Per-Encounter Medical Market is estimated to be valued at USD 156.25 Bn in 2025 and is expected to reach USD 441.6 Bn in 2032, exhibiting a compound annual growth rate (CAGR) of 16% from 2025 to 2032.
The increasing incidence of chronic diseases and the expanding elderly population are pivotal factors fueling the demand for routine medical check-ups and procedures, thus propelling the global per-encounter medical devices market. This market is segmented into product, application, distribution channel, and region, with consumables like disposable syringes, needles, and catheters dominating the product segment due to their extensive usage.
Global Per-Encounter Medical Market Regional Insights:
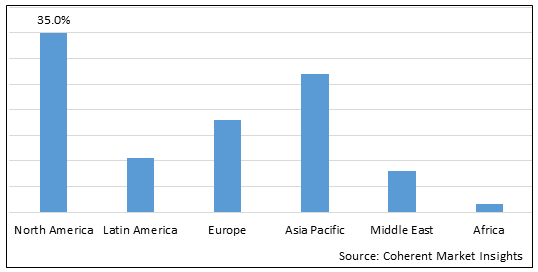
- North America is expected to be the largest market for per-encounter medical market during the forecast period, which accounted for over 35% of the market share in 2025. The market growth in North America is due to the high prevalence of chronic diseases, advanced healthcare infrastructure, and favorable reimbursement policies in the region.
- The Europe market is expected to be the second-largest market for per-encounter medical market, which accounted for over 18% of the market share in 2025. The market growth in Europe is due to the rising geriatric population, presence of major market players, and increasing adoption of minimally invasive surgeries in the region.
- The Asia Pacific market is expected to be the fastest-growing market for per-encounter medical market, with a CAGR of over 27% during the forecast period. The market growth in Asia Pacific is due to the improving healthcare infrastructure, rising disposable incomes, and expanding patient base in the region.
Figure 1. Per-Encounter Medical Market Share (%), By Region, 2025
To learn more about this report, Download Free Sample
Analyst View of the Global Per-Encounter Medical Market
Global per-encounter medical market is expected to grow steadily driven by increasing healthcare costs and focus on value-based care. As medical costs continue rising, payers and providers are exploring pay-for-performance and episode-based payment models to curb expenditure. This will drive greater adoption of per-encounter payments.
North America dominates the market due to active shift towards alternative payment models in the U.S. However, Asia Pacific is expected to offer lucrative opportunities owing to improving accessibility and universal healthcare initiatives in countries like India, China, and Malaysia. European countries are also incentivizing providers to deliver more cost-effective and coordinated care, thus benefitting the per-encounter model.
Lack of data infrastructure and healthcare fragmentation pose challenges for providers to effectively transition to this payment approach. Reimbursement uncertainty is another potential restrain as providers face financial risks if patient visits exceed budgeted rates. However, growing focus on patient-centered care, healthcare and multi-stakeholder partnerships will help overcome such restraints.
Upfront costs are capable revamping revenue cycle functions and help in deterring some providers, and the per-encounter model aligns incentives across the care continuum and rewards value over volume. It has the potential to lower unwarranted variation and healthcare costs.
Global Per-Encounter Medical Market Drivers:
- Rising prevalence of chronic diseases: The rising prevalence of chronic diseases such as cardiovascular diseases, cancer, diabetes, and musculoskeletal disorders is increasing the demand for regular medical check-ups, diagnostic testing, and surgical procedures. This is driving the adoption of single-use per-encounter medical devices like syringes, needles, Intravenous catheters, and drainage bags used during these procedures. For instance, on September 2023, according to the data published by World Health Organization (WHO), the cardiovascular diseases account for most noncommunicable diseases (NCDs) deaths, or 17.9 million people annually, followed by cancers (9.3 million), chronic respiratory diseases (4.1 million), and diabetes (2.0 million including kidney disease deaths caused by diabetes).
- Growth in geriatric population: The growing geriatric population across the globe is a key factor fueling the demand for per-encounter medical devices. Older people are more susceptible to acute and chronic diseases, thus leading to increased doctor visits and hospitalizations. This expands the patient pool undergoing diagnostics tests and treatments, thereby driving the adoption of per-encounter devices like prefilled syringes, test kits, and surgical blades. The rising elderly population is expected to boost market growth.
- Technological advancements in medical devices: Continuous advancements in medical device technologies are leading to new product developments in the per-encounter devices market. Advanced materials like engineered plastics, silicone, and metal alloys allow improved designs for single-use devices. Technologies like 3D printing facilitate customized device fabrication. The integration of electronics leads to smart devices like auto-disable and retractable syringes. Such technological advances improve performance, safety and drive new product adoption among end users.
- Favorable government regulations: Supportive regulations by governments and healthcare agencies regarding medical device standards and patient safety are conducive to the growth of single-use per-encounter devices. Mandates for safe injection practices, proper sharps disposal, and transition to high-quality devices boost market prospects.
Per-Encounter Medical Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2024 | Market Size in 2025: | USD 156.25 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2025 To 2032 |
| Forecast Period 2025 to 2032 CAGR: | 16% | 2032 Value Projection: | USD 441.6 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Catalent Inc., Smiths Medical Inc., Cardinal Health, Schott, Nipro Corporation, Terumo Corporation, Gerresheimer AG, Becton, Dickinson and Company, B. Braun Melsungen AG, AngioDynamics Inc., Teleflex Incorporated, Argon Medical Devices, Inc., ICU Medical, Inc., Medical Components, Inc., and Vygon (UK) Ltd |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Global Per-Encounter Medical Market Opportunities:
- Emerging markets expansion: Developing economies in Asia Pacific, Latin America, and Africa represent significant growth opportunities for per-encounter device manufacturers due to improving healthcare infrastructure and increasing ghealthcareexpenditure. Large underserved patient populations and increasing access to healthcare services in these regions will fuel the adoption of syringes, catheters, surgical tools and Intravenous therapy products. Market players are expanding their footprint in emerging markets through distribution partnerships and localized product offerings.
- Rising home healthcare services: The growing adoption of home healthcare services such as home dialysis, infusion therapy, and remote patient monitoring is creating opportunities for single-use medical devices tailored for home use. Devices which are designed for easy self-administration, pre-filled with medications, and integrating connectivity for telehealth allow convenient at-home treatment. Market players are developing specialized devices for home healthcare applications to tap this high-growth market segment.
- Development of eco-friendly products: Rising focus on sustainability among healthcare providers is leading to increasing demand for eco-friendly single-use medical devices made from biodegradable materials and recyclable packaging. Devices like syringes, tubing, and wound care products which are made using renewable feedstock and following end-of-life disposal guidelines allow healthcare facilities to lower their environmental impact. This presents an opportunity for manufacturers to develop innovative eco-friendly per-encounter devices.
- 3D Printing of medical devices: The adoption of 3D printing technology for on-demand fabrication of patient-specific single-use medical devices that are customized to anatomy and surgical needs supports market growth. 3D printing of per-encounter devices like surgical tools, plastic components, and dental accessories facilitates onsite-manufacturing and cost-efficiency for healthcare facilities. Device manufacturers leveraging 3D printing can provide customized offerings to gain market share.
Global Per-Encounter Medical Market Trends:
- Integration of electronics and connectivity: The integration of electronics components and connectivity technologies like Bluetooth and Near-field communication in single-use smart medical devices for data capture and communication is an emerging trend. Smart syringes with dosage tracking, safety needles with retraction sensors, and IV catheters with patency monitors allow medication delivery data to be transmitted to Electronic Health Record (EHR) systems. Device manufacturers are incorporating electronics and wireless connectivity for patient monitoring and adherence tracking to add value.
- Shift to self-administration devices: Due to the convenience and cost benefits of self-administered healthcare, demand is increasing for easy-to-use disposable devices that are designed for patient self-injection and testing. This includes devices like auto injectors, prefilled syringes, insulin pens, and wearable sensors for managing chronic conditions at home. Key device makers are developing self-administration focused products like intuitive injection systems and micro needles for transdermal drug delivery.
- Materials and design innovations: Advanced materials like engineered composites, antimicrobial coatings, and silicone are being leveraged to improve the performance and safety of per-encounter devices such as ventilator circuits, wound care products, and surgical tools. Ergonomic designs, human-centric engineering, and patient comfort considerations are driving product innovations and adoption. Companies are investing in research and development (R&D) on new materials and designs to gain a competitive edge.
- Sustainability: Medical device companies are adopting sustainable manufacturing practices by using recyclable and eco-friendly materials, by optimizing packaging, and developing take-back programs for responsible device disposal. Single-use devices that are designed with biodegradable polymers, minimal packaging, and reusability allow hospitals to meet their sustainability goals. Device makers are also offering carbon footprint data to customers and thus pursuing initiatives to lower environmental impact.
Global Per-Encounter Medical Market Restraints:
- Stringent regulatory requirements: Stringent approval and compliance requirements by regulatory agencies like the U.S. FDA for medical devices pose challenges to product development and commercialization. Lengthy approval timelines need for clinical evidence, quality management system mandates, and post-market surveillance add costs and restrict the product pipeline. This negatively impacts the introduction of newer per-encounter devices, thereby limiting market prospects.
- Pricing pressure: Pricing pressure from large group purchasing organizations, consolidated healthcare systems, and cost-containment efforts by payers is leading to declining prices for per-encounter devices like syringes, surgical kits, and IV therapy systems. To sustain profit margins, medical device companies are forced to pursue low-cost manufacturing, localized production, and materials substitution. While this impacts industry profitability, but enables lower per-unit prices temper market growth to some extent.
- Reuse of medical devices: The rising trend of reusing single-use medical devices like surgical tools, endoscopes, and catheters, after reprocessing is a major factor restricting the per-encounter devices market growth. Reprocessing allows hospitals to lower device costs. However, reusing single-use disposable items goes against regulatory mandates and raises safety concerns due to issues like incomplete sterilization. Although the practice is discouraged, cost pressures drive reuse, thereby hindering market prospects.
- Counterbalance: The practice of reusing medical devices should be encouraged by the key market players in order to drive market growth.
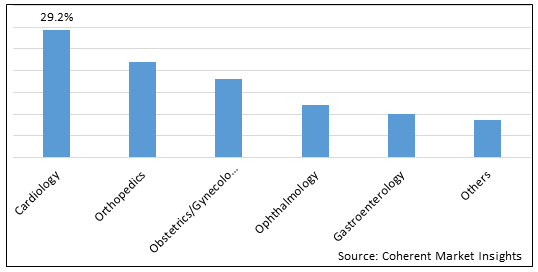
Figure 2. Global Per-Encounter Medical Market Share (%), By Applications, 2025
To learn more about this report, Download Free Sample
Recent Developments:
New product Approvals and launches:
- On February 11, 2020, Bluegrass Vascular Technologies, a medical technology company, received a De Novo classification order from the U.S. Food and Drug Administration (FDA) for its Surfacer Inside-Out Access Catheter System. The system is developed to obtain central venous access for catheter insertion into the central venous system for patients with upper body venous occlusions.
- On May 5, 2022, Access Vascular, Inc. (AVI), a manufacturing company of biomaterial-based vascular access devices, announced that it received U.S. Food and Drug Administration (FDA) 510(k) clearance for its HydroPICC Dual-Lumen catheter. Designed and constructed using the same proprietary hydrophilic biomaterial as AVI’s single-lumen HydroPICC and HydroMID catheters. These devices showed significant reduction in complications such as occlusions, replacements, deep vein thrombosis, and phlebitis in studies.
Agreement and partnerships:
- On November 19, 2021, Numedico, a global company in medical technology, signed a global distribution partnership with a medical devices distributor PBC Medicals L.L.C. Numedico. The partnership worth US$ 5.5 million allowed Numedico in distributing their ClickZip Safety Syringe technology into the global markets.
- On May 24, 2021, as part of the national response to COVID-19, the Department of Defense (DOD), in coordination with the Department of Health and Human Services (HHS), signed an agreement with Retractable Technologies, Inc. (RTI), a company manufactures safety medical devices with automated retraction has expand the production of the low dead-space (LDS) safety syringes and needles. The government allocated fund worth US$ 27.3 million.
Top Companies in Global Per-Encounter Medical Market
- Catalent Inc.
- Smiths Medical Inc.
- Cardinal Health
- Schott
- Nipro Corporation
- Terumo Corporation
- Gerresheimer AG
- Becton, Dickinson and Company
- Braun Melsungen AG
- AngioDynamics Inc.
- Teleflex Incorporated
- Argon Medical Devices, Inc.
- ICU Medical, Inc.
- Medical Components, Inc.
- Vygon (UK) Ltd
Definition: Per-encounter medical devices, which encompass items such as syringes, needles, catheters, guide wires, and surgical gowns, are single-use medical instruments which are utilized in various medical procedures and examinations.
Share
Share
About Author
Komal Dighe is a Management Consultant with over 8 years of experience in market research and consulting. She excels in managing and delivering high-quality insights and solutions in Health-tech Consulting reports. Her expertise encompasses conducting both primary and secondary research, effectively addressing client requirements, and excelling in market estimation and forecast. Her comprehensive approach ensures that clients receive thorough and accurate analyses, enabling them to make informed decisions and capitalize on market opportunities.
Missing comfort of reading report in your local language? Find your preferred language :
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
Select a License Type
EXISTING CLIENTELE
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients




