The Global Ablation Devices Market was valued at USD 4.86 Bn in 2025 and is expected to reach USD 9.59 Bn by 2032, growing at a compound annual growth rate (CAGR) of 10.20% from 2025 to 2032.
The Global Ablation Devices Market is growing due to rising cardiovascular diseases, technological advancements, and an aging population. Ablation devices remove abnormal tissue minimally invasively, avoiding surgery. Radiofrequency ablation (RFA) uses high-frequency waves to heat and destroy tumors in organs like the liver and lungs, offering quick recovery. Microwave ablation (MWA) uses microwaves to more effectively treat larger tumors. For instance, in 2023, microwave ablation was successfully used to shrink a large liver tumor, demonstrating its effectiveness and patient benefits.
|
Event |
Description and Impact |
|
Technological Advancement and Innovation Wave |
|
|
Regulatory Landscape Evolution |
|
|
Global Healthcare Infrastructure Transformation |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
The Ablation Devices Market in Cardiac Arrhythmia is witnessing robust innovation, particularly in Ablation Devices for Atrial Fibrillation. Several radiofrequency (RF) ablation devices like the KODEX-EPD (EPD Solutions) and VARIPULSE Pulsed Field Ablation System (Biosense Webster) are in late-stage clinical trials, targeting improved efficacy and safety for atrial fibrillation treatment. Cryoablation devices such as Boston Scientific’s POLARx Cryoablation System are also progressing through global Phase III trials.
In the realm of Ablation Devices by Interventional Radiology, microwave ablation systems like Perseon Medical’s PERSEON device are undergoing Phase II trials for liver tumor applications, expanding minimally invasive cancer therapies. Laser ablation devices, including Monteris Medical’s LITT-MR system for brain tumors, are advancing MRI-guided intraoperative ablation approaches.
Emerging pulsed field ablation (PFA) technologies, including Boston Scientific’s FARAPULSE and Galaxy Medical’s CENTAURI systems, show promise in atrial fibrillation management with ongoing pivotal and feasibility studies. Ultrasound ablation devices, such as InSightec’s ExAblate Neuro, are in pivotal trials for neurological disorders, highlighting diversification of ablation applications.
Next-generation intraoperative ablation devices, like AngioDynamics’ IRE NanoKnife for pancreatic cancer, are entering early-phase clinical evaluations, demonstrating the market’s expansion into oncology and other specialties.
An analysis of the ablation devices market shows innovation efforts are centered around device enhancement to improve user experience and optimize patient outcomes. Most key patents relate to improvements in electrodes that are designed to achieve controlled and uniform lesion formation as with advanced electrodes.
Novel applications related to multi-electrode arrays and energy delivery systems providing temperature-controlled delivery with precision ablation system released within the last 3 years. Other innovations include AI-assisted navigation and real-time imaging systems aimed at enhancing guidance accuracy.
Differentiation among leading device manufacturers is expected as a result. Moreover, some patents safeguard advanced flexible and user-friendly minimally invasive catheters tailored to the rising demand for outpatient and intraoperative ablation procedures. From a geographical standpoint, patent applications are primarily filed in the United States, Europe, and Japan, which is indicative of a strong regulatory framework coupled with market opportunity.
There is a growing number of licensing agreements and cross-licensing deals initiated by startups and experienced medical device firms which is expediting innovation and entry into the market. To sum up, the active patent applications and grants illustrate the competitive framework within the ablation devices market and the fundamental importance of patents in acquiring and defending technological market leadership drive competition.
The Ablation Devices Market faces a global reimbursement system which is intertwined with device adoption and patient access. The blank reimbursement is attributed to ablative procedures CPT codes which contain of pathologic modalities, for example CPT 93656 for atrial fibrillation and CPT 47370 for liver tumor ablation. It is also facilitated by supporting diagnosis codes from ICD-10 and device specific hcpcs codes.
In the US, reimbursement policies set by CMS NCDs and LCDs, are likely to cover these ablation procedures within Medicare Parts A and B. Medicare covers catheter ablation procedures at an estimated price of 15,000 to 25,000 dollars and pays approximately 80 percent of the approved costs post deductible, while the patient coinsurance is 20 percent on average. Medicaid coverage differs from state to state but does provide coverage for most ablation procedures.
In Europe, MCDA decisions are made by the national HTA bodies such as NICE UK and G-BA Germany, working within EMA regulatory frameworks. In the Asia pacific region, Japan’s MHLW and China’s NMPA supervise a fragmented reimbursement ecosystem, which is becoming more supportive as ablation therapies are increasingly used. As discussed, the objectives regarding other regions highlight the importance that these policies have on commercial and widespread use of ablation devices, irrespective of oncology and cardiac arrhythmia indications.
Factors influencing prescribers in the Ablation Devices Market for Cardiac Arrhythmia Management are device efficiency, safety, procedural intricacy, and patient-centered considerations. During early stage atrial fibrillation (AF), pharmacological options such as Class IC antiarrhythmics and beta-blockers are still the most prescribed.
However, in the case of young and symptomatic patients with paroxysmal AF, catheter ablation is increasingly viewed as first-line treatment, especially when quality of life is significantly impacted. For secondary treatment lines, when medications are ineffective or intolerable, there is a preference for ablation devices equipped with contact-force sensing catheters like ThermoCool SmartTouch® and TactiCath®. In the setting of atrial flutter, conventional radiofrequency ablation of the cavotricuspid isthmus is commonly performed because of its high rate of success.
These cases are often more resistant such as persistent AF or long-standing AF where ablation is the dominant strategy. Physicians frequently use advanced mapping systems such as CARTO® or EnSite® along with contact-force catheters and in some cases, employ hybrid strategies using surgically implanted devices like AtriCure with catheter ablation.
For Ventricular Tachycardia, the preferred treatment involves the use of irrigated-tip catheters which enable or promote effectiveness in complex arrhythmias. In all stages of arrhythmias, technology is focused on improving clinical and procedural outcomes which remains the highest priority for all prescribers.
The radiofrequency ablation segment leads the ablation devices market with a significant 35.5% revenue share in 2025. This dominance is driven by its widespread use in treating cancerous tumors and cardiovascular diseases due to its minimally invasive nature and faster patient recovery. Technological advancements and growing clinical adoption further support the segment’s leadership.
Microwave ablation holds a notable share as well, favored for its effectiveness in treating larger tumors. Other technologies, including cryoablation, ultrasound ablation, and hydrothermal ablation, contribute to the market but have comparatively smaller shares. Continuous innovation and expanding treatment applications are expected to fuel growth across these segments.
To learn more about this report, Download Free Sample
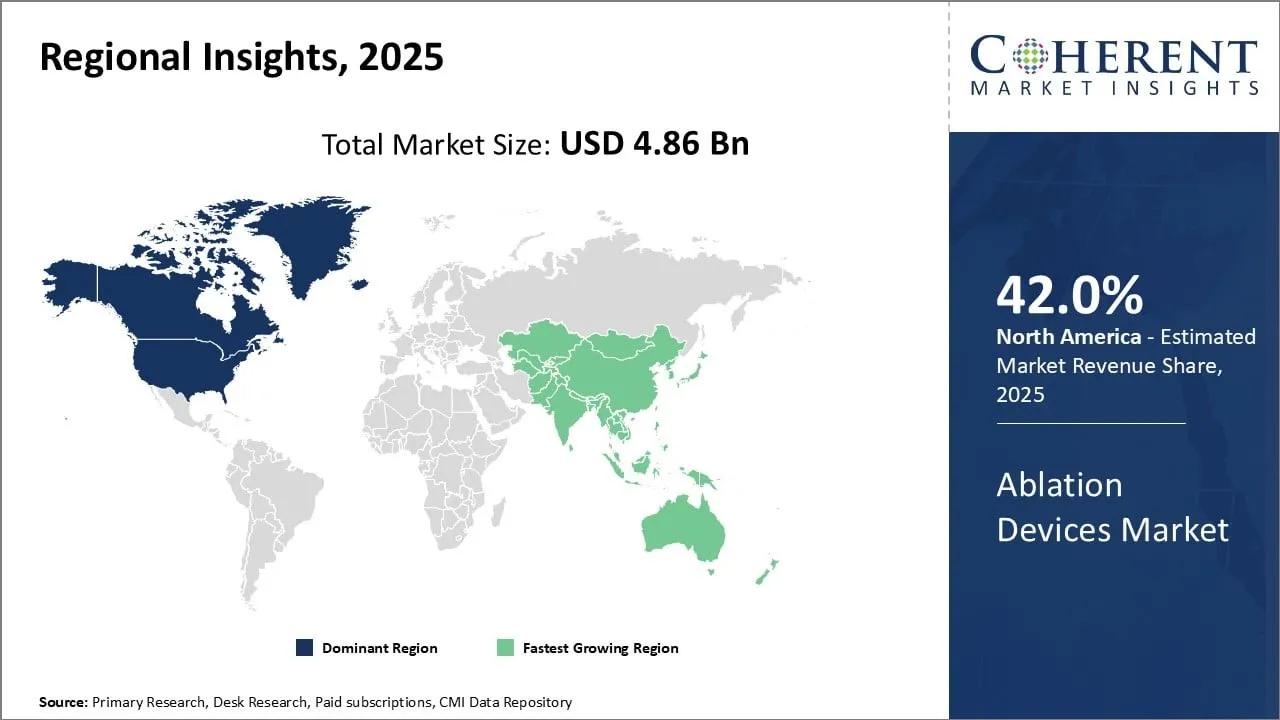
North America leads the global ablation devices market, holding a dominant 42.0% share in 2025. This leadership stems from the growing preference among physicians for laser ablation techniques to treat conditions such as atrial fibrillation and cancerous tumors. Laser ablation offers enhanced precision and control over tissue removal compared to other technologies, driving its widespread adoption. The region benefits from advanced healthcare infrastructure, strong clinical expertise, and high healthcare expenditure, supporting continued market growth.
Europe is the second-largest market for ablation devices, capturing a 29% share in 2025. The market expansion is primarily driven by the extensive adoption of radiofrequency ablation therapy for cardiac arrhythmias. Favorable reimbursement frameworks, increasing awareness among healthcare providers, and ongoing technological innovations in ablation therapy contribute to the steady growth of the market across European countries.
Asia Pacific holds the third-largest share at 18% in 2025, with rapid growth fueled by the rising use of ultrasound ablation technology. This technology’s safety and precision advantages make it a preferred choice for treating certain medical conditions. Improving healthcare infrastructure, increased investment in medical technologies, and growing awareness of minimally invasive treatment options are key factors driving market adoption in the region.
The U.S. dominates the North American market, driven by widespread use of laser ablation procedures and strong healthcare R&D investments. High adoption rates are supported by a large patient pool with cardiac and oncological conditions and well-established reimbursement policies.
Germany leads the European ablation devices market, supported by its advanced healthcare system and extensive use of radiofrequency ablation therapy. The country’s focus on clinical research and technology adoption ensures sustained growth in this segment.
China is a key market in Asia Pacific, with growing adoption of ultrasound ablation driven by expanding healthcare infrastructure and increased access to advanced medical technologies. Government initiatives promoting healthcare modernization and rising patient awareness contribute to the rapid market expansion.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2024 | Market Size in 2025: | USD 4.86 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2025 To 2032 |
| Forecast Period 2025 to 2032 CAGR: | 10.2% | 2032 Value Projection: | USD 9.59 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Medtronic, Johnson & Johnson Services, Inc., Abbott, Smith+Nephew., Boston Scientific Corporation, AngioDynamics, ATRICURE, INC., Olympus Corporation, CONMED Corporation, Merit Medical Systems |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
The increasing incidence of cancer cases around the world is a major factor fueling the expansion of the global ablation devices market. Cancer has become one of the leading causes of death worldwide, with nearly 10 million deaths in 2020, according to the World Health Organization. The growing burden of various types of cancer such as liver cancer, lung cancer, kidney cancer, and others, is driving greater demand for effective ablation techniques that can help in cancer treatment.
Ablation techniques have emerged as viable, minimally invasive alternatives to surgery in recent years for cancer treatment. Ablation devices help destroy cancerous tumors or unnecessary tissues by imparting heat or cold. Various devices in this market, such as radiofrequency ablation devices, microwave ablation devices, cryoablation devices, hydrothermal ablation devices, and others, are being widely adopted.
This is due to advantages such as shorter hospital stays, reduced recovery time, and less pain for patients compared to conventional surgery. The rising preference for minimally invasive procedures will aid in the increasing incorporation of ablation techniques in clinical oncology.
Ablation treatment is increasingly being used for age-related chronic diseases and conditions like atrial fibrillation, cancer, and cardiovascular diseases, which are highly prevalent in older age groups. For example, according to the World Health Organization (WHO), atrial fibrillation, a type of irregular heartbeat that can lead to strokes, heart failure, and other complications, affects around 6 million people in the European Region alone, and its incidence rises with age from less than 1% at age 50 to almost 10% at age 80. Similarly, the American Cancer Society (ACS) estimates that around 66% of all cancers are diagnosed in people aged 65 years or older.
Ablation offers minimally invasive techniques to treat such diseases with fewer side effects than alternative options like open surgeries for the aging population. As people across the world opt for radiofrequency, cryoablation, microwave, and other catheter-based techniques over surgical treatments, the demand and scope for the application of ablation systems for chronic disease management in older age segments are surging rapidly. This steady rise in diseases and conditions afflicting the ballooning senior citizen demographic globally will remain a key driver propelling the multibillion ablation devices industry forward in the forthcoming years.
The global market for ablation devices is receiving a boost due to new and emerging technologies for ablation devices. Ablation is a minimally invasive procedure that employs technologies such as radiofrequency, laser and light, ultrasound, and others to destroy the tissues that are damaged.
The latest generations of ablation devices tend to enable physicians to manage complex medical problems more accurately and more effectively. For example, some modern laser systems make it possible to ablate tumors in the lungs- even in inaccessible locations, which aids considerably in making surgical cuts.
These improvements make the risks and recovery times faster and better for patients. One additional aspect of importance as to why the global market for ablation devices is expanding is the development of Magnetic Resonance Guided Focused Ultrasound (MRgFUS ) technology by prominent players within the industry. MRgFUS incorporates MRI imaging and focused ultrasound beams to non-invasively ablating tissues like fibroids at the uterus. MRgFUS is gaining acceptance for essential tremors and bone metastases.
Unlike conventional surgical procedures, MRgFUS Does not introduce any form of malignancy. According to the U.S. National Institute of Biomedical Imaging and Bioengineering (2020), more than 20,000 patients had received treatment with gynecological MR-guided focused ultrasound systems worldwide by 2020.
The emerging economies in Asia Pacific and Latin America regions are well positioned to drive significant growth in the global ablation devices market in the coming years. Countries like India, China, Brazil, and Mexico have very large patient populations combined with improving access to healthcare facilities. As disposable incomes and standards of living rise rapidly in these developing nations, access to advanced medical technologies and procedures will also increase dramatically.
Ablation devices are increasingly used in minimally invasive treatments for cancer, cardiac arrhythmias, and other conditions. As the burden of non-communicable diseases grows in the developing world due to lifestyle changes and aging populations, demand for ablation technologies from physicians and patients will continue to surge. These emerging markets still have much lower device utilization and procedure rates compared to developed countries currently. This indicates a massive potential for future procedure volume growth as affordability barriers reduce over time.
Growing applications of ablation devices in a variety of medical procedures have created significant opportunity in the global ablation devices market. Ablation procedures are increasingly being used as minimally invasive alternatives for surgeries across various therapeutic areas like cardiology, gynecology, urology, and many others.
According to data published in June 2024, the Center for Disease Control and Prevention, cancer incidence and mortality rates have declined annually from 2016 to 2019 in the U.S., partly due to early detection and advanced interventional procedures like ablation that help treat cancer without the need for major invasive surgeries.
This ablation devices are becoming the standard of care for treating arrhythmias and replacing the need for lifelong medication. According to the World Health Organization, cardiovascular diseases are the leading cause of mortality worldwide, responsible for over 17 million premature deaths annually. Ablation offers effective treatment with high success and lower risks than alternative drug therapies or extensive surgical procedures for heart conditions. Industry projections suggest more than half a million cardiac ablation procedures are performed worldwide each year, and this figure is expected to grow substantially in the coming years.
It is another promising area for ablation devices. The National Cancer Institute estimated that there were over 1.8 million new cancer cases diagnosed in the U.S. in 2020. Minimally invasive techniques such as ablation help address the increasing cancer burden while offering patients less trauma, quicker recovery times, and potentially lower costs compared to open surgeries. The ability of ablation technologies to treat both benign and cancerous tumors across different parts of the body without the need for general anesthesia makes them an attractive option.
The rural communities across the globe present a massive untapped market potential for ablation devices. With a major chunk of the world's population residing in rural and semi-urban areas, enabling access to advanced medical technologies in these regions can help address significant unmet healthcare needs.
Ablation, being a minimally invasive treatment option for several cardiovascular, oncological, and urological conditions, has the ability to bring effective solutions closer to patients living in remote locations. At present, a lack of proper infrastructure and skilled medical practitioners restrict the availability of ablation devices and associated procedures only to major urban centers. However, with coordinated efforts of governments, non-profit organizations, and private players, this barrier can be overcome.
Telehealth and telerobotics offer promising options to virtually connect patients in rural clinics to expert interventional radiologists and electrophysiologists based in cities. Mobile care facilities and training of local healthcare workers will also help in conducting simple ablation treatments near patients' homes. This localized model of decentralized healthcare can become highly beneficial for aging populations in underdeveloped regions.
According to the WHO, non-communicable diseases account for over 70% of deaths globally, with a disproportionately higher burden on poorer nations. Addressing this critical need through innovations in rural ablation care can boost quality of life as well as health outcomes.
As awareness and acceptance grow amongst rural communities, manufacturers stand to gain substantial returns on investments made in building local supply chains and post-sales support systems tailored for remote infrastructure. Recent initiatives by the Australian and Canadian governments to improve telehealth infrastructure and skills in their rural heartlands indicate the potential opportunities if similar focused steps are taken elsewhere. This untapped rural growth segment can fuel the global expansion of the ablation devices industry in the long run.
The adoption of robot-assisted ablation devices is having a significant impact on the global ablation devices market. Robot-assisted systems allow for improved precision, consistency, and a reduced risk of complications compared to conventional manual ablation procedures. These advantages are driving increasing demand for robotic systems among healthcare facilities and physicians.
Robot-assisted devices allow electrophysiologists and surgeons to perform minimally invasive cardiac ablation procedures through small incisions or ports with enhanced visualization. This enables precise catheter manipulation and lesion delivery to complex anatomies that would otherwise be difficult to access manually. The robotic systems filter out any physiologic tremor and ensure steadier contact during ablation, helping achieve optimal treatment results. These capabilities are leading to higher success rates for treating atrial fibrillation and other cardiac arrhythmias. These examples indicate lucrative market potential and a promising outlook for robot-assisted ablation devices.
The rapidly evolving trends towards minimally invasive procedures for the treatment of cardiac arrhythmias and cancer have significantly contributed to the rising demand for radiofrequency ablation devices globally. With advancements in medical technology, radiofrequency ablation has emerged as one of the most preferred techniques for precisely targeting and destroying tumors or abnormal tissue through the application of heat energy. It allows for precise treatment with minimal procedural risks and associated complications for patients.
A key factor propelling the adoption of radiofrequency ablation devices is the growing preference for outpatient procedures among healthcare professionals as well as patients. Radiofrequency ablation can often be performed on an outpatient basis as it does not require long hospital recovery time. This has also strengthened its appeal as a cost-effective alternative to traditional open surgeries.
*Definition: Ablation devices are designed to ablate (remove or destroy) tissue for therapeutic purposes and often used in the treatment of various types of diseases and medical conditions, including cardiac arrhythmias, cancer tumors, and other dysfunctional tissues. These devices function by delivering energy to the targeted tissues and can employ various techniques based on the type of energy they utilize, such as radiofrequency ablation, cryoablation, laser ablation, microwave ablation, electrical ablation, ultrasound ablation, etc.
Share
Share
About Author
Komal Dighe is a Management Consultant with over 8 years of experience in market research and consulting. She excels in managing and delivering high-quality insights and solutions in Health-tech Consulting reports. Her expertise encompasses conducting both primary and secondary research, effectively addressing client requirements, and excelling in market estimation and forecast. Her comprehensive approach ensures that clients receive thorough and accurate analyses, enabling them to make informed decisions and capitalize on market opportunities.
Missing comfort of reading report in your local language? Find your preferred language :
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients