The pediatric interventional cardiology market is estimated to be valued at USD 3.18 Bn in 2025 and is expected to reach USD 6.41 Bn by 2032, exhibiting a compound annual growth rate (CAGR) of 10.5% from 2025 to 2032.
Discover market dynamics shaping the industry: Download Free Sample
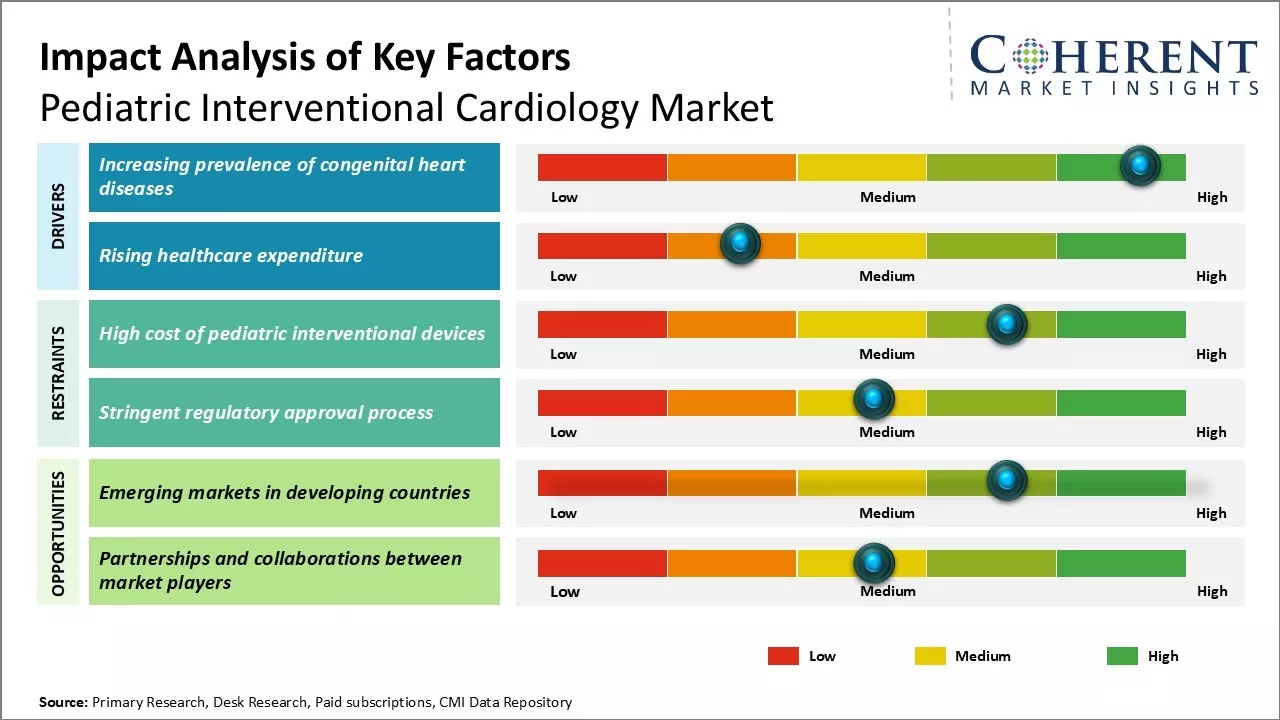
The rising prevalence of congenital heart diseases among infants and young children is a key factor driving the demand for pediatric interventional cardiology procedures. According to the Centers for Disease Control and Prevention, congenital heart defects affect nearly 1% of all newborns in the U.S. each year. Technological advancements in devices and equipment for pediatric cardiology interventions have further enhanced clinical outcomes for affected pediatric patients. This has created a conducive environment for market growth.
Discover high revenue pocket segments and roadmap to it: Download Free Sample
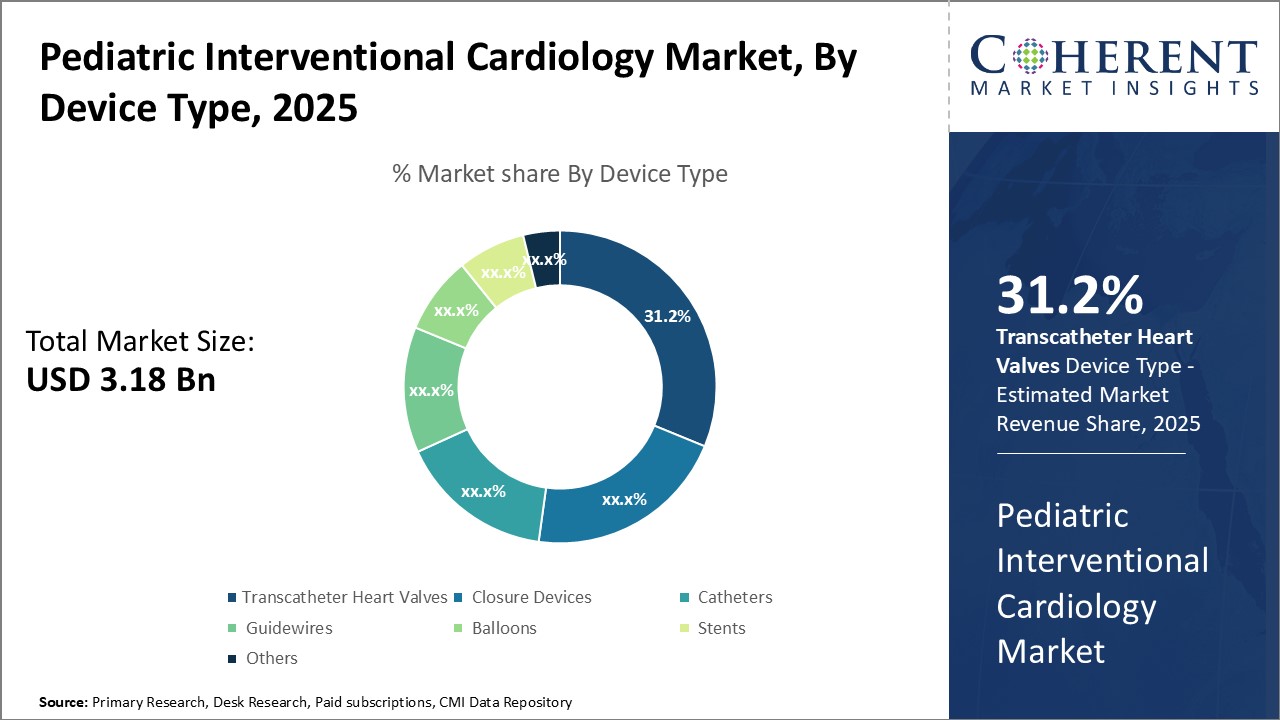
Insights By Device Type - Growing minimally invasive procedure demand is boosting the transcatheter heart valves segment
In terms of device type, transcatheter heart valves segment is expected to contribute the highest market share of 31.2% in 2025, due to increasing demand for minimally invasive procedures among pediatric patients. These valves facilitate valve replacement without open-heart surgery, reducing complications, scarring, and recovery time, making them suitable for high-risk infants who may not tolerate traditional surgery.
Insights By Procedure - Prevalence of congenital heart defects drives angioplasty segment growth
In terms of procedure, angioplasty segment is expected to contribute the highest market share of 44.3% in 2025, owing to the high prevalence of congenital heart defects. Angioplasty is used to open narrowed or blocked blood vessels caused by conditions like pulmonary valve stenosis. The rising incident rates of congenital heart anomalies drives the need for angioplasty procedures to repair defects without open heart surgery.
Insights By End User – High demand expected from hospitals as cardiac care expands
In terms of end user, hospitals segment is expected to contribute the highest market share of 61.9% in 2025, due to their capacity to deliver comprehensive cardiac care. Multi-specialty hospitals provide advanced facilities, specialized teams, and essential support for complex pediatric interventions, making them the primary setting for these critical procedures.
Need a Different Region or Segment? Download Free Sample
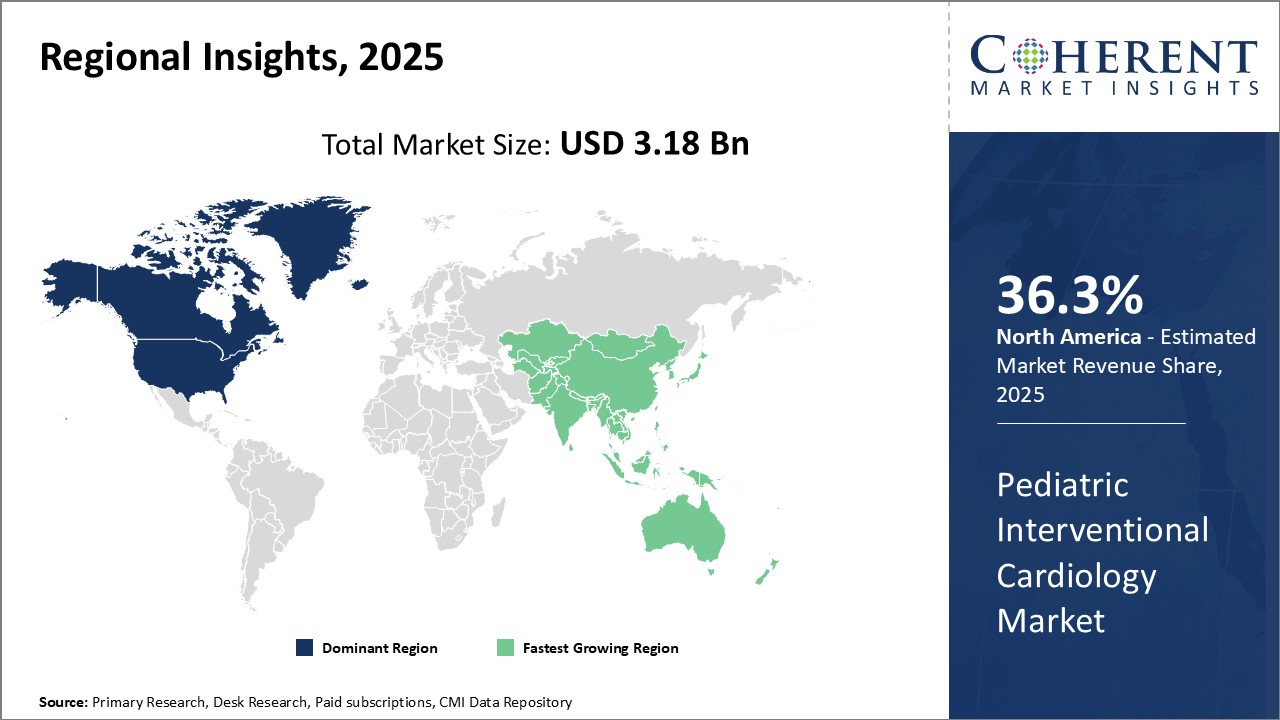
Dominating Region: North America
North America is expected to hold the highest market share of 36.3% in 2025. The dominance can be attributed to a strong presence of leading medical device companies and advanced healthcare infrastructure in countries like the U.S.
Fastest-Growing Region: Asia Pacific
Asia Pacific region exhibits the fastest growth with 19.6% market share in 2025, driven by factors like rising medical tourism, growing private healthcare spending, and increasing focus of market players on expanding their presence beyond developed markets. Countries such as India and China have emerged as top destinations for pediatric cardiac procedures owing to their large patient pools and improving technical expertise.
Pediatric Interventional Cardiology Market Outlook for Key Countries
Investment by key market players in the U.S.
The pediatric interventional cardiology industry in the U.S. is greatly enhanced by substantial investments in innovation from industry leaders like Boston Scientific Corporation. Their focus on developing minimally invasive treatment options is transforming the landscape, improving patient outcomes, and expanding access to advanced cardiac care for children.
Japan’s government initiatives prioritizing pediatric healthcare
Japan pediatric interventional cardiology industry is experiencing steady growth, driven by government initiatives prioritizing pediatric healthcare and promoting advanced medical technologies. These efforts enhance access to specialized care, improve treatment outcomes, and encourage innovation in medical devices, ensuring that children receive the best possible cardiac interventions.
India’s expansion in cardiac care for children
India pediatric interventional cardiology market is rapidly expanding as super-specialty hospitals enhance their capabilities to deliver high-quality cardiac care for children. This growth is attracting patients from across Asia Pacific and the Middle East, drawn by advanced treatments and specialized services that cater to complex pediatric heart conditions.
China’s expanding access through national medical insurance coverage
China pediatric interventional cardiology market is gaining momentum, fueled by the government's commitment to expanding access through national medical insurance coverage and fostering public-private partnerships. These initiatives are establishing centers of excellence for pediatric cardiology, enhancing care quality and attracting patients from neighboring regions seeking specialized treatments.
Get actionable strategies to beat competition: Download Free Sample
Top Strategies Followed by Pediatric Interventional Cardiology Market Players
Emerging Startups in the Pediatric Interventional Cardiology Market
Key Takeaways from Analyst
Pediatric Interventional Cardiology Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2024 | Market Size in 2025: | USD 3.18 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2025 To 2032 |
| Forecast Period 2025 to 2032 CAGR: | 10.5% | 2032 Value Projection: | USD 6.41 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
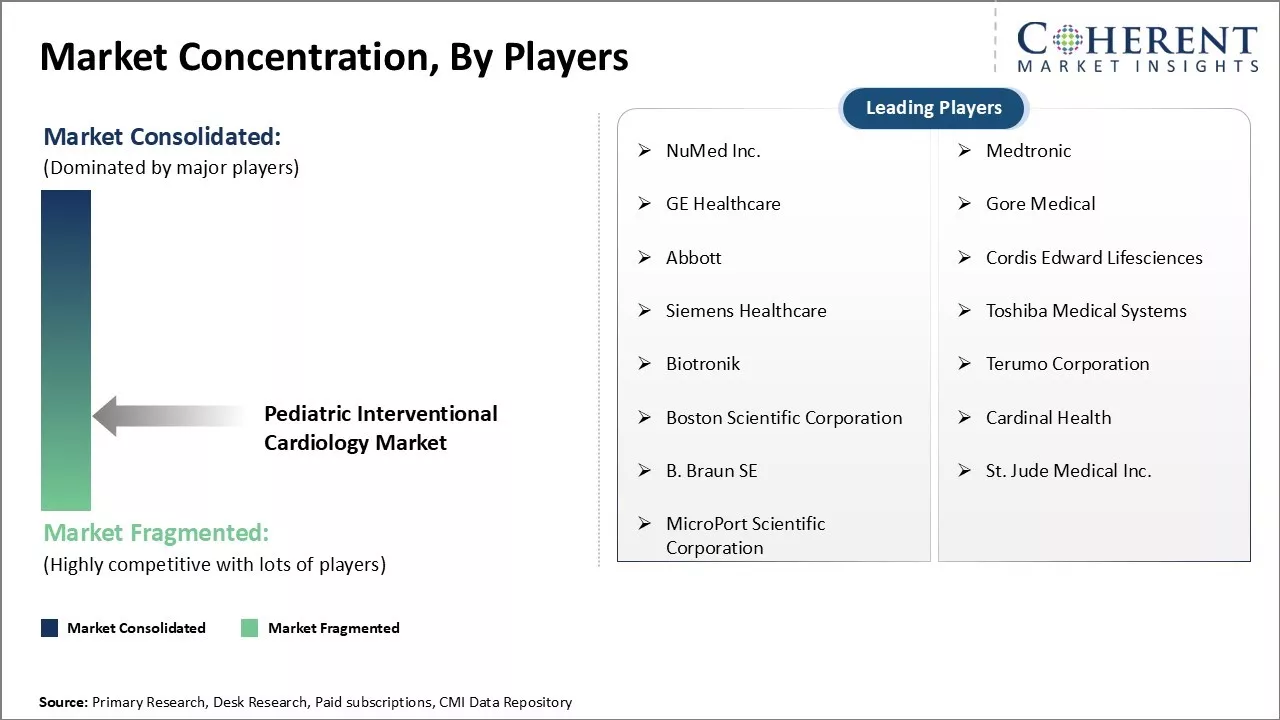
NuMed Inc., Medtronic, GE Healthcare, Gore Medical, Abbott, Cordis Edward Lifesciences, Siemens Healthcare, Toshiba Medical Systems, Biotronik, Terumo Corporation, Boston Scientific Corporation, Cardinal Health, B. Braun SE, St. Jude Medical Inc., and MicroPort Scientific Corporation |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Market Driver - Increasing prevalence of congenital heart diseases
The increasing prevalence of congenital heart diseases drives the market growth. On October 18, 2024, according to the Centers for Disease Control and Prevention, the number of children born each year with congenital heart defects is steadily increasing, affecting nearly 1% of live births in the U.S., which translates to over 40,000 infants annually. Factors contributing to this rise include increasing maternal age, lifestyle changes, environmental pollution, and genetic disorders passed through generations.
Market Challenge - High cost of pediatric interventional devices
One of the major challenges in the pediatric interventional cardiology market is the high cost of specialized devices like cardiac catheters and occluders. These devices must be smaller than adult versions, necessitating extensive research and design modifications, which increase manufacturing costs. Additionally, low production volumes due to a smaller patient pool leads to higher prices, making affordability a significant barrier, especially in developing countries.
Market Opportunity - Emerging Markets in Developing Countries
Emerging economies present a significant opportunity for the pediatric interventional cardiology industry. Despite a similar global prevalence of congenital heart diseases, many children in developing nations lack access to advanced cardiac care due to inadequate infrastructure. As these economies grow, healthcare investments are increasing, allowing specialized centers to reach rural areas. This affluence enhances patients' ability to afford therapies, enabling medical device companies to introduce affordable products and innovative reimbursement models tailored for these markets.
Share
Share
About Author
Komal Dighe is a Management Consultant with over 8 years of experience in market research and consulting. She excels in managing and delivering high-quality insights and solutions in Health-tech Consulting reports. Her expertise encompasses conducting both primary and secondary research, effectively addressing client requirements, and excelling in market estimation and forecast. Her comprehensive approach ensures that clients receive thorough and accurate analyses, enabling them to make informed decisions and capitalize on market opportunities.
Missing comfort of reading report in your local language? Find your preferred language :
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients