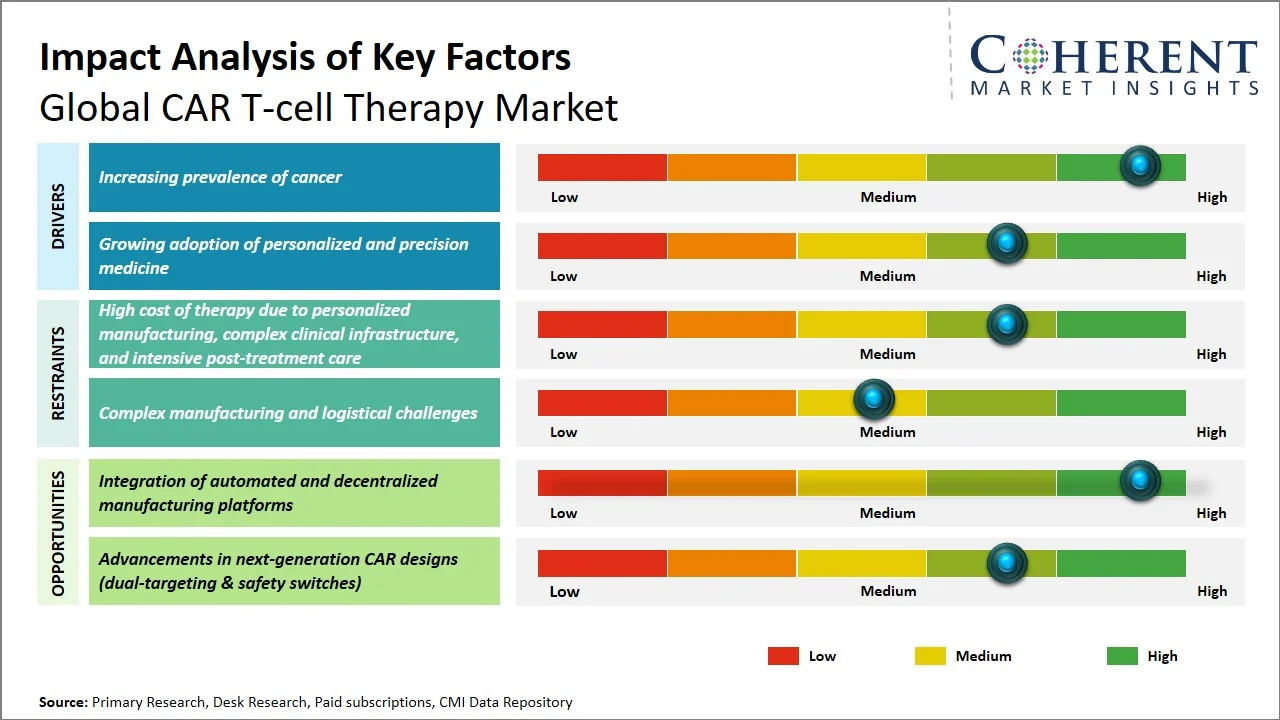
The global CAR T-cell therapy market is expected to grow from USD 4.83 Bn in 2026 to USD 18.46 Bn by 2033, registering a compound annual growth rate (CAGR) of 21.1%. The global CAR T-cell therapy market is poised for significant expansion, fueled by rising global burden of cancer, particularly hematological malignancies.
According to the Global Cancer Observatory (GLOBOCAN), there were approximately 19.3 million new cancer cases and nearly 10 million cancer-related deaths worldwide in 2022, highlighting a substantial and growing patient pool. In addition, leukemia accounted for over 470,000 new cases globally, reinforcing the increasing prevalence of blood cancers where CAR-T therapy has demonstrated strong clinical efficacy. Furthermore, GLOBOCAN estimates indicate over 53.5 million prevalent cancer cases (5-year prevalence) worldwide, reflecting a significant population living with cancer and requiring ongoing and advanced treatment solutions
(Source: Global Cancer Observatory)
CD19 is projected to hold a market share of 75.0% in 2026, owing to its high and stable expression in B cell malignancies including acute lymphoblastic leukemia (ALL) and diffuse large B-cell lymphoma (DLBCL) and thus its suitability for use as an ideal and validated target. For instance, research published by National Library of Medicine in May 2025, indicated that CD19 CAR T-cell therapies yield high response rates of about 83% in B cell non-Hodgkin’s lymphoma and 90% in B cell acute lymphoblastic leukemia, which shows the high level of efficacy for the treatments. (Source: National Library of Medicine)
CAR-T therapies targeting CD19 are among first to receive U.S. Food and Drug Administration regulatory approvals, making it one of the pioneering markets in the industry. Drugs like Kymriah and Yescarta have been proven to have a higher response rate, which increases the assurance of physicians and leads to quicker adoption. Furthermore, due to active clinical testing and label expansion, the domination of this segment in the industry becomes even more pronounced.
To learn more about this report, Request Free Sample
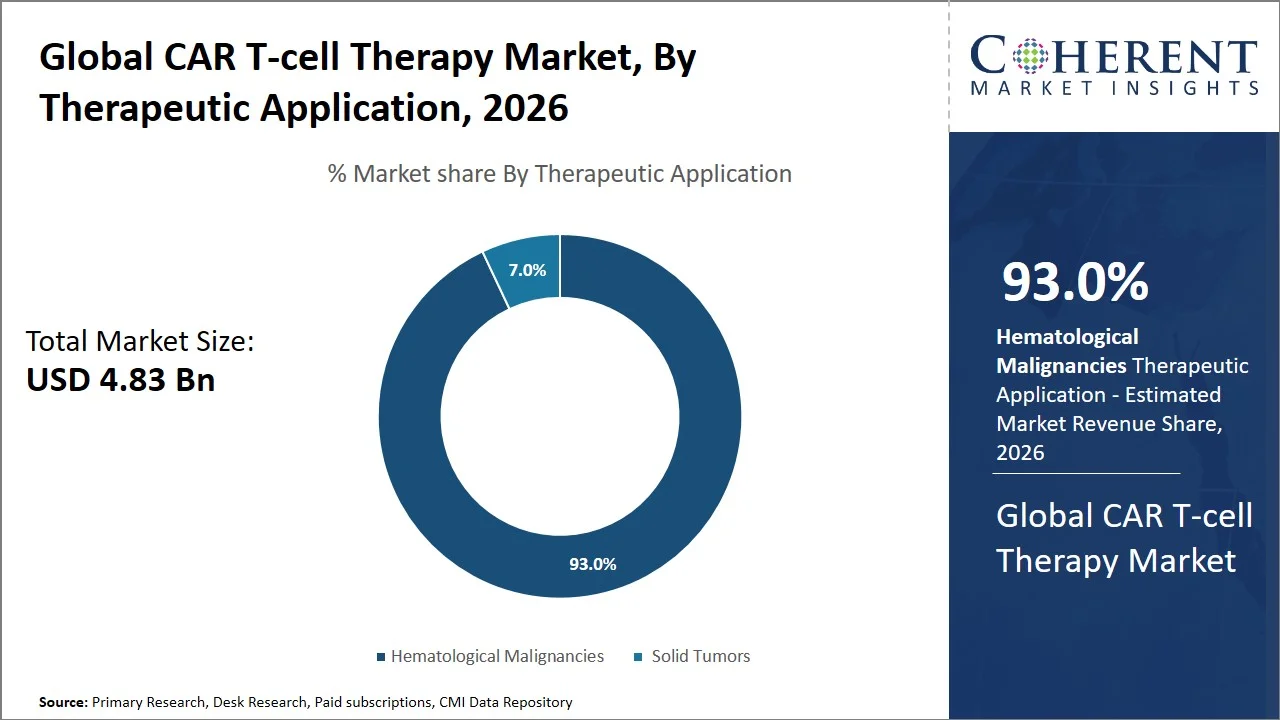
Hematological Malignancies are projected to hold a market share of 93.0% in 2026 based on its relative accessibility and well-defined antigens that can facilitate the targeted action of genetically engineered T-cells. While solid tumors have a number of physical characteristics making it difficult for the genetically engineered T-cells to access them and survive in such an environment, hematology does not possess any such challenges, thus ensuring a high success rate for the CAR T-cell therapy. Besides, the vast number of clinical trials and successful approvals associated with the use of CAR T-cell therapy in the treatment of hematology ensures that this application continues to dominate the market.
For instance, according to an August 2024 meta-analysis study published by Frontiers Media SA, the overall response rates (ORR) rate was 70–90%, while complete response (CR) was 40–60% for relapsed or refractory patients with hematologic cancer using CAR-T therapy, where a considerable number of these patients achieved a sustained response rate exceeding 12 months. (Source: Frontiers Media SA)
The autologous CAR T-cell therapy segment is projected to hold a market share of 90.0% in 2026, due to its proven clinical safety and established regulatory approvals. Since this therapy involves using T-cells from the patients themselves, it greatly minimizes the risks of immune rejection such as graft-versus-host disease (GVHD), and other complications, thus contributing to better treatment results.
Moreover, autologous treatments are the first ones that have gained commercial success through effective production practices and robust clinical trial results for hematological cancers. For example, the approval and subsequent acceptance of Kymriah (2017) and Yescarta (2017), two of the first autologous chimeric antigen receptor-T cell (CAR-T) therapies have received U.S. FDA approval. These treatments showed high remission success in the treatment of relapsed or refractory B-cell cancers such as acute lymphocytic leukemia (ALL) and diffuse large B-cell lymphoma (DLBCL), resulting in their immediate use in practice and making autologous CAR-T the prevailing commercial form.
In spite of difficulties with regard to manufacturing times and costs, the effectiveness and quality of autologous treatments continue playing a crucial role in their demand. For instance, in December 2024, clinical study published by Haymarket Media, Inc. reported an overall response rate (ORR) of 72.8% and a complete response (CR) rate of 50.8% in patients with relapsed or refractory diffuse large B-cell lymphoma treated with autologous CAR-T therapy, highlighting its strong and durable clinical effectiveness. (Source: Haymarket Media, Inc.)
|
Current Events |
Description and its Impact |
|
Approval of First Allogeneic CAR T-Cell Therapy Clinical Advancements (Ongoing Regulatory Progress) (2023-Present) |
|
|
U.S. FDA Accelerated Approval Pathways for CAR T-Cell Therapies (Regulatory Shift Toward Faster Approvals) (2017-Present) |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
(Source: U.S. Food and Drug Administration, U.S. Food and Drug Administration)
To learn more about this report, Request Free Sample
To learn more about this report, Request Free Sample
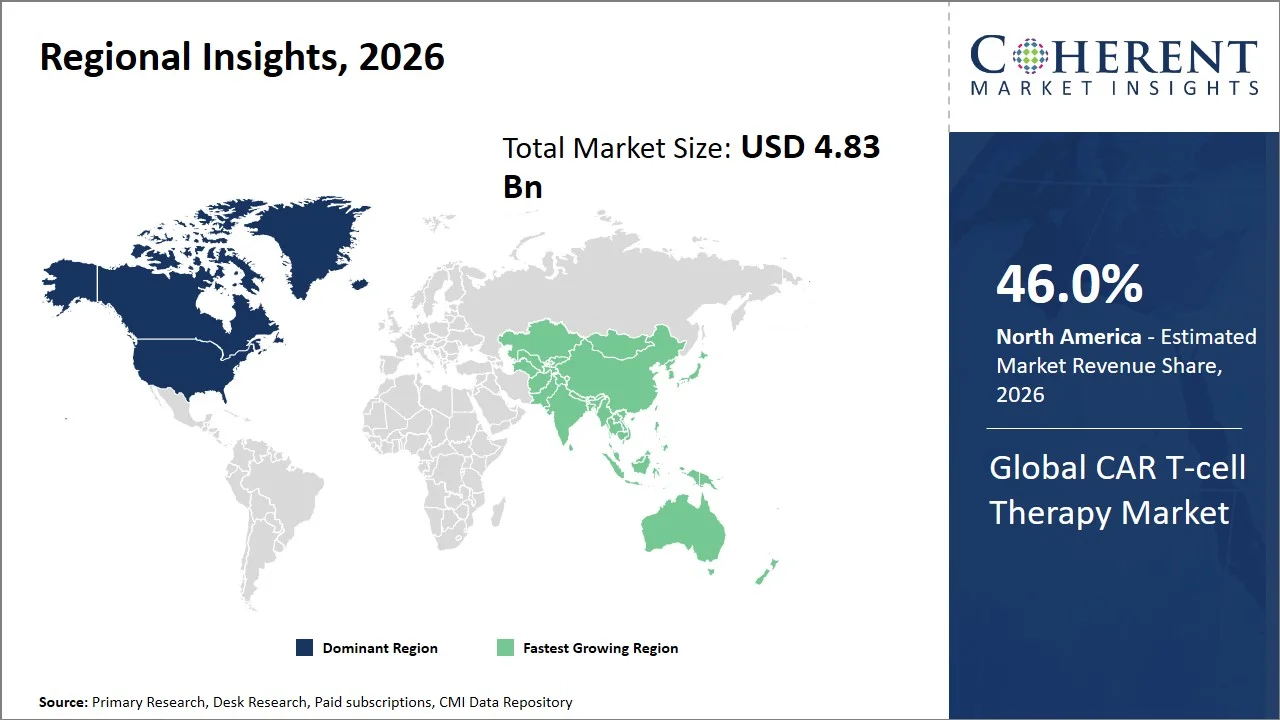
North America leads the global CAR T-cell Therapy market, accounting for an estimated 46.0% share in 2026, attributed to the highly developed healthcare systems, established regulatory framework, and well-developed reimbursement schemes that allow rapid integration of novel therapies into practice (such as, Medicare coverage through the Inpatient Prospective Payment System (IPPS) and New Technology Add-on Payment (NTAP), as well as private payer reimbursement from UnitedHealthcare, Aetna, and Blue Cross Blue Shield). There is also an elevated frequency of hematological cancer cases within the region, coupled with an advanced infrastructure of healthcare facilities specializing in cell-based treatment. For instance, according to the National Cancer Institute (SEER Program), leukemia incidence stands at approximately 14.4 cases per 100,000 population annually, while non-Hodgkin lymphoma incidence is around 19.6 cases per 100,000 population. (Source: National Cancer Institute)
Additionally, multiple myeloma incidence is reported at approximately 7.3 cases per 100,000 population, further contributing to the regional disease burden. (Source: National Cancer Institute) Moreover, with significant investments being made into the biotechnology industry (with the National Institutes of Health (NIH) investing nearly USD 48 billion annually in biomedical research, including approximately USD 35.3 billion in research grants), alongside the presence of major pharma/biotech firms (such as, Pfizer Inc., Johnson & Johnson, Merck & Co., Bristol-Myers Squibb, Gilead Sciences, Amgen Inc., AbbVie Inc., Biogen Inc.), there is constant innovation and expansion of the product pipeline.
The Asia Pacific CAR T-cell therapy market is expected to exhibit the fastest growth with an estimated contribution of 19.0% share to the market in 2026, owing to the rapid development of clinical trials, increased governmental support, and rise in cancer cases in the region. Asia Pacific has emerged itself as a global hub for oncology and CAR-T research, with countries such as China, Japan, and South Korea leading in innovation and trial participation. For instance, China has already carried out 1,006 CAR-T clinical trials, which demonstrates the potential of the region in terms of innovation. (Source: National Library of Medicine)
Moreover, the region contributes to a considerable percentage of precision oncology studies on a global scale and attracts investment due to cheaper trials and faster recruiting of patients. Furthermore, the presence of many governmental programs and favourable regulatory changes (including Japan’s Act on the Safety of Regenerative Medicine (ASRM) and Pharmaceuticals and Medical Devices Act (PMD Act), China’s National Medical Products Administration (NMPA) accelerated approval pathways and Drug Regulatory Science Action Plan, South Korea’s Advanced Regenerative Medicine and Advanced Biological Products (ARM/ABP) framework, and Singapore’s Cell, Tissue and Gene Therapy Products (CTGTP) regulations) in the sphere facilitate further developments in the market.
The U.S. is the leader in innovations and implementations within the CAR T-cell therapy market due to its robust regulatory backing and early approvals from the U.S. FDA, enabling faster commercialization of advanced therapies The country also boasts government support in terms of funding as the National Institutes of Health (NIH) spends billions of dollars each year on biotechnology and cancer studies. For instance, the National Institutes of Health invests most of its nearly USD 48 billion for medical research in fiscal year 2024, wherein a considerable chunk was dedicated to oncology and cell and gene therapy research, contributing to continued innovation in CAR-T therapy. (Source: National Institutes of Health)
Moreover, the presence of top-notch pharmaceutical and biotech firms (such as Pfizer Inc., Gilead Sciences, Inc.) coupled with a solid system of clinical trials (549 trials) helps to facilitate innovations. The country has a solid infrastructure that allows for quick adoptions, with a large number of specialized medical centers (such as MD Anderson Cancer Center, Memorial Sloan Kettering Cancer Center, Mayo Clinic, Dana-Farber Cancer Institute, City of Hope National Medical Center) and a favorable reimbursement policy (such as Medicare coverage under the Inpatient Prospective Payment System (IPPS), New Technology Add-on Payment (NTAP), and private payer coverage from UnitedHealthcare, Aetna, and Blue Cross Blue Shield) being the driving force behind this achievement.
U.K. represents an ideal environment for the CAR T-cell therapy market owing to its regulatory and reimbursement framework provided by the National Health Service along with recommendations by the National Institute for Health and Care Excellence. Access to this therapy among patients can be easily achieved by utilizing managed access agreements and centralized funds for facilitating better access to expensive therapies.
Furthermore, there exists a strong research community along with specialized treatment centers within the U.K. (such as University College London Hospitals NHS Foundation Trust, The Christie NHS Foundation Trust, King’s College Hospital NHS Foundation Trust, Oxford University Hospitals NHS Foundation Trust, Manchester Royal Infirmary) which support the implementation of advanced treatments.
China stands out as one of the most important regions with respect to the future growth of the CAR T-cell therapy market owing to the fast growth rates that are observed in the clinical trials carried out within the country (China has already carried out 1,006 CAR-T clinical trials). The Chinese National Medical Products Administration has adopted strategies (such as Priority Review and Approval (PRA) pathway, Breakthrough Therapy Designation (BTD), Conditional Approval pathway, and the Drug Regulatory Science Action Plan) that will ensure faster approval processes for innovations.
Further, the existence of local biotechnology firms (such as CARsgen Therapeutics, Legend Biotech, JW Therapeutics, Gracell Biotechnologies, IASO Biotherapeutics) and improved manufacturing capabilities makes its even stronger.
Germany emerges as a leader in the Europe CAR T-cell therapy market, as it offers state-of-the-art health care infrastructure along with favorable reimbursement policies (such as German Diagnosis-Related Group (G-DRG) system, NUB (Neue Untersuchungs- und Behandlungsmethoden) reimbursement pathway, AMNOG (Arzneimittelmarkt-Neuordnungsgesetz) pricing and reimbursement framework) and early adoption of innovative therapies. Germany enjoys the advantage of having efficient hospitals and specialized centers (such as University Hospital Heidelberg, Charité – Universitätsmedizin Berlin, University Hospital Munich (LMU Klinikum), University Hospital Frankfurt, University Hospital Cologne) that can provide the latest therapies in terms of cell-based treatments. Moreover, the Diagnosis-Related Group (DRG) based reimbursement system makes it easy to fund expensive drugs such as CAR-T treatments, thus making them easily available to patients.
The market for CAR-T cell therapy in Japan is witnessing growth due to its robust regulatory framework and increasing acceptance of such innovative therapies. Several CAR-T cell therapies have been approved in Japan for the treatment of hematological malignancies (such as Kymriah, Yescarta, Tecartus, Breyanzi, Abecma), suggesting the trend towards commercialization of innovative products. Furthermore, the regulatory framework of Japan, particularly those pertaining to the Pharmaceuticals and Medical Devices Act (PMD Act) and Act on the Safety of Regenerative Medicine (ASRM), enable conditional and accelerated approvals of cell and gene-based products, thereby aiding product innovation.
|
Region |
Key Regulatory Bodies |
Guidelines |
Key Highlights |
|
North America |
U.S. Food and Drug Administration (FDA), Health Canada |
U.S. FDA Center for Biologics Evaluation and Research (CBER), Regenerative Medicine Advanced Therapy (RMAT) designation |
Strong regulatory support with expedited pathways (RMAT, Breakthrough Therapy); well-defined approval process for cell and gene therapies; robust post-market surveillance |
|
Europe |
European Medicines Agency (EMA), National Competent Authorities |
Advanced Therapy Medicinal Products (ATMP) Regulation (EC No. 1394/2007) |
Centralized approval process; stringent quality, safety, and efficacy requirements; strong focus on pharmacovigilance and long-term follow-up |
|
Asia Pacific |
PMDA (Japan), NMPA (China), CDSCO (India) |
Japan’s Act on the Safety of Regenerative Medicine; China’s cell therapy guidelines; India’s National Guidelines for Stem Cell Research |
Rapidly evolving regulatory landscape; accelerated approvals in Japan; increasing clinical trial activity in China; supportive government policies |
|
Latin America |
ANVISA (Brazil), COFEPRIS (Mexico) |
National biologics and advanced therapy regulations |
Emerging regulatory framework; improving approval timelines; increasing adoption of advanced therapies with evolving guidelines |
|
Middle East & Africa |
Saudi FDA (SFDA), South African Health Products Regulatory Authority (SAHPRA) |
National regulatory frameworks for biologics and gene therapies |
Nascent but developing regulatory ecosystem; growing focus on advanced therapeutics; increasing investments in healthcare infrastructure |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
The adoption of automation and decentralization of manufacturing systems is creating ample growth opportunities in the CAR-T cell therapy market by overcoming one of its major limitations which is the process of manufacturing that requires significant time and investment. For example, systems such as the automated closed-systems, like the CliniMACS Prodigy, make it possible for CAR-T cells to be processed from start to finish in one machine. Decentralized manufacturing approaches, on the other hand, permit the manufacture of the CAR-T products closer or even in hospital facilities, hence greatly reducing the vein-to-vein duration of the process. (Source: Miltenyi Biotec) In addition, these developments allow corporations to extend their geographical reach, improve logistics management, and better utilize their resources, leading to faster deployment of CAR-T treatments worldwide.
To learn more about this report, Request Free Sample
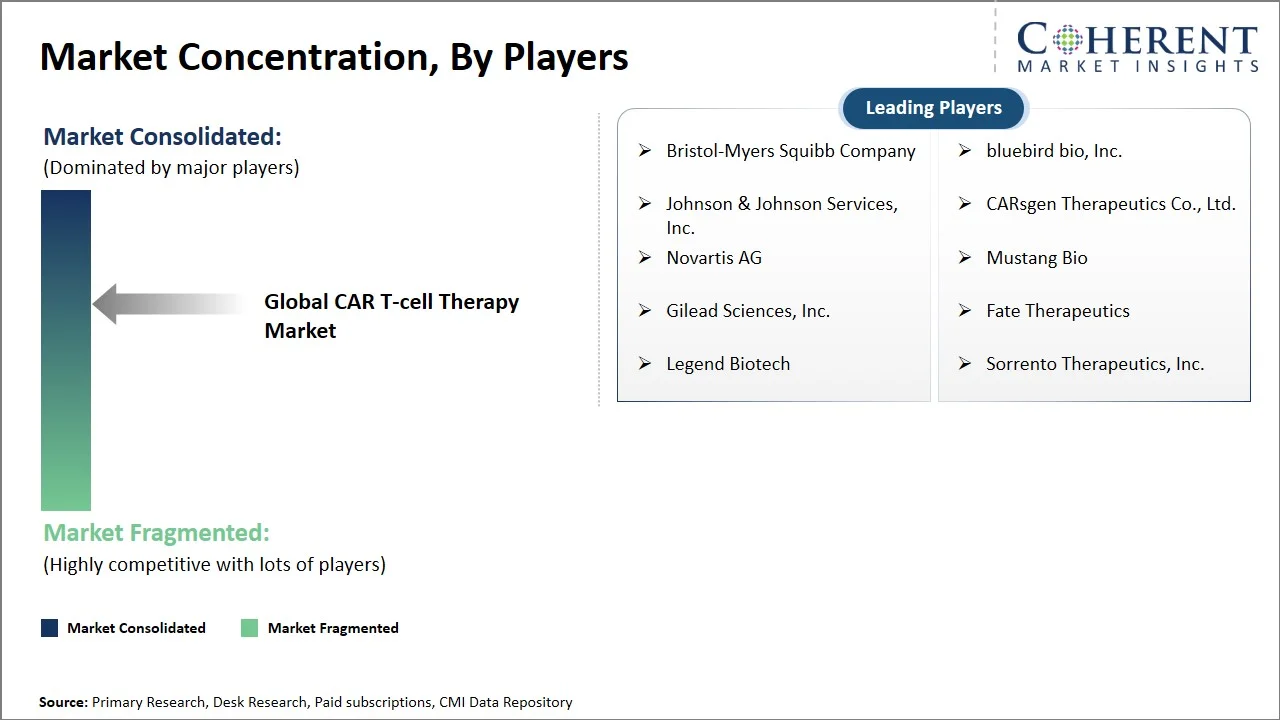
The global CAR T-cell therapy market is highly competitive yet moderately consolidated, with few well-known companies dominating the market for commercialized therapies, while a number of new biotech companies are making the competition in the pipeline even stronger. The market is driven by ongoing improvements in cell engineering, strong clinical pipelines, and more regulatory approvals. Companies are working hard to expand their indications beyond blood cancers to solid tumors. Companies that have advanced manufacturing capabilities and have invested in allogeneic (off-the-shelf) platforms are in a better position to get ahead of the competition. Key focus areas include:
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 4.83 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 21.1% | 2033 Value Projection: | USD 18.46 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Bristol-Myers Squibb Company, Johnson & Johnson Services, Inc., Novartis AG, Gilead Sciences, Inc., Legend Biotech, bluebird bio, Inc., CARsgen Therapeutics Co., Ltd., Mustang Bio, Fate Therapeutics, and Sorrento Therapeutics, Inc. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Nikhilesh Ravindra Patel is a Senior Consultant with over 8 years of consulting experience. He excels in market estimations, market insights, and identifying trends and opportunities. His deep understanding of the market dynamics and ability to pinpoint growth areas make him an invaluable asset in guiding clients toward informed business decisions. He plays a instrumental role in providing market intelligence, business intelligence, and competitive intelligence services through the reports.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients