The global nasal polyps treatment market is estimated to be valued at USD 3.65 Billion in 2025 and is expected to reach USD 6.19 Billion by 2032, exhibiting a compound annual growth rate (CAGR) of 7.8% from 2025 to 2032.
Discover market dynamics shaping the industry: Download Free Sample
The global nasal polyps treatment market is projected to experience positive growth driven by the rising prevalence of chronic sinusitis and nasal polyps worldwide. Increasing adoption of biologic therapies, such as dupilumab, and advancements in minimally invasive surgical techniques are significant market drivers. However, challenges such as high treatment costs, misdiagnosis, and limited awareness may restrain the market growth. Additionally, the need for personalized medicine and ongoing research into new treatment options will further shape the market landscape. Overall, the combination of rising demand and innovative therapies positions the nasal polyps treatment market for substantial growth in the coming years.
Discover high revenue pocket segments and roadmap to it: Download Free Sample
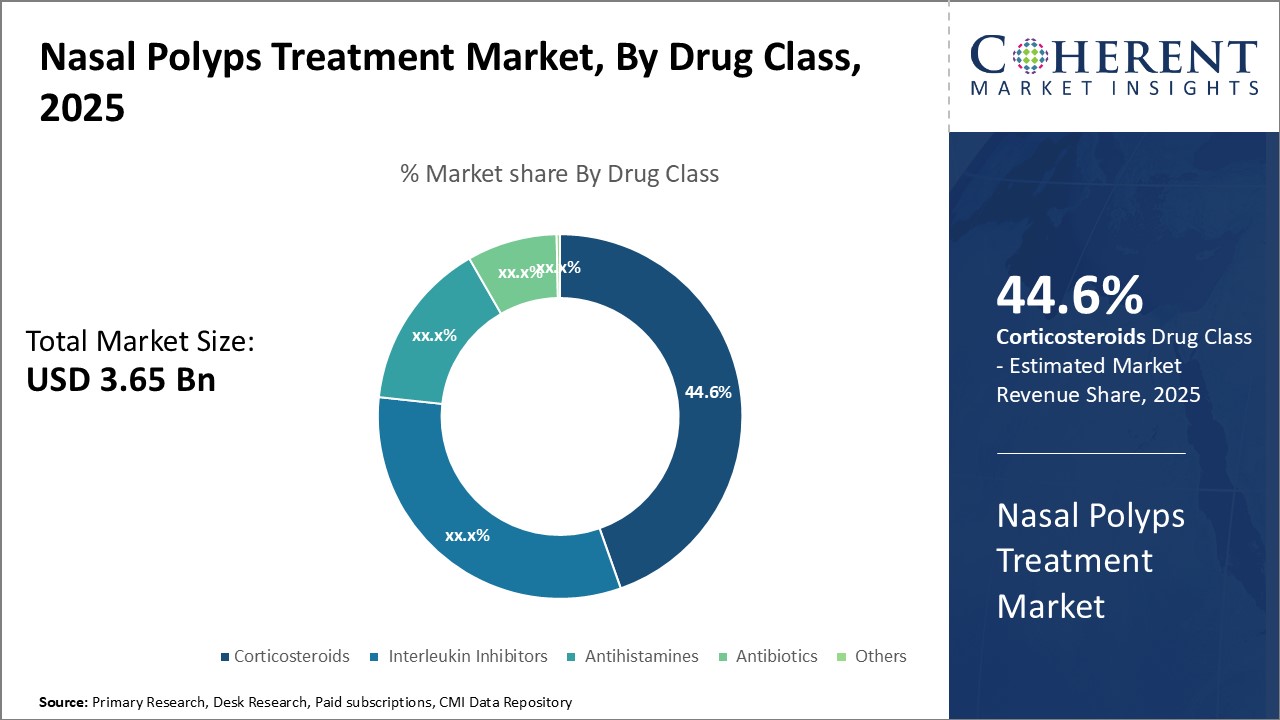
Insights By Drug Class- Robust management of nasal polyps establishes corticosteroids’ popularity
In terms of drug class, corticosteroids is expected to contribute the highest share of 44.6% in 2025, owing to its high acceptance and efficacy in managing nasal polyp symptoms. As nasal polyps are non-cancerous growths caused by inflammation, corticosteroids work by reducing inflammation and swelling of the nasal passages. The administration of corticosteroids directly to the nasal cavity also allows for higher drug concentrations at the site of inflammation with minimal systemic side effects.
Insights By Route of Administration- Nasal segment dominates due to ease of administration
In terms of route of administration, the nasal segment is expected to contribute the highest share of 62.1% in 2025. This is because nasal polyps originate and develop within the nasal cavity allowing direct delivery of drugs to the site of action via the intranasal route. Compared to oral and parenteral administration, nasal delivery provides better efficacy by achieving higher drug concentrations within the nasal tissue and paranasal sinuses with minimal systemic absorption and side effects.
Insights By Distribution Channel- Hospital pharmacies dominate owing to convenience and accessibility
In terms of distribution channel, the hospital pharmacies segment is expected to contribute the highest share of 48.7% in 2025. This is because patients undergoing nasal polyps treatment may require follow-up consultations and check-ups. Thus, having the medications dispensed at the hospital makes it very easy for them. Hospital pharmacies allow patients to obtain their drugs right at the location where their treatment is taking place, saving them time and effort.
Need a Different Region or Segment? Download Free Sample
Regional Analysis: Global Nasal Polyps Treatment Market
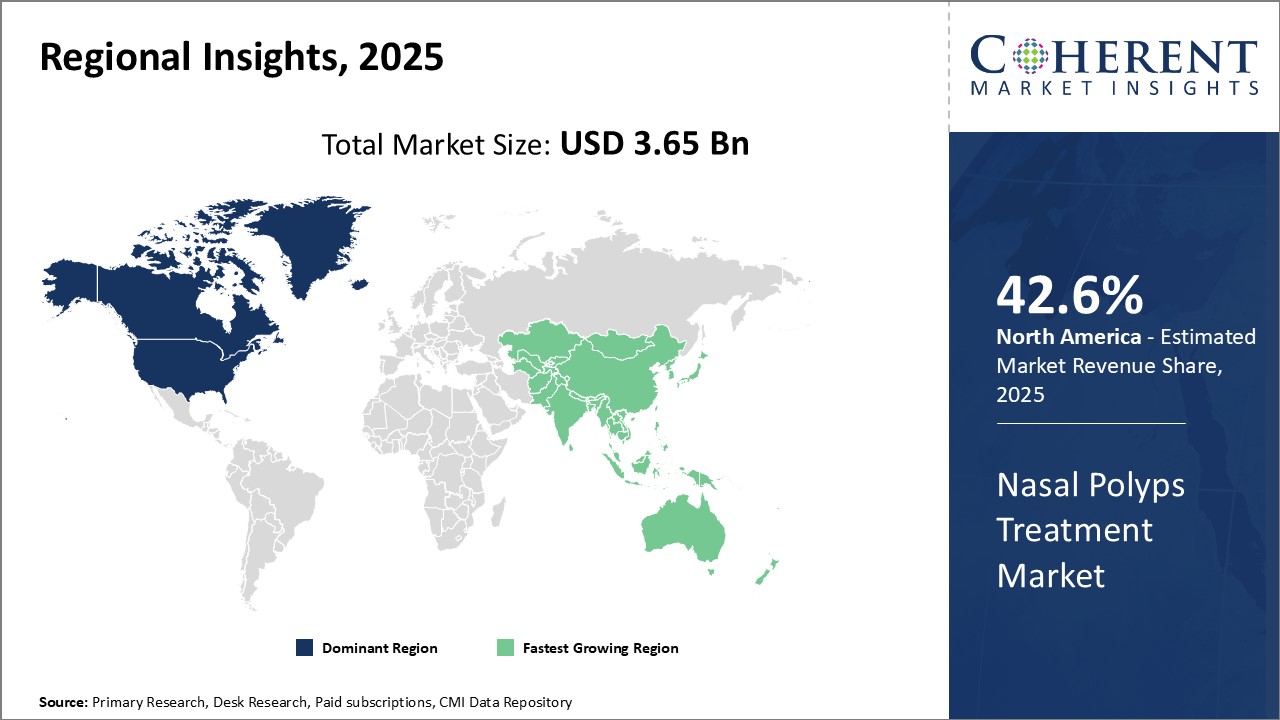
Dominating Region: North America
North America is expected to dominate the global nasal polyps treatment industry, with an estimated market share of 42.6% in 2025, due to factors such as the strong presence of leading pharmaceutical and medical device companies, rising healthcare expenditure, and favorable reimbursement scenario.
Fastest-Growing Region: Asia Pacific
Asia Pacific region exhibits the fastest growth in the nasal polyps treatment industry, with an estimated market share of 23.9% in 2025, due to the large patient pool, increasing awareness about nasal polyps, and growing focus of international players on expanding their footprint in emerging countries.
Nasal Polyps Treatment Market Outlook for Key Countries
High adoption of advanced therapies in the U.S.
The U.S. nasal polyps treatment industry is characterized by a high adoption of advanced therapies and procedure-based treatments. Leading U.S.-based companies are introducing innovative drugs and devices that significantly enhance clinical outcomes, driving the market growth. This focus on cutting-edge solutions positions the U.S. as a leader in effective nasal polyps management.
Strategic collaborations between domestic and foreign industries in China
The nasal polyps treatment industry in China is experiencing robust growth, fueled by strategic collaborations between domestic and foreign industry stakeholders. The government is actively implementing initiatives to enhance local manufacturing capabilities and improve access to care, ensuring that innovative treatments are more readily available to patients across the country.
Low-cost drug manufacturing in India
India continues to excel in the nasal polyps treatment industry, driven by its leadership in low-cost drug manufacturing. Several Indian pharmaceutical companies provide affordable generic medications globally, leveraging extensive distribution networks to dominate the local market. This accessibility enhances treatment options for patients suffering from nasal polyps, supporting broader healthcare initiatives.
Research and Development in Japan
The nasal polyps treatment sector in Japan is advanced due to substantial R&D investments from prominent medical technology companies. These investments have led to rapid approvals of innovative therapies, such as GSK's Nucala (mepolizumab), which is gaining preference among healthcare providers. This focus on cutting-edge treatments enhances patient care and positions Japan as a leader in the market.
Get actionable strategies to beat competition: Download Free Sample
Top Strategies Followed by Global Nasal Polyps Treatment Market Players
Emerging Startups in the Global Nasal Polyps Treatment Market
Key Takeaways from Analyst
Nasal Polyps Treatment Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2024 | Market Size in 2025: | USD 3.65 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2025 To 2032 |
| Forecast Period 2025 to 2032 CAGR: | 7.8% | 2032 Value Projection: | USD 6.19 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
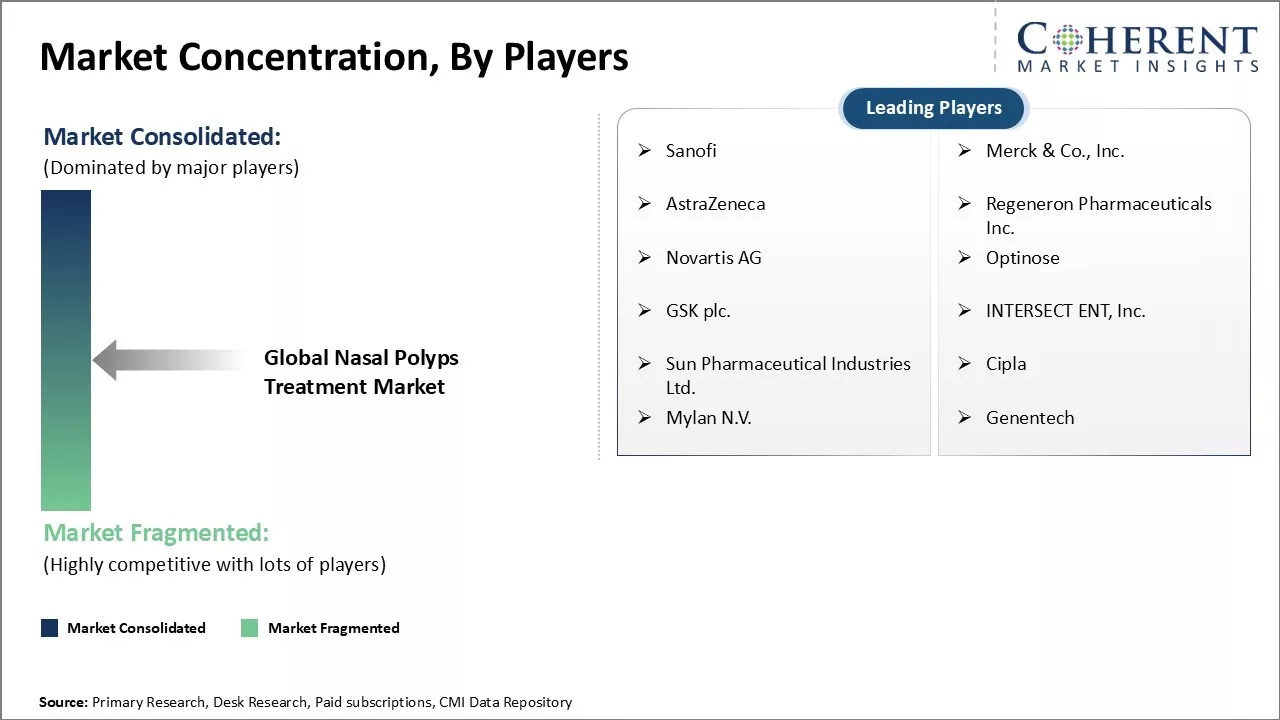
Sanofi, Merck & Co., Inc., AstraZeneca, Regeneron Pharmaceuticals Inc., Novartis AG, Optinose, GSK plc., INTERSECT ENT, Inc., Sun Pharmaceutical Industries Ltd., Cipla, Mylan N.V., and Genentech |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
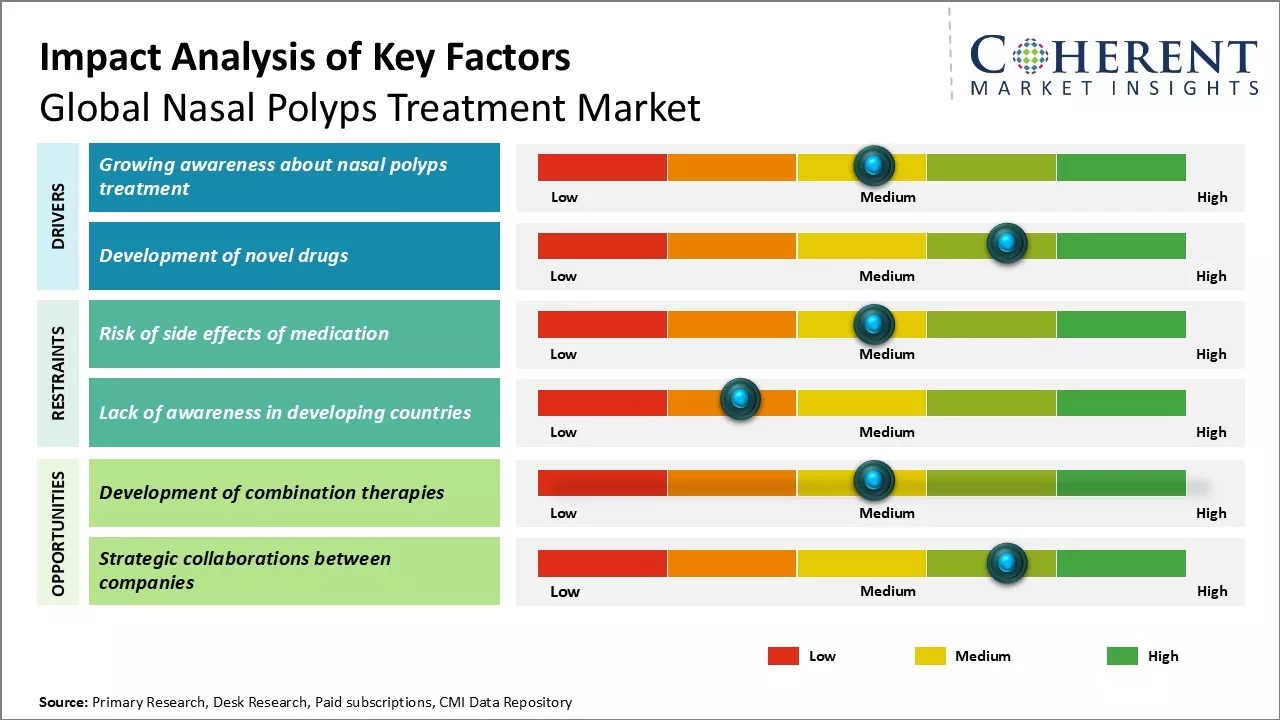
Market Driver - Growing awareness about nasal polyps
There has been a significant rise in awareness regarding nasal polyps and their treatment options globally. This increase is attributed to better access to health-related information online, improved health literacy, government initiatives, and promotional efforts by pharmaceutical companies. As a result, people are now more likely to recognize symptoms such as chronic nasal obstruction and seek medical advice for effective treatment.
Market Challenge - Risk of side effects of medication
The use of medications for nasal polyps, particularly corticosteroids, often leads to side effects such as headaches, nausea, and nasal irritation with long-term use. Complications like insomnia, anxiety, and high blood pressure can also arise, making patients hesitant to adhere to treatment schedules. These risks contribute to non-adherence, negatively impacting treatment effectiveness and increasing the likelihood of polyp recurrence. Consequently, the occurrence of side effects presents significant challenges for physicians and hinders market growth in the nasal polyps treatment sector.
Market Opportunity - Development of combination therapies for market
The growing demand for more effective and safer treatment options presents a significant opportunity in the nasal polyps treatment market. There is considerable potential for developing novel combination therapies that integrate corticosteroids with other drug classes such as monoclonal antibodies and antihistamines. Collaborative partnerships between drug manufacturers to create well-researched combination formulations represent an untapped area that could capture a substantial market share, positioning companies at the forefront of addressing the unmet needs in advanced polyp management therapies.
Share
Share
About Author
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Missing comfort of reading report in your local language? Find your preferred language :
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients