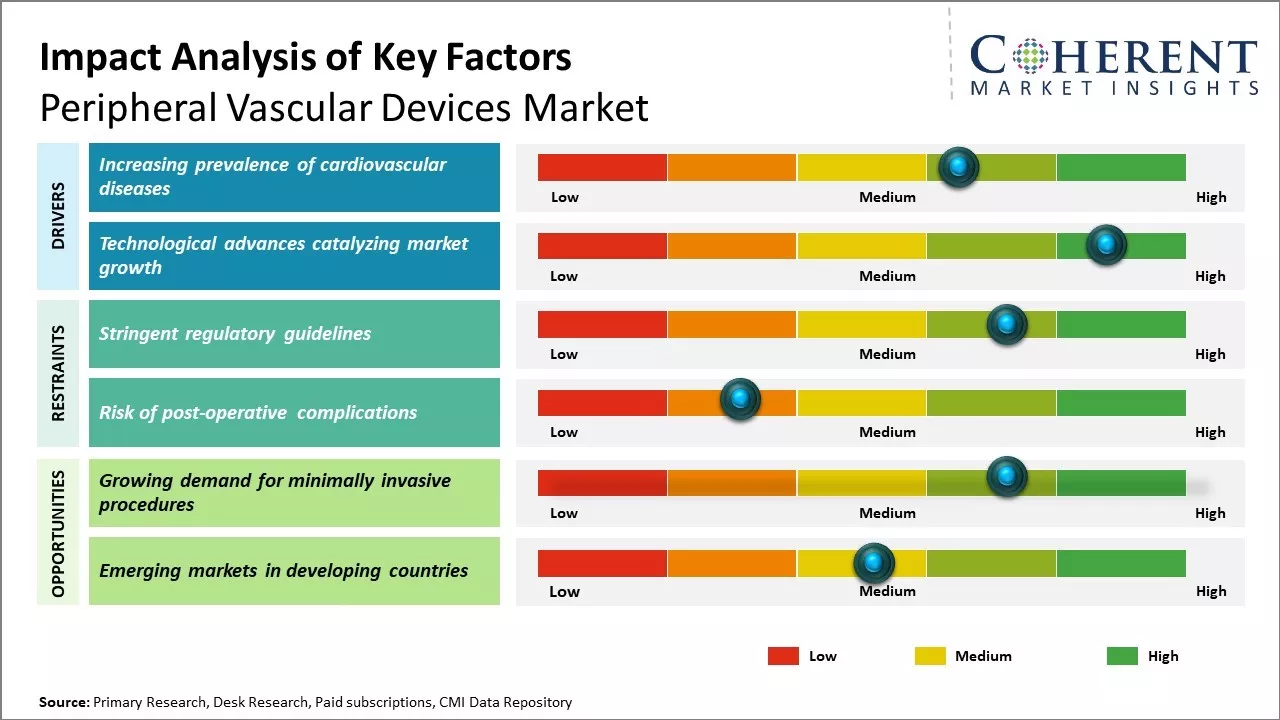
Rising prevalence of cardiovascular diseases has emerged as a key driver bolstering growth of the peripheral vascular devices market. Cardiovascular diseases such as arteriosclerosis, peripheral artery disease (PAD), and other atherosclerotic conditions that obstruct blood circulation to limbs are increasingly common worldwide.
Growing geriatric population base has further added to the rising disease incidence given the increased susceptibility of cardiovascular disorders with advancing age. PAD is characterized by a blockage in arteries other than the heart, restricting blood flow to limbs. This has driven uptake of devices like angioplasty balloons, stents, embolic protection devices, atherectomy devices, and chronic total occlusion devices. The need for minimally invasive treatment options has further bolstered adoption.
Continual technological advancements are responsible for fueling demand for next-generation peripheral vascular devices. Strategic investments by device manufacturers towards the development of technologically superior products with enhanced efficacy, safety and cost-effectiveness have broadened the horizon for peripheral vascular procedures globally.
For instance, industry giants are focusing on nanotechnologies and bioabsorbable materials to develop drug-eluting stents, bioresorbable scaffolds, and other innovative stents. These novel product lines are delivering superior clinical outcomes and reducing complications arising from metal allergy as compared to conventional metallic stents.
Moreover, the advent of advanced imaging modalities like optical coherence tomography (OCT) has enabled improved visualization during procedures and assessment of the outcomes. OCT facilitates high-resolution, three-dimensional cross-sectional imaging to help physicians optimize device placement.
The increasing demand for procedures that are less invasive and allow for quicker recovery presents a major opportunity for companies in the peripheral vascular devices market. Over the past decade, there has been significant shift towards minimally invasive surgeries as patients prefer techniques that minimize incisions, scarring and pain while reducing recovery time. Procedures such as balloon angioplasty, stenting and catheter-based therapy have gained popularity as alternatives to open surgeries for conditions affecting arteries and veins.
Product type segment is sub-segmented into peripheral vascular stents, peripheral transluminal angioplasty balloon catheters, PTA guidewires, atherectomy devices, chronic total occlusion devices, aortic stents, synthetic surgical grafts, and others. Peripheral cardio vascular stents segment is anticipated to hold 25.7% of the market share in 2025. Peripheral artery disease, or PAD, affects people worldwide and causes decreased blood flow and impaired function in the legs.
A large contributing factor to PAD's prevalence is the growing rates of risk factors like diabetes, smoking, obesity, and old age. When lifestyle changes and medication are insufficient to manage PAD symptoms, minimally invasive procedures using peripheral stents are often the standard of care. Stents are small expandable tubes that are placed inside narrowed or blocked arteries to help keep them open and improve blood flow. Compared to previous treatment methods, stents provide better and longer-lasting outcomes while reducing recovery times for patients.
End user segment is sub-segmented into hospitals, specialty clinics, ambulatory surgical centers, and others. Hospitals segment is estimated to contribute the highest share of the peripheral vascular devices market and is projected to hold 48.1% of the market share in 2025. Their centralized operating rooms, specialized clinical staff, and capacity for complex cases make hospitals the natural focal point for performing revascularization procedures.
Most patients with advanced PAD that require angioplasty, stenting, or surgical bypass end up undergoing their treatment in a hospital setting rather than ambulatory centers.
A major driver of hospital-based care is the rising prevalence of comorbidities among PAD patients. As more individuals live with diabetes, chronic kidney disease, and cardiac issues, their peripheral ischemia is often only one part of complex multi-system diseases. Hospitals can best manage comorbidity-related risks that may arise intra- or post-operatively.
Their added capacity for intensive post-op care also assures patients adequate monitoring as they recover. Reimbursement structures further motivate hospitals to serve as hubs for PAD care. Procedure volumes are important for maintaining reimbursement levels as hospital reimbursement shifts to value-based payment model.
To learn more about this report, Download Free Sample
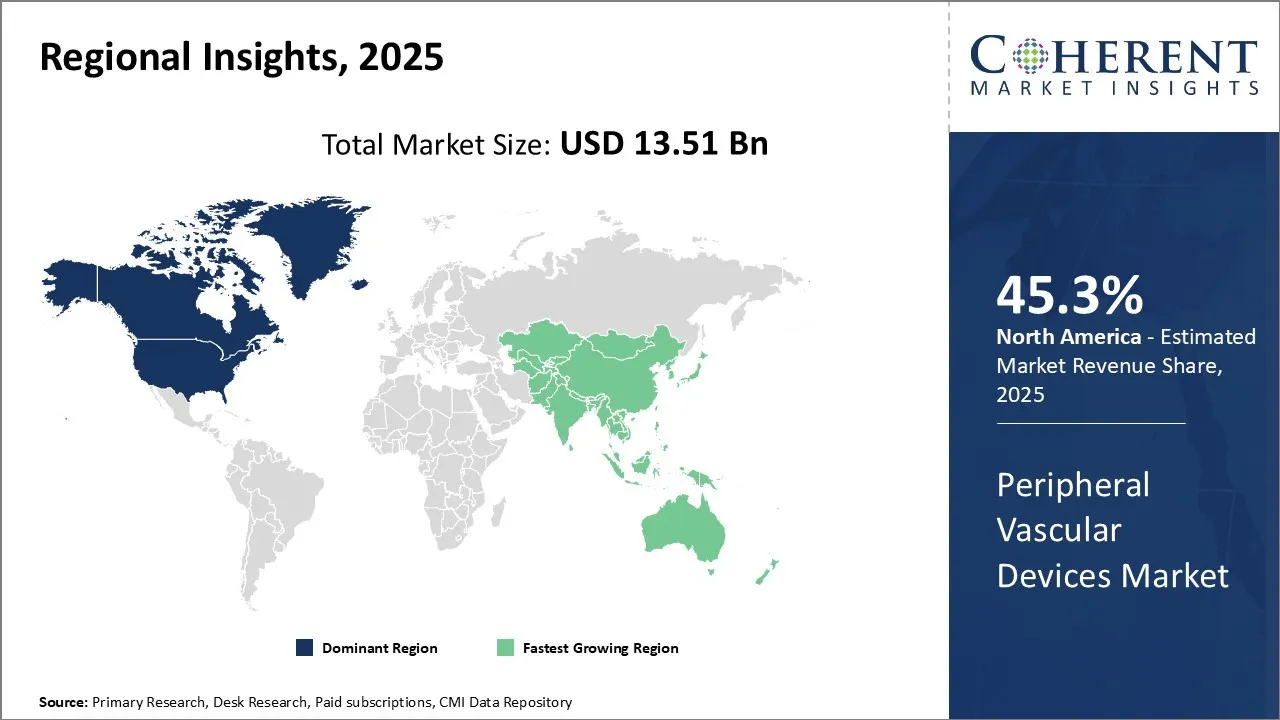
North America is projected to dominate the global peripheral vascular devices market with an estimated 45.3% share in 2025, supported by a well-developed healthcare infrastructure and the strong presence of leading industry players. Major companies such as Medtronic, Boston Scientific, and Abbott Vascular—headquartered in the U.S.—invest significantly in R&D to introduce cutting-edge vascular technologies, reinforcing the region’s leadership.
Favorable reimbursement frameworks from both public and private payers facilitate the adoption of new devices, while relatively high procedure volumes and premium pricing further boost revenue generation. These factors collectively position North America as a critical hub for innovation, commercialization, and market growth.
Asia Pacific is rapidly emerging as the fastest-growing market for peripheral vascular devices, driven by a rising prevalence of peripheral artery disease and expanding healthcare access across key countries like China and India. In China, government initiatives such as Healthy China 2030 aim to modernize healthcare delivery and increase affordability, fostering broader adoption of advanced medical devices.
Global manufacturers are expanding their footprint in the region through local production and distribution networks, while domestic players are accelerating R&D investments. Despite challenges such as high import duties, growing foreign direct investment and expanding trade volumes indicate strong market potential and increasing international interest.
The United States leads the global peripheral vascular devices market, supported by a robust healthcare system, strong clinical demand, and a high rate of device innovation. U.S.-based medical technology firms continually expand their portfolios through next-generation stents, catheters, and drug-coated balloons, backed by favorable FDA pathways and extensive payer coverage.
China plays a pivotal role in Asia Pacific’s market expansion, with rising disease prevalence and healthcare reforms driving demand for vascular interventions. Strategic government policies, expanding middle-class healthcare spending, and increased multinational investment are transforming China into both a consumption and manufacturing powerhouse for peripheral vascular devices.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2024 | Market Size in 2025: | USD 13.51 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2025 To 2032 |
| Forecast Period 2025 to 2032 CAGR: | 6.5% | 2032 Value Projection: | USD 21.00 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
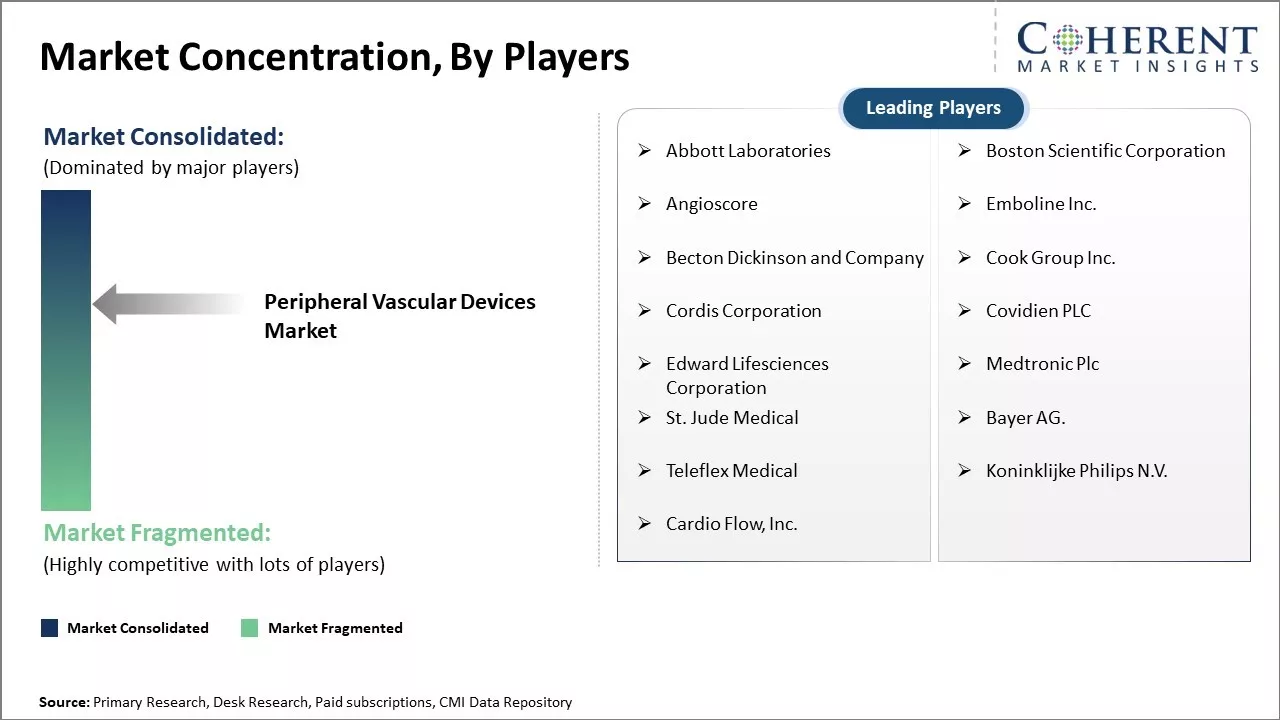
Abbott Laboratories, Boston Scientific Corporation, Angioscore, Emboline Inc., Becton Dickinson and Company, Cook Group Inc., Cordis Corporation, Covidien PLC, Edward Lifesciences Corporation, Medtronic Plc, St. Jude Medical, Bayer AG., Teleflex Medical, Koninklijke Philips N.V., Cardio Flow, Inc. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
*Definition: Peripheral Vascular Devices Market includes medical devices that are used for diagnosis and treatment of peripheral vascular diseases. Some key products in this market are peripheral stents, inferior vena cava filters, angioplasty balloons, angioplasty guidewires, atherectomy devices, venous stents, embolic protection devices, etc. These devices are used by interventional cardiologists and vascular surgeons to open blocked arteries and veins, treat aneurysms, repair damaged vessels, and address other peripheral diseases.
Share
Share
About Author
Komal Dighe is a Management Consultant with over 8 years of experience in market research and consulting. She excels in managing and delivering high-quality insights and solutions in Health-tech Consulting reports. Her expertise encompasses conducting both primary and secondary research, effectively addressing client requirements, and excelling in market estimation and forecast. Her comprehensive approach ensures that clients receive thorough and accurate analyses, enabling them to make informed decisions and capitalize on market opportunities.
Missing comfort of reading report in your local language? Find your preferred language :
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients