Gene Therapy for Rare Disease Market - Insights
Gene therapy involves insertion of genetic material into cells of the patients to provide new function or to restore missing or inappropriate functioning of the cells due to cancer or gene mutations. According to National Center for Advancing Translational Sciences, although around 7,000 rare diseases are identified, only a few hundred have approved treatments. Moreover, developing gene therapy for rare diseases is gaining significant traction, as more than 80% of rare diseases have a known monogenic (single-gene) cause. Small molecule drugs often treat symptoms only rather than curing underlying cause of the disease. This requires frequent administration of the drugs to manage the condition. Gene therapy holds the potential to correct underlying genetic defects. Moreover, a clinically sound and successful gene therapy may only require a single dose to cure a disease rather than requiring a lifetime treatment. Commercially approved gene therapies include Kymriah (Novartis AG), Yescarta (Gilead Sciences, Inc.), Luxturna (Spark Therapeutics), and Strimvelis (Orchard Therapeutics).
Increasing drug approvals and launches of novel gene therapies for treatment of various rare diseases are expected to boost the gene therapy for rare disease market growth over the forecast period
In recent past, key players in the market have gained regulatory approvals and have launched their therapies in the market. Frequent approvals of novel gene therapies and their launches in the developed regions are expected to significantly drive gene therapy for rare disease market growth over the forecast period. For instance, in December 2017, Spark Therapeutics, Inc. received the U.S. Food and Drug Administration (FDA) approval for its LUXTURNA (voretigene neparvovec-rzyl), a one-time gene therapy product indicated for the treatment of patients with confirmed biallelic RPE65 mutation-associated retinal dystrophy. Furthermore, in August 2018, Novartis International AG received European Commission’s (EC) approval for Kymriah (tisagenlecleucel, formerly CTL019) indicated for treatment of patients up to 25 years of age with relapse or refractory B-cell acute lymphoblastic leukemia (ALL) and for the treatment of adult patients with relapsed or refractory (r/r) diffuse large B-cell lymphoma (DLBCL) after two or more lines of systemic therapy. Moreover, in August 2018, Kite Pharma, Inc., a Gilead Company, received European Commission (EC) Marketing approval for its novel Yescarta (axicabtagene ciloleucel), as a treatment for adult patients with relapsed or refractory diffuse large B-cell lymphoma (DLBCL) and primary mediastinal large B-cell lymphoma (PMBCL), after two or more lines of systemic therapy.
The global gene therapy for rare disease market size was valued at US$ 16.3 million in 2017, and is expected to witness a robust CAGR of 35.0% over the forecast period (2018–2026).
Figure No.1: Global Gene Therapy for Rare Disease Market Share, By Drug (%), 2018 -2026 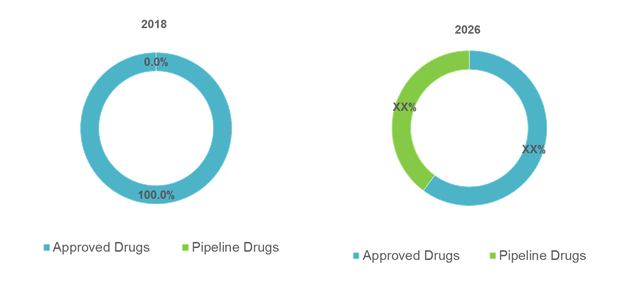
To learn more about this report, Download Free Sample
Source: Coherent Market Insights Analysis (2018)
Ongoing research and development in gene therapies for rare diseases is expected to boost growth of the global gene therapy for rare disease market
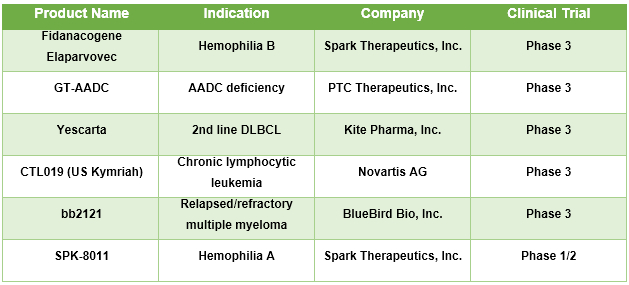
Presence of multiple novel gene therapies for various rare disease treatment in pipeline, which are expected to gain approval is expected to boost the market growth over the forecast period. For instance, Spark Therapeutics, Inc. is developing gene therapy namely Fidanacogene Elaparvovec, a novel, investigational gene therapy indicated for the treatment of patients suffering from hemophilia B. Currently, the therapy is under Phase 3 clinical trial and is expected to receive approval in near future. In August 2018, uniQure N.V. treated the first patient in its Phase IIb dose-confirmation study of AMT-061, an investigational AAV5-based gene therapy incorporating the FIX-Padua variant for the treatment of patients with severe and moderately severe hemophilia B. In May 2018, Orchard Therapeutics Plc. received Rare Pediatric Disease Designation from the U.S. Food and Drug Administration (FDA) for its gene therapy candidate OTL-200, indicated for treatment of patients with metachromatic leukodystrophy (MLD). PTC Therapeutics, In. has GT-AADC in phase 3 clinical trials and is indicated for Aromatic L-Amino Acid Decarboxylase (AADC) deficiency.
Table No.1: Gene Therapy for Rare Disease Pipeline
To learn more about this report, Download Free Sample
Source: Company Websites, Clinicaltrials.gov
However, lack of awareness among population regarding rare diseases and their treatment is expected to negatively affect growth of the gene therapy for rare disease market. For instance, in 2017, uniQure N.V. dropped their plant to pursue the renewal of its gene therapy Glybera, which was approved in 2012, for treatment for a small subset of patients with familial lipoprotein lipase deficiency (LPLD). The company dropped their plan due to very low demand of therapy in the market
Some of the major players operating in the global gene therapy for rare disease market include Kite Pharma, Inc. (Gilead Sciences, Inc.), Novartis International AG, Juno Therapeutics Inc. (Celgene Corporation), Bluebird Bio, Inc., Spark Therapeutics, Inc., uniQure N.V, Orchard Therapeutics Plc., PTC Therapeutics, Inc., and BioMarin Pharmaceutical Inc.
Share
Share
Missing comfort of reading report in your local language? Find your preferred language :
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
Select a License Type
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients