Acromegaly Treatment Market Size and Forecast – 2025 – 2032
The Acromegaly Treatment Market size is estimated to be valued at USD 1.45 billion in 2025 and is expected to reach USD 2.34 billion by 2032, exhibiting a compound annual growth rate (CAGR) of 6.8% from 2025 to 2032.
Global Acromegaly Treatment Market Overview
Acromegaly treatment products are pharmaceutical formulations developed to manage the overproduction of growth hormone caused by pituitary gland tumors. These include somatostatin analogs, growth hormone receptor antagonists, and dopamine agonists, available in injectable or oral forms. Common drugs such as octreotide, lanreotide, and pegvisomant help control hormone levels and alleviate symptoms like enlarged hands, feet, and facial features. These treatments work by inhibiting growth hormone secretion or blocking its action on tissues. Advanced formulations offer extended-release mechanisms for reduced dosing frequency and improved patient adherence.
Key Takeaways
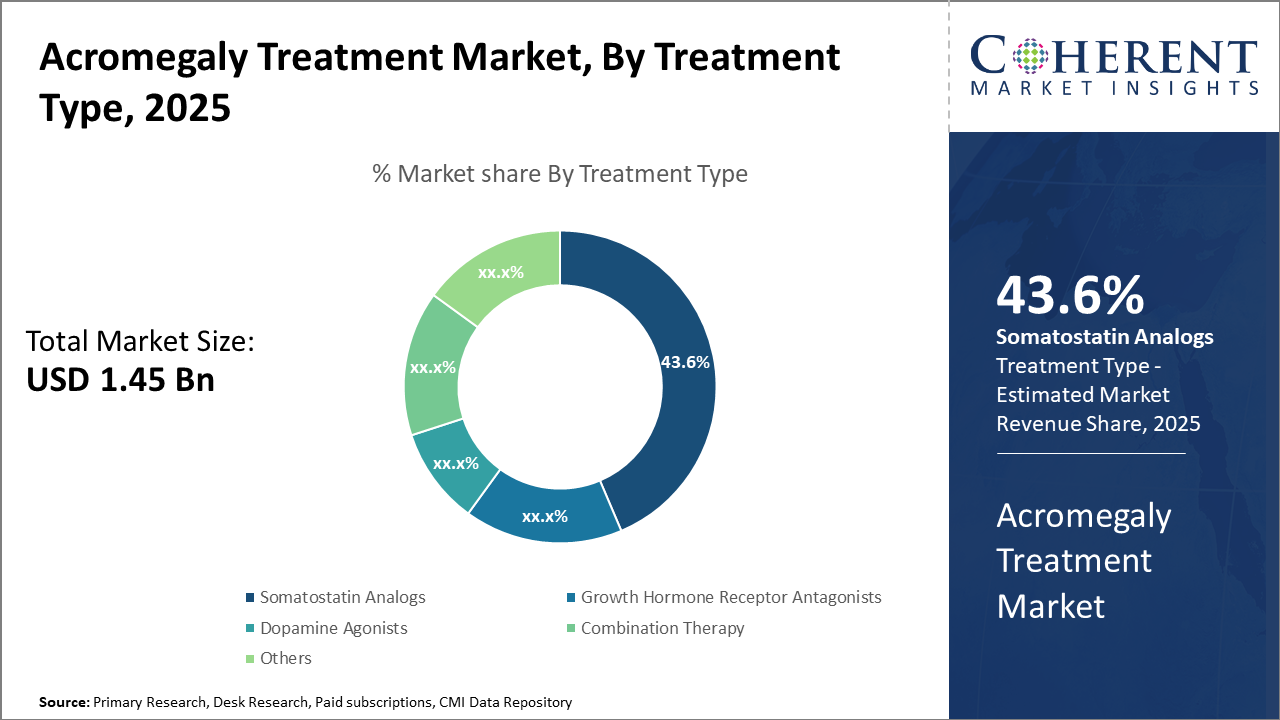
Market segments reflect a strong preference for somatostatin analogs, dominating over 43% of the industry share due to efficacy and established clinical protocols.
Injectable routes continue to prevail as the leading administration method due to optimized pharmacokinetics and patient outcomes.
Hospitals and clinics remain the largest end-user segment, supported by integrated treatment workflows and specialized care expertise.
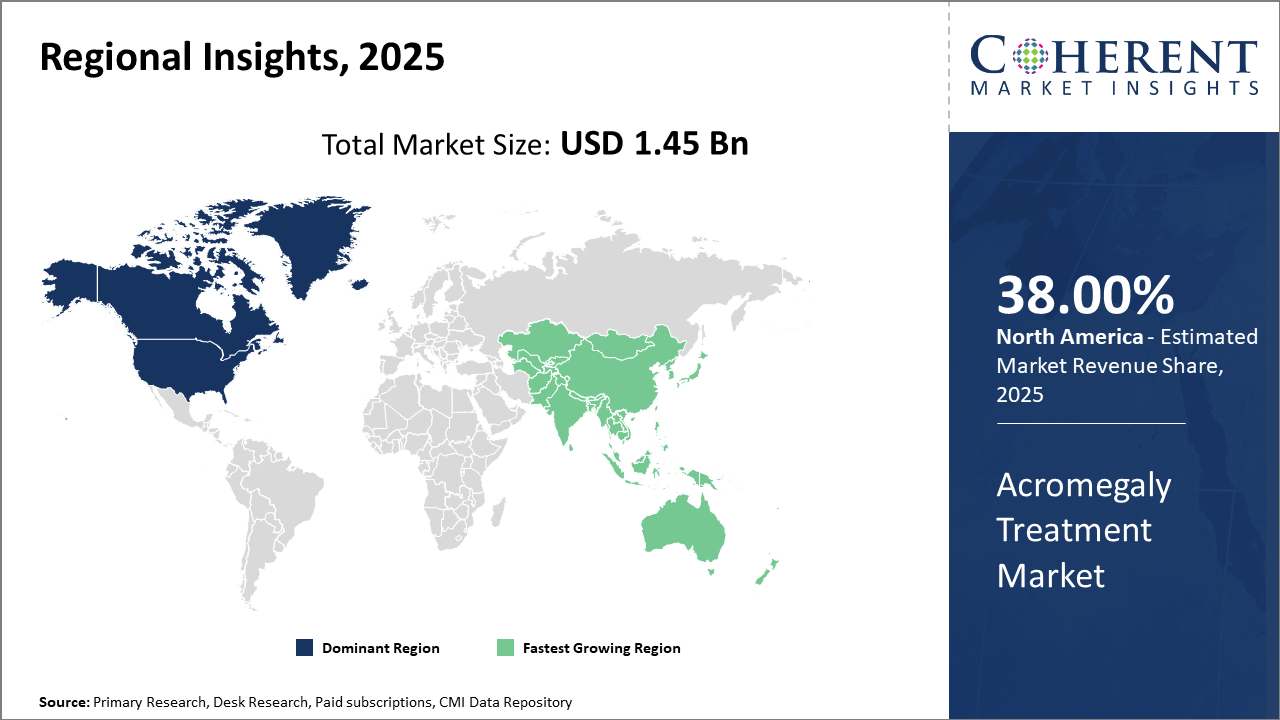
Regionally, North America accounts for approximately 38% industry share, propelled by advanced healthcare ecosystems and reimbursement frameworks.
Asia Pacific is identified as the fastest-growing region, registering a significant CAGR due to increasing healthcare infrastructure investments and broadened patient awareness.
These key takeaways offer critical market insights to support targeted growth initiatives.
Acromegaly Treatment Market Segmentation Analysis
To learn more about this report, Download Free Sample
Acromegaly Treatment Market Insights, By Treatment Type
Somatostatin Analogs dominate the market share with 43.6%, due to their proven efficacy in inhibiting excess growth hormone release, supported by rising prescription trends globally. The fastest-growing subsegment is Combination Therapy, which has seen increased clinical adoption due to its enhanced efficacy in drug-resistant acromegaly cases, driven by robust outcomes reported in 2025 trials. Growth Hormone Receptor Antagonists provide an alternative for patients intolerant to analogs, whereas Dopamine Agonists are occasionally prescribed in mild cases, maintaining niche usage. Others comprise emerging therapies still in clinical development stages.
Acromegaly Treatment Market Insights, By Route of Administration
Injectable administration is the dominant segment, attributed to its high bioavailability and established use; it accounted for over 58% revenue share, given patient preference for long-acting formulations that reduce treatment burden. The oral route represents the fastest-growing subsegment, spurred by recent approvals of oral somatostatin analog formulations, which simplify administration, increase patient compliance, and expand treatment accessibility beyond hospital settings.
Acromegaly Treatment Market Insights, By End-User
Hospitals & Clinics dominate market share due to centralized diagnostic facilities and comprehensive care teams managing complex acromegaly cases. Specialty Clinics are the fastest-growing subsegment owing to the rise of dedicated endocrinology centers, improving specialized patient management and follow-up care. Home Care Settings account for a smaller but steadily growing share, supported by increasing adoption of patient-friendly therapies and telehealth services.
Acromegaly Treatment Market Trends
The acromegaly treatment market is witnessing significant evolution marked by three key trends. First, the adoption of oral formulations as an alternative to injectable drugs is gaining rapid traction, exemplified by regulatory approvals in the U.S. and Europe since late 2024, leading to improved patient compliance and broadened market adoption.
Second, digital health integration including telemedicine and adherence monitoring tools has emerged as a niche but rapidly growing segment, with clinical centers reporting a 14% increase in treatment adherence due to these technologies in 2025.
Third, globalization of clinical trials, particularly in Asia Pacific, is boosting R&D productivity and accelerating market launch cycles for novel therapies.
Acromegaly Treatment Market Insights, By Geography
To learn more about this report, Download Free Sample
North America Acromegaly Treatment Market Analysis and Trends
In North America, the dominance in the Acromegaly Treatment Market is attributed to advanced healthcare infrastructure, comprehensive insurance reimbursement, and high awareness levels. The region holds approximately 38% market share globally, with the U.S. spearheading innovation and patient access. Established pharmaceutical companies have bolstered their presence through targeted product launches and strategic collaborations, ensuring sustained market leadership.
Asia Pacific Acromegaly Treatment Market Analysis and Trends
Meanwhile, Asia Pacific exhibits the fastest growth with a CAGR surpassing 8.2%, driven by expanding health insurance coverage, increasing disease awareness, and rising government investments in rare disease treatment ecosystems. Countries like China and India are witnessing notable increases in treatment initiation rates, supported by collaborations between local and international pharmaceutical companies facilitating market penetration.
Acromegaly Treatment Market Outlook for Key Countries
USA Acromegaly Treatment Market Analysis and Trends
The USA’s acromegaly treatment market is the largest globally, propelled by advanced diagnostic capabilities and extensive insurance coverage contributing to over 30% of the total market revenue in 2024. Key companies such as Novartis and Ipsen have introduced novel formulations and digital support tools targeting American healthcare providers, which has augmented market share through improved patient outcomes. Additionally, regulatory support and accelerated approval pathways for rare disease drugs continue to stimulate vibrant pipeline activities.
Germany Acromegaly Treatment Market Analysis and Trends
Germany’s acromegaly treatment market represents the leading position within Europe, accounting for nearly 18% of the regional market share. A strong presence of specialized endocrine centers and well-established reimbursement frameworks fuel prescription growth. Strategic alliances between pharmaceutical companies and healthcare providers in Germany have enhanced patient access programs, resulting in a 9% year-over-year increase in revenue during 2025. The country’s emphasis on personalized medicine is also reshaping treatment protocols favorably.
Analyst Opinion
Pricing Dynamics and Drug Accessibility: Recent data from 2024 indicate that the average treatment cost for acromegaly medications has seen a marginal decrease of 3% owing to competitive generic entries and improved biosimilar availability. This shift in pricing dynamics has enhanced treatment accessibility in emerging markets by approximately 15%, impacting market share distribution positively.
Advanced Drug Delivery Systems: Injectable prolonged-release formulations accounted for nearly 58% of total market revenue in 2024, driven by patient preference for less frequent dosing schedules. Supporting this, pharmaceutical companies reported a 22% increase in the adoption of implantable delivery devices in clinical settings during 2025, signifying strong demand for convenience-oriented therapeutic approaches.
Growing Use Cases in Comorbid Conditions: The application of acromegaly treatments among patients with coexisting cardiovascular and metabolic disorders rose by 12% in 2025, reflecting the growing recognition of integrated disease management strategies. Clinical treatment protocols in major hospitals suggest this trend has enhanced overall market revenue due to medication adherence improvements in these patient subsets.
Import-Export Flux and Market Penetration: Export volumes of acromegaly-specific therapies from North America increased by 8.5% in 2024, reflecting robust supply chain efficiencies and expanding trade agreements with Asia Pacific regions. In contrast, import data reveal a 10% uptick in Latin America’s pharmaceutical imports, indicating evolving demand-side growth factors aligned with healthcare infrastructure development.
Market Scope
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2025: | USD 1.45 billion |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2025 To 2032 |
| Forecast Period 2025 to 2032 CAGR: | 6.8% | 2032 Value Projection: | USD 2.34 billion |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: | Novartis AG, Pfizer Inc., Ipsen Group, Recordati S.p.A., Chiasma Inc., Ferring Pharmaceuticals, AstraZeneca PLC, Sun Pharmaceutical Industries Ltd., Merck KGaA, UCB S.A., HRA Pharma. | ||
| Growth Drivers: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Acromegaly Treatment Market Growth Factors
The steady rise in global acromegaly prevalence, driven by enhanced diagnostic protocols using IGF-1 biomarkers, is a primary growth catalyst. Studies from 2025 indicate improved early diagnosis rates by nearly 20%, contributing directly to higher treatment initiation volumes. Increasing healthcare expenditure on rare disease therapeutics, notably in North America and Europe, accelerates market expansion, supported by government reimbursement policies that boost patient access. Additionally, technological advancements in drug delivery—especially oral and long-acting injectable forms—address adherence challenges, fostering increased physician and patient acceptance. Lastly, growing investments in clinical trials focusing on combination therapies underscore the evolving therapeutic landscape, driving significant market revenue uplift anticipated through 2032.
Acromegaly Treatment Market Development
In September 2025, Crinetics Pharmaceuticals secured FDA approval for Palsonify (paltusotine), the first once-daily oral therapy for adults with acromegaly who either did not respond adequately to surgery or couldn’t have surgery. It is a somatostatin receptor type-2 (SST2) nonpeptide agonist. In clinical trials (PATHFNDR-1 & PATHFNDR-2), it showed rapid, durable, and consistent biochemical control of IGF-1 and GH levels, along with reduced symptom burden.
In October 2024, Camurus AB had its drug Oclaiz™ (CAM2029), “octreotide subcutaneous depot” formulation’s NDA (New Drug Application) accepted by the FDA for treatment of acromegaly; its PDUFA date was set for October 21, 2024. Oclaiz is designed for once-monthly self-administration with enhanced octreotide plasma exposure, intended to reduce treatment burden compared to more frequent injections.
Key Players
Leading Companies of the Market
Novartis AG
Pfizer Inc.
Ipsen Group
Recordati S.p.A.
Chiasma Inc.
Ferring Pharmaceuticals
AstraZeneca PLC
Sun Pharmaceutical Industries Ltd.
Merck KGaA
UCB S.A.
HRA Pharma
Competitive strategies among leading market players emphasize portfolio diversification and mergers & acquisitions to strengthen their foothold. For instance, Novartis’ strategic acquisition of specialized biotech firms in 2024 accelerated its R&D capabilities, resulting in a 9% increase in market revenue share. Ipsen Group leveraged exclusive licensing agreements in North America, contributing to an 11% year-over-year sales uplift within acromegaly treatments. Pfizer’s investment in next-generation formulations, focusing on patient-centric delivery systems, boosted its product adoption rates by 18% in key European markets during 2025.
Acromegaly Treatment Market Future Outlook
Future progress in acromegaly treatment is expected to focus on precision medicine, targeted biologics, and improved drug delivery systems. Research into combination therapies and oral formulations may enhance convenience and efficacy. The use of digital health tools for remote hormone monitoring and patient management will likely expand. Gene-based and peptide therapies could offer more durable control over hormone levels. Continued clinical research and innovation are expected to provide safer, more effective, and individualized treatment solutions, driving better long-term outcomes for acromegaly patients worldwide.
Acromegaly Treatment Market Historical Analysis
Historically, acromegaly treatment relied heavily on surgical interventions and radiation therapy to manage excess growth hormone secretion caused by pituitary adenomas. With advancements in biotechnology and pharmacology, medical therapies such as somatostatin analogs, dopamine agonists, and growth hormone receptor antagonists emerged, transforming disease management. The introduction of long-acting injectable formulations significantly improved patient compliance and outcomes. Over time, the integration of diagnostic imaging and hormone assays enhanced early detection and treatment precision. These developments collectively reduced disease complications and improved the quality of life for patients.
Sources
Primary Research interviews:
Endocrinologists
Clinical Pharmacologists
Hospital Procurement Specialists
Biopharma Researchers
Databases:
National Center for Biotechnology Information (NCBI)
World Health Organization (WHO) Database,
GlobalData Healthcare Reports
Magazines:
PharmaTimes
Pharmaceutical Technology
BioPharma Dive
Endocrine Today
Journals:
The Journal of Clinical Endocrinology & Metabolism
Endocrine Reviews
Hormone Research in Pediatrics
Clinical Endocrinology Journal
Newspapers:
The Economic Times (Healthcare)
The Hindu Business Line (Pharma)
The Guardian (Health)
The New York Times (Science & Health)
Associations:
The Endocrine Society
Pituitary Network Association
American Association of Clinical Endocrinology (AACE)
International Society of Endocrinology (ISE)
Share
Share
About Author
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Missing comfort of reading report in your local language? Find your preferred language :
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
Select a License Type
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients