Intracranial Stents Market – Insights
Narrowing of the blood vessels or blood clots due to building up of fats, plaque, and other substances in the blood vessels is called intracranial stenosis. This condition marks with a stroke either to the carotid artery in the neck or in an intracranial artery. Stents during this condition act as a permanent implant which allows blood flow to the brain by opening the blocked arteries. Angioplasty (use of balloon catheter) along with placement of a stent may be an appropriate treatment for the patients who have had a stroke or transient ischemic attack (TIA). However, since these stents are designed for use in the heart, it is complex to use them in the arteries of the brain. Though, with the advent of the new self-expanding brain stents, the rate of reoccurrence of artery narrowing can be lowered. For instance, Acclino microstent offered by Acandis GmbH, shows a satisfactory safety profile and a promising rate of immediate and mid-term complete aneurysm occlusion for stent-assisted coil embolization in wide-neck intracranial aneurysms.
The global intracranial stents market size was valued at US$ 313.5 million in 2017, and is expected to witness a CAGR of 15.6% over the forecast period (2018 – 2026).
Figure 1. Global Intracranial Stents Market Share Analysis (%), By Region, 2017
To learn more about this report, Download Free Sample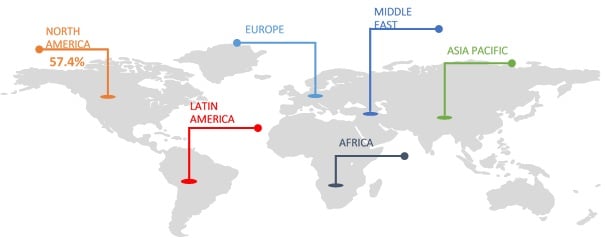
Source: Coherent Market Insights Analysis (2018)
Intracranial Stents Market Dynamics
Increasing incidence of stroke and brain aneurysm is propelling demand for intracranial stents. According to the Brain Aneurysm Foundation (BAF), brain aneurysm accounts for around 500,000 deaths worldwide annually. Moreover, BAF estimates that aneurysm is predominant in the age group of 35–60 years. Furthermore, high prevalence of stroke among geriatric population worldwide is also expected to propel demand for intracranial stents in the near future. According to the data published in the Medscape, 2010, around 75-89% of strokes occur in individuals aged > 65 years. Of these strokes, 50% occur in people who are aged ≥ 70 years and nearly 25% occur in individuals who are aged > 85 years.
Integration of advanced software with existing products are gaining significant traction among various market players of intracranial stents with affordable costs thus, expected to drive intracranial stents market growth. For instance, in September 2016, Medtronic plc received the U.S. Food and Drug Administration (FDA) approval for its StealthStation, which is integrated with Medtronic’s latest O-arm Imaging System, enabling clinicians to place the deep brain stimulation (DBS) leads with high level of accuracy during the cranial procedures.
However, high cost of the procedure and limited use of various intracranial stents to a small population may impact the overall market to some extent. For instance, in August 2012, the U.S. FDA approved label changes for the Wingspan Stent System offered by Stryker Corporation, reflecting changes in indications for use, and limiting the system to a much smaller group of patients between 22 and 80 years with intracranial stenosis.
Intracranial Stents Market Regional Outlook
On the basis of geography, the global intracranial stents market is segmented into North America, Europe, Asia Pacific, Latin America, Middle East, and Africa. North America is expected to hold dominant position in this market, owing to increasing geriatric population who are more prone to brain diseases and adoption of technologically advanced products such as adoption of cranio maxillofacial implants. Emerging economies are also projected to witness significant growth in market over the forecast period, owing to high prevalence of intracranial strokes. For instance, according to the American Journal of Neuroradiology, 2011, intracranial cerebral atherosclerosis disease (ICAD) accounts for around 8%–10% of all ischemic strokes with a higher reported incidence in the Asian, African, and Hispanic descent populations. The factors responsible for disease prevalence in these regions includes cigarette smoking, hypertension, insulin-dependent diabetes mellitus, and hypercholesterolemia. Thus, emerging economies such as Asia Pacific and Latin America is likely to be the fastest growing region throughout the forecast period.
Major companies operating in the global intracranial stents market include Medtronic Plc, Balt Extrusion S.A., MicroPort Scientific Corporation, Abbott Laboratories, Stryker Corporation, Admedes Schuessler GmbH, Cardiatis, S.A, DePuy Synthes, Acandis GmbH, and Penumbra, Inc.
Share
Share
Missing comfort of reading report in your local language? Find your preferred language :
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
Select a License Type
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients