Transcatheter Aortic Valve Replacement Market Size and Forecast – 2026 to 2033
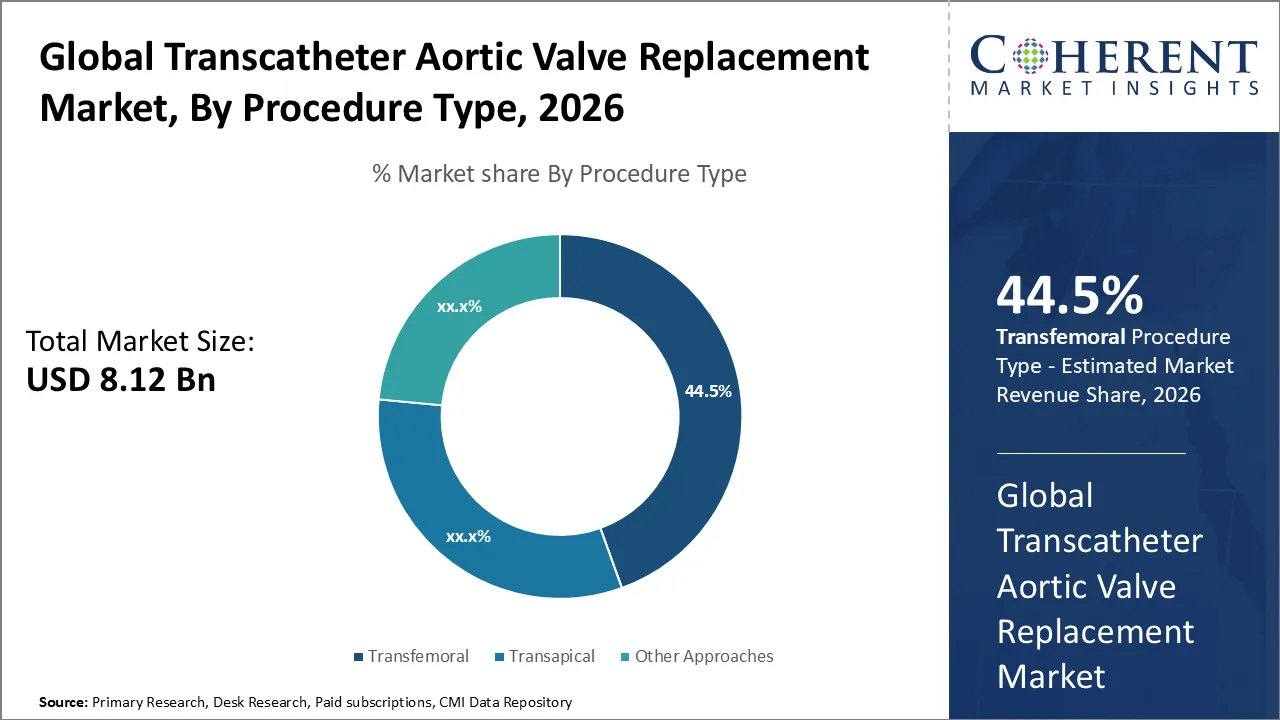
The Transcatheter Aortic Valve Replacement Market is estimated to be valued at USD 8.12 Bn in 2026 and is expected to reach USD 13.76 Bn by 2033, exhibiting a compound annual growth rate (CAGR) of 8.2% from 2026 to 2033.
Key Takeaways
- Based on Procedure Type, Transfemoral segment is projected to account for 44.5% of the global market in 2026, owing to its minimally invasive nature and faster recovery times compared to traditional surgical methods.
- Based on Device Type, Balloon-Expandable Valves segment is expected to capture 57%share of the market in 2026, due to their quicker implantation and track record of positive clinical outcomes.
- By End User, Hospitals are projected to account 39.2% share in 2026, due to state-of-the-art infrastructure and multidisciplinary teams.
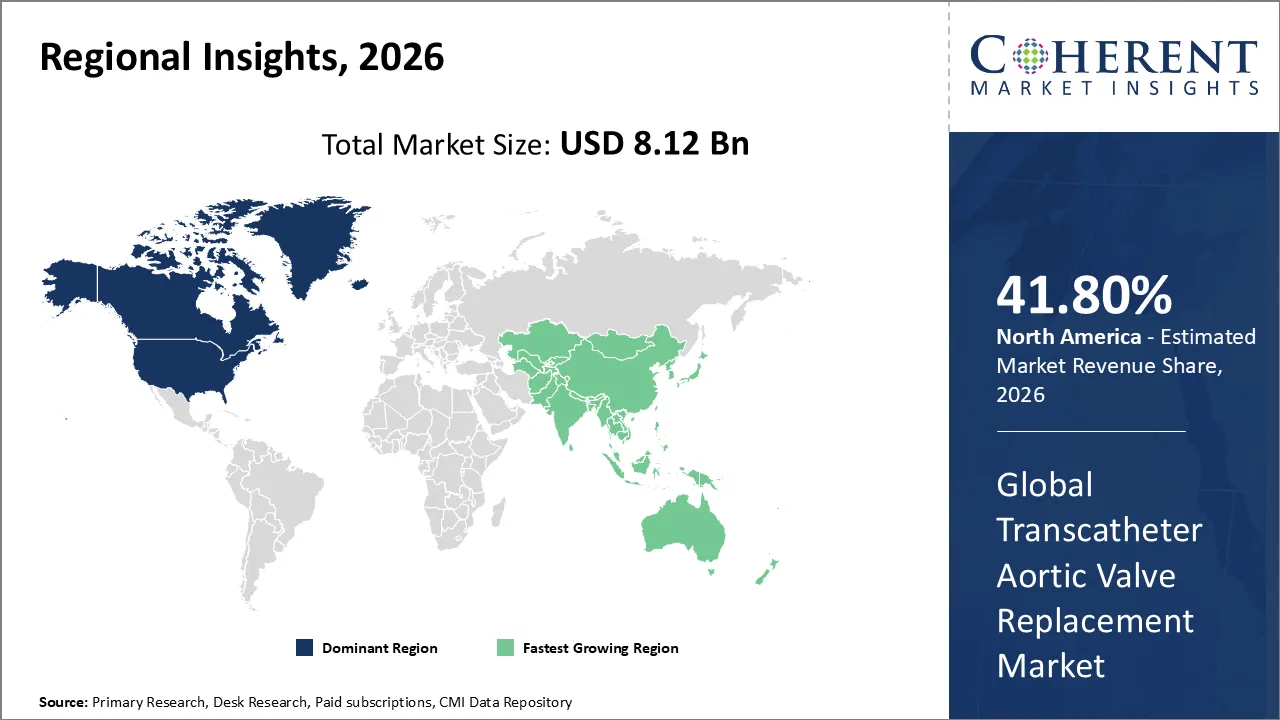
- Based on Region, North America is set to dominate the global market with a 41.8% share in 2026 owing to the favorable reimbursement & insurance support.
Market Overview
The Transcatheter Aortic Valve Replacement (TAVR) market continues to grow as healthcare providers increasingly adopt minimally invasive cardiac procedures to treat aortic stenosis, especially in elderly and high-risk patients. Innovations in valve design, delivery systems, and imaging enhance procedural safety and clinical outcomes, encouraging wider acceptance. Rising hospital demand, improved physician expertise, supportive reimbursement policies in developed regions, and continuous clinical research collectively drive global market expansion and innovation.
Current Events and Its Impact on the Transcatheter Aortic Valve Replacement Market
|
Current Events |
Description and its impact |
|
Regulatory and Policy Developments |
|
|
Technological Advancements and Innovation |
|
|
Competitive Landscape Shifts |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Reimbursement Analysis in Transcatheter Aortic Valve replacement
Transcatheter aortic valve replacement (TAVR) reimbursement and economic impact have been extensively analyzed in health services research. In the U.S., Medicare is the dominant payer for TAVR, and procedure reimbursement varies widely by institution, with reported CMS payments ranging roughly from ~$28,000–$111,000 per case (median ≈ $45,884). Higher reimbursement alone was not independently linked to use of additional devices like embolic protection once site factors were adjusted, with procedural volume more strongly associated with practice patterns than payment level.
Transcatheter Aortic Valve Replacement Market Insights, By Procedure Type: Transfemoral contribute the highest share of the market owing to its minimally invasive nature & clinical advantages.
Transfemoral segment is projected to account for 44.5% of the global market in 2026. The transfemoral route in Transcatheter Aortic Valve Replacement (TAVR) is becoming increasingly popular because it minimizes procedural trauma and speeds patient recovery. Advancements in delivery systems allow clinicians to place valves more precisely and treat a wider range of anatomies. Clinical studies show improved safety and outcomes, prompting physicians to adopt this approach. Hospitals implement it to shorten stays and reduce complications, while growing awareness, an aging population, and robust healthcare infrastructure continue to expand its use as the preferred TAVR access route. For instance, in April 2025, Edwards Lifesciences announced that its SAPIEN M3 mitral valve system received CE Mark for transcatheter treatment of symptomatic mitral regurgitation in patients unsuitable for surgery or TEER, becoming the world’s first transfemoral transcatheter valve replacement for MR.
Transcatheter Aortic Valve Replacement Market Insights, By Device Type: Balloon -Expandable Valves contribute the highest share of the market owing to its clinical advantages & procedural precision.
Balloon-Expandable Valves segment is expected to capture 57% share of the market in 2026. Clinicians increasingly choose balloon-expandable valves in Transcatheter Aortic Valve Replacement (TAVR) for their precise deployment and consistent performance, which minimize valve misplacement and paravalvular leakage. These valves deliver predictable outcomes and facilitate treatment in anatomically challenging patients. Ongoing innovations in valve design and delivery systems expand their use, while robust clinical evidence boosts physician confidence. Hospitals implement balloon-expandable valves to enhance procedural success, support faster patient recovery, and reinforce their standing in minimally invasive cardiac care. For instance, in September 2025, Edwards Lifesciences launched its transcatheter heart valve platform in India for aortic stenosis, featuring balloon-expandable valves with calcification-resistant tissue to reduce valve deterioration and reintervention.
Transcatheter Aortic Valve Replacement Market Insights, By End User: Hospitals contribute the highest share of the market owing to its advanced clinical infrastructure & capabilities.
Hospitals are projected to account 39.2% share in 2026. Hospitals actively adopt Transcatheter Aortic Valve Replacement (TAVR) by utilizing advanced infrastructure, such as hybrid operating rooms and specialized imaging systems, to perform complex procedures safely. They form multidisciplinary heart teams to provide comprehensive care from preoperative evaluation to postoperative recovery. High patient referrals and supportive reimbursement policies make TAVR financially sustainable, while extensive clinical experience increases physician confidence. Hospitals also implement TAVR to improve patient outcomes, minimize complications, and establish themselves as leading centers of excellence in cardiac care. For instance, Hinduja Hospital, Khar, became the first hospital in Mumbai/Western India authorized to perform TAVI without a Proctor, significantly reducing wait times for high-risk patients.
Regional Insights
To learn more about this report, Download Free Sample
North America Transcatheter Aortic Valve Replacement Market Trends
North America is set to dominate the global market with a 41.8% share in 2026. The North American Transcatheter Aortic Valve Replacement (TAVR) market grows as hospitals and clinics adopt minimally invasive procedures, supported by advanced healthcare infrastructure and early regulatory approvals for new devices. In the United States, active clinical research, frequent technology launches, and extensive reimbursement coverage drive adoption, while expanding healthcare capacity in Canada further increases uptake. Rising cases of aortic valve disease among the aging population, along with continuous innovation from leading manufacturers, strengthen the region’s position and drive higher procedural volumes. For instance, Medtronic announced the U.S. market expansion of its newest self-expanding Evolut™ FX TAVR system for transcatheter aortic valve replacement.
Asia Pacific Transcatheter Aortic Valve Replacement Market Trends
Countries across Asia Pacific are actively expanding the Transcatheter Aortic Valve Replacement (TAVR) market by strengthening healthcare infrastructure and increasing access to minimally invasive cardiac care. Healthcare providers perform more procedures as rising aortic valve disease prevalence among aging populations and growing awareness of TAVR benefits drive demand in China, Japan, India, and Southeast Asia. Regulators advance approvals, and local manufacturers increase device availability, while improved training programs and referral networks help clinicians integrate TAVR into standard cardiac practice throughout the region. For instance, in January 2025, Abbott launched the Navitor Vision in India, a cutting-edge valve for treating severe aortic stenosis in high-risk patients, advancing its transcatheter aortic valve (TAVI/TAVR) technology.
United States Transcatheter Aortic Valve Replacement Market Trends
Healthcare providers in the United States actively drive the Transcatheter Aortic Valve Replacement (TAVR) market by adopting minimally invasive therapy as an alternative to open-heart surgery, supported by advanced cardiovascular centers and growing clinical evidence of safety and effectiveness. Physicians perform TAVR on a wider range of patient risk profiles, leveraging FDA approvals and continuous device innovations. Favorable reimbursement policies and the high prevalence of aortic valve disease among aging Americans sustain procedural demand, while leading manufacturers introduce new technologies and form strategic partnerships to compete. For instance, JenaValve Technology, a California-based company developing transcatheter aortic valve replacement (TAVR) solutions, raised USD 100 million in a Series C financing round.
China Transcatheter Aortic Valve Replacement Market Trends
Hospitals and healthcare providers in China are actively expanding minimally invasive Transcatheter Aortic Valve Replacement (TAVR) procedures as advanced cardiac care units grow in major cities and referral networks strengthen, increasing procedural volumes. Rising cardiovascular disease prevalence and greater patient awareness boost demand, while local manufacturers and regulatory authorities enhance access to affordable valve technologies. Hospitals and clinicians also implement investments, clinical training programs, and strategic partnerships with global manufacturers to integrate TAVR into standard cardiac practice throughout the country.
End-user Feedback and Unmet Needs in the Transcatheter Aortic Valve Replacement Market
- Procedure Safety and Complication Management: End-users emphasize the need to further minimize procedural complications, such as vascular injury, paravalvular leakage, and conduction abnormalities. Clinicians seek devices and delivery systems that enhance deployment accuracy and reduce adverse events, ensuring patient safety while maintaining efficacy, especially in complex anatomies or high-risk populations.
- Device Versatility and Anatomical Adaptability: Physicians highlight limitations in treating patients with diverse anatomies, including small or heavily calcified vessels. There’s an unmet need for more versatile valves and delivery systems capable of adapting to varied anatomical challenges while maintaining optimal hemodynamic performance and ease of use across all patient populations.
- Training and Procedural Support: Healthcare providers report challenges in mastering TAVR procedures due to their technical complexity. End-users request enhanced training programs, simulation tools, and on-site procedural support to improve operator confidence, reduce learning curves, and standardize outcomes, particularly in emerging markets where TAVR experience remains limited.
Transcatheter Aortic Valve Replacement Market Trend
Expansion of Minimally Invasive Procedures
Healthcare providers are increasingly favoring TAVR over traditional open-heart surgery due to its minimally invasive nature, which shortens recovery times and reduces procedural risks. Hospitals are investing in hybrid operating rooms and advanced imaging to support complex interventions. The trend reflects growing physician confidence, rising patient preference, and broader adoption across intermediate- and low-risk populations, driving TAVR integration into standard cardiac care pathways across developed and emerging healthcare systems.
Technological Innovations in Valve Design
Manufacturers are introducing next-generation valves with enhanced sealing skirts, low-profile delivery systems, and improved hemodynamic performance. Innovations focus on precise deployment, reducing paravalvular leak, and accommodating diverse patient anatomies. These advances support wider clinical application, including in patients with challenging vascular access, and encourage physicians to adopt TAVR for broader populations. Continuous improvements in device reliability and ease of use differentiate offerings in a competitive market while supporting better long-term outcomes.
Transcatheter Aortic Valve Replacement Market Opportunity
Expansion to Low- and Intermediate-Risk Patients
With growing clinical evidence supporting safety and efficacy, TAVR is increasingly being considered for low- and intermediate-risk patients, beyond high-risk populations. This creates opportunities for hospitals and manufacturers to expand procedural volumes, introduce targeted patient education programs, and develop valves optimized for diverse anatomies. Physicians can now offer minimally invasive solutions to a broader population, increasing adoption and improving long-term outcomes while reinforcing the market’s growth potential in previously underserved segments.
Market Report Scope
Transcatheter Aortic Valve Replacement Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 8.12 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 8.2% | 2033 Value Projection: | USD 13.76 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Edwards Lifesciences Corp., Medtronic Plc., Abbott Laboratories., Boston Scientific Corp., MicroPort Scientific Corp., JenaValve Technology GmbH., Cardiovascular Systems, Inc., Symetis SA., Lepu Medical Technology Co., Ltd., Terumo Corporation., St. Jude Medical (now part of Abbott)., AtriCure, Inc., Vascular Solutions, Inc., Biotronik SE & Co. KG., and Cook Medical |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Analyst Opinion (Expert Opinion)
- The Transcatheter Aortic Valve Replacement (TAVR) market is advancing beyond conventional expectations, driven not merely by demographic tailwinds but by substantive clinical validation and competitive realignment. Procedural adoption has transcended its original high‑risk niche, with transfemoral access now accounting for an overwhelming majority of cases in mature centers (≥85–90%), and the United States contributing over half of global volumes, surpassing 100,000 annual procedures in recent years. Transfemoral TAVR consistently demonstrates lower in‑hospital mortality and shorter lengths of stay compared with surgical alternatives, reinforcing cardiologist confidence in its procedural efficacy and operational throughput.
- While market trackers report divergent growth projections, the underlying consensus points to robust technological momentum. Innovations such as retrievable and repositionable valves, reduced delivery profiles, and advanced imaging integration have materially reduced historical complications like paravalvular leak and pacing dependency, with experienced centers now reporting procedural success rates exceeding 95%.
- Competition has sharpened substantially. The recent U.S. FTC action blocking Edwards Lifesciences’ acquisition of JenaValve underscores regulatory scrutiny over perceived market concentration in next‑generation devices for conditions such as aortic regurgitation. This decision reflects concern that consolidation could stifle innovation and limit therapeutic alternatives for underserved patient subsets. At the same time, clinical setbacks—such as Boston Scientific’s withdrawal of certain TAVR systems following trial results indicating elevated adverse events—have temporarily redistributed share toward incumbents like Edwards, Medtronic and Abbott.
- Cost and access remain the most credible constraints to universal adoption. TAVR procedures typically incur 2–3× the cost of surgical replacements in developed markets, compounded by capital‑intensive infrastructure requirements and uneven reimbursement frameworks. These economic pressures are particularly acute in emerging markets where penetration rates remain below 5% of eligible patients.
- Strategically, the TAVR landscape is shifting from a product‑centric model to a system‑wide value proposition. Procedure optimization through conscious sedation protocols, same‑day discharge pathways, and computational tools for personalized valve selection is becoming a competitive differentiator. Investments in AI and patient‑specific planning are translating into measurable reductions in complication rates and improved long‑term outcomes.
Market Segmentation
- By Procedure Type Insights
- Transfemoral
- Transapical
- Other Approaches
- By Device Type Insights
- Balloon-Expandable Valves
- Self-Expanding Valves
- By End User Insights
- Hospitals
- Ambulatory Surgical Centers
- Independent Cardiac Catheterization Labs
- Others
- By Regional Insights
- North America
- U.S.
- Canada
- Latin America
- Brazil
- Argentina
- Mexico
- Rest of Latin America
- Europe
- Germany
- U.K.
- Spain
- France
- Italy
- Russia
- Rest of Europe
- Asia Pacific
- China
- India
- Japan
- Australia
- South Korea
- ASEAN
- Rest of Asia Pacific
- Middle East
- GCC Countries
- Israel
- Rest of Middle East
- Africa
- South Africa
- North Africa
- Central Africa
- Key Players Insights
- Edwards Lifesciences Corp.
- Medtronic Plc.
- Abbott Laboratories.
- Boston Scientific Corp.
- MicroPort Scientific Corp.
- JenaValve Technology GmbH.
- Cardiovascular Systems, Inc.
- Symetis SA.
- Lepu Medical Technology Co., Ltd.
- Terumo Corporation.
- Jude Medical (now part of Abbott).
- AtriCure, Inc.
- Vascular Solutions, Inc.
- Biotronik SE & Co. KG.
- Cook Medical
Sources
Primary Research Interviews
- Interventional Cardiologists
- Medical Device Manufacturers Executives
- Hospital Procurement Managers
- Cardiac Surgeons
- Others
Databases
- FDA Medical Device Database
- ClinicalTrials.gov
- Medicare Provider Analysis and Review (MEDPAR)
- Global Health Observatory (WHO)
- Others
Magazines
- Cardiovascular Business
- Medical Device & Diagnostic Industry (MD+DI)
- Interventional Cardiology Review
- Cardiology Magazine
- Others
Journals
- Journal of the American College of Cardiology
- JACC: Cardiovascular Interventions
- European Heart Journal
- Others
Newspapers
- The Wall Street Journal
- Financial Times
- Reuters Health News
- Bloomberg Healthcare
- Others
Associations
- American College of Cardiology (ACC)
- Society for Cardiovascular Angiography and Interventions (SCAI)
- European Society of Cardiology (ESC)
- Transcatheter Valve Therapy (TVT) Registry
- Others
Public Domain Sources
- Centers for Medicare & Medicaid Services (CMS)
- U.S. Food and Drug Administration (FDA)
- European Medicines Agency (EMA)
- World Health Organization (WHO)
- Others
Proprietary Elements
- CMI Data Analytics Tool
- Proprietary CMI Existing Repository of information for last 8 years
Share
Share
Missing comfort of reading report in your local language? Find your preferred language :
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
EXISTING CLIENTELE
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients