Paracetamol IV Market Size and Trends
Global paracetamol IV market is estimated to be valued at USD 867.5 Mn in 2025 and is expected to reach USD 1,104.5 Mn by 2032, exhibiting a compound annual growth rate (CAGR) of 3.5% from 2025 to 2032. Paracetamol is widely used to manage pain and fever. Rising cases of chronic diseases and surgical procedures can boost the demand for paracetamol formulations.
Discover market dynamics shaping the industry: Download Free Sample
Paracetamol IV market can witness steady growth over the forecast period, due to increasing healthcare spending and expansion of healthcare infrastructure in developing countries. Rising geriatric population suffering from chronic pain and fever can also boost the demand for paracetamol.
Discover high revenue pocket segments and roadmap to it: Download Free Sample
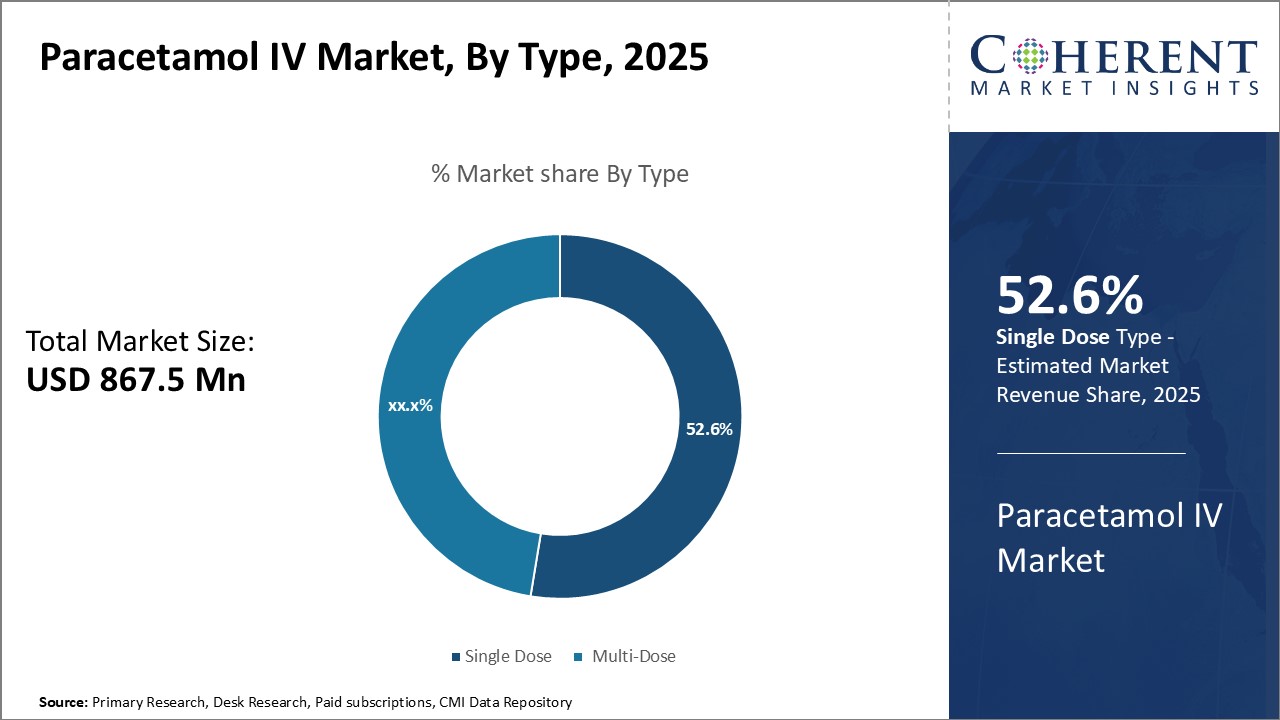
Insights By Type- Convenience of Consumption Drives Single Dose’s Dominance
By type, single dose segment dominates the paracetamol IV industry, with an estimated market share of 52.6% in 2025, due to their convenience. Patients benefit from simplified treatment, avoiding multiple dosing schedules. Medical staff also prefer single-dose vials, reducing the risk of incorrect dosing and wastage. Single-dose vials ensure efficient medication use, as the entire vial is consumed in one administration.
Insights By Application- Wide Applicability Fuels Pain Management’s Dominance
By application, pain management segment dominates the paracetamol IV industry, with an estimated market share of 53.7% in 2025, due to its broad use for acute and post-surgical pain relief. It effectively alleviates pain from cluster headaches, backaches, and surgeries, with a favorable safety profile for vulnerable populations. Paracetamol IV is also commonly used alongside opioids to reduce narcotic doses, minimizing side effects and risks. Its mild analgesic properties, flexibility, and tolerability make it the first-line treatment for mild-to-moderate pain, enhancing its dominance in acute pain management.
Insights By End User- Role in Streamlined Care Drives Hospitals’ Dominance
By end user, hospitals segment dominates the paracetamol IV industry, with an estimated market share of 40.6% in 2025, due to their need for efficient pain management in complex, high-volume settings. Paracetamol IV’s rapid onset of action and precise dosing meet the demands of hospitals managing acute pain from surgeries and trauma. Its intravenous form offers 100% bioavailability and quick relief, ensuring effective pain control. This, combined with fewer administration steps compared to oral medications, makes paracetamol IV indispensable for standardized pain management in hospitals, supporting streamlined inpatient care workflows.
Regional Insights
Need a Different Region or Segment? Download Free Sample
Regional Insights
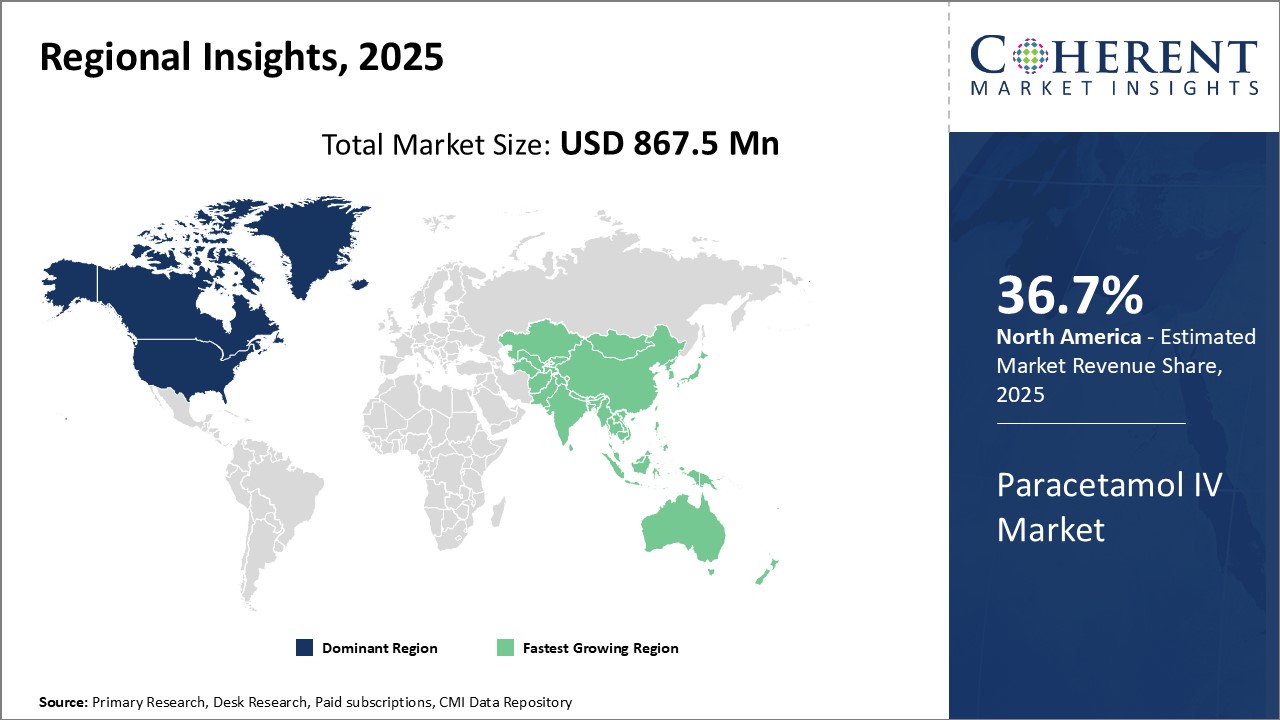
Dominating Region- North America
North America dominates the paracetamol IV industry with share of 36.7% in 2025 due to robust healthcare infrastructure, advanced medical treatments, and the presence of major players like Pfizer Inc. innovating in intravenous pain management therapies. The region's established healthcare system supports widespread adoption of effect IV pain relief solutions.
Fastest-Growing Region- Asia Pacific
Asia Pacific is the fastest-growing region due to rising healthcare expenditure, aging population prone to chronic diseases, and increasing emphasis on pain management in developing nations. This growth is bolstered by expanding healthcare access and evolving pain relief strategies.
Paracetamol IV Market Outlook for Key Countries
U.S. Leads the Demand for Pain Management
The U.S. leads the paracetamol IV industry due to its advanced healthcare infrastructure and high surgical rates. Growing preference for non-opioid pain management solutions in hospitals can also boosts the demand. On January 30, 2025, according to PubMed Central, 77.5% to 83.5% of patients in the U.S. reported experiencing moderate to severe postoperative pain, highlighting the demand for effective pain relief options. Intravenous paracetamol's favorable safety profile makes it suitable for multimodal pain management strategies.
Growing Focus on Postoperative Care in Canada
Canada is increasingly adopting paracetamol IV in postoperative care settings, with a notable 25.5% rise in its usage for pain management following surgeries, as per the data from Canadian Journal of Anesthesia published in September 2023. This trend reflects a shift toward multimodal analgesia strategies that prioritize non-opioid pain relief. Paracetamol IV’s rapid onset and favorable safety profile makes it an essential part of Canada’s pain management approach, enhancing patient recovery and minimizing reliance on opioids.
Innovative Pain Management Practices in Japan
Japan's healthcare system is increasingly integrating paracetamol IV for effective pain relief. In August 2023, the Japanese Society of Anesthesiologists reported an increase in usage of IV paracetamol in surgical procedures, emphasizing its role in reducing opioid dependence and improving recovery outcomes.
Rapid Expansion in Healthcare Access in China
China paracetamol IV market is growing rapidly due to government initiatives like the Healthy China 2030 to improve healthcare access. As per the report from the China Health Commission released in 2023, there has been 30.5% increase in the availability of IV paracetamol in hospitals due to rising surgical procedures and demand for effect IV e-pain management.
Emerging Market for Non-opioid Solutions in India
India is witnessing huge adoption of paracetamol IV, particularly in urban healthcare settings. In November 2023, the Indian Journal of Pain reported a 40.5% increase in the usage of IV paracetamol for managing postoperative pain, reflecting a growing awareness of non-opioid alternatives.
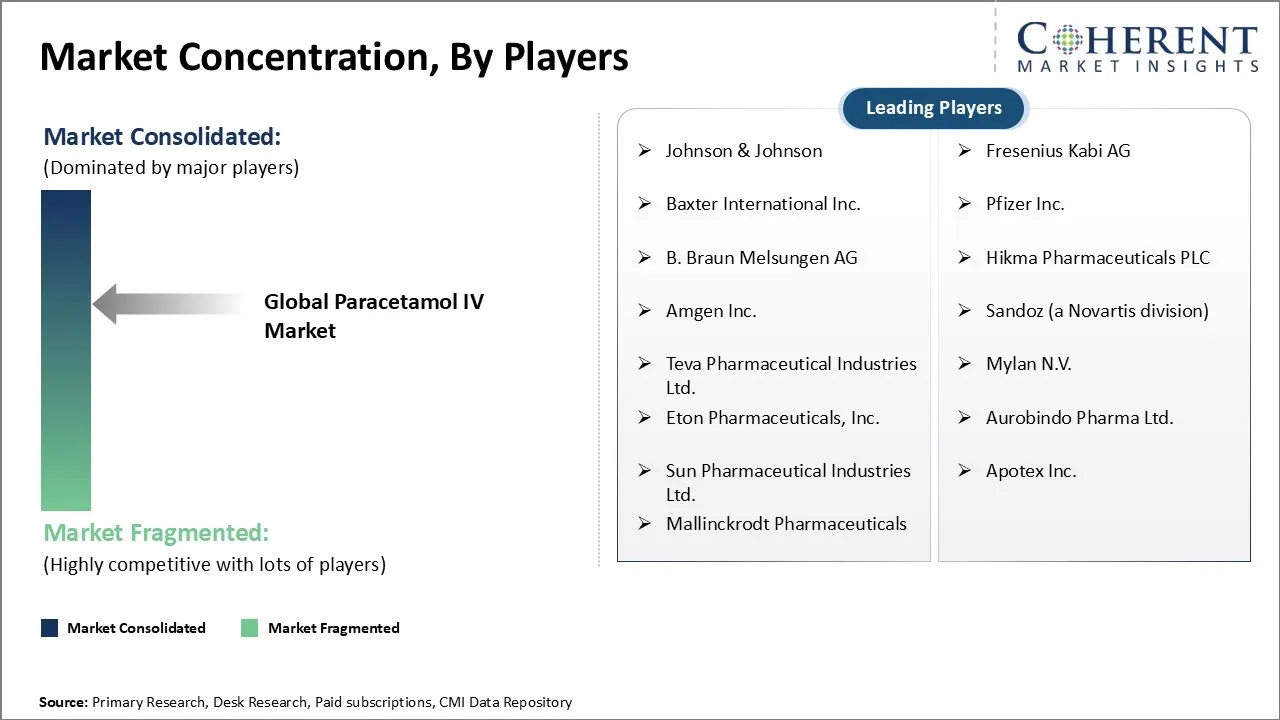
Market Concentration and Competitive Landscape
Get actionable strategies to beat competition: Download Free Sample
Top Strategies Followed by Global Paracetamol IV Market Players
- Established Players: R&D Investment - Leading companies like Johnson & Johnson in the global paracetamol IV market focus extensively on research and development to innovate new products. For example, Pfizer, a top player in pharmaceutical sector, dedicated over US$ 8 billion to R&D in 2020 to launch improved injectable analgesics in new markets worldwide.
- Mid-Level Players: Cost-Effective Solutions - Mid-sized companies in the market focus on delivering quality products at affordable price points. These introduce generic and value-for-money paracetamol IV solutions to tap the needs of price-conscious consumers. For example, Siemens Healthcare partnered with UNICEF, United Nations International Children's Emergency Fund, to optimize point-of-care diagnostics networks in sub-Saharan Africa.
- Small-Scale Players: Niche Specialization - Emerging small players look to capture untapped segments by offering specialized products. In October 2024, AdvaCare Pharma developed a paracetamol solution for pediatric pain management in pre-filled syringes. This addresses the need for easy-to-administer formulations for young patients.
Emerging Startups in the Global Paracetamol IV Market
Startups are bringing innovation and sustainability to the paracetamol IV market with advanced technologies and eco-friendly solutions. NanoMed, a biotech startup, announced on September 2024 the development of a nanoparticle-enhanced IV paracetamol formulation
BioSolve Inc. focuses on reducing plastic waste by producing biodegradable paracetamol IV bags from sugarcane-derived materials. GreenCare Solutions emphasizes sustainability by using renewable energy and recycling over 95% of its production waste. GreenMedic Inc. offers fully recyclable IV solutions made from recycled plant fibers. These startups are not only driving environmental sustainability but also creating new revenue streams by reprocessing medical waste, with GreenMedic recognized in Forbes' "Top 10 Healthcare Startups to Watch" in 2020.
Key Takeaways from Analyst
- Global paracetamol IV market can witness growth over the forecast period, owing to rising prevalence of fever and pain disorders worldwide. Growing geriatric population who are more susceptible to chronic diseases can also boost demand for paracetamol injectables. Furthermore, rapid development of infusion and injection techniques encourages healthcare professionals to prefer IV mode of paracetamol administration over oral dosage forms. North America currently dominates the market but Asia Pacific is expected to emerge as the fastest growing region due to rising healthcare expenditure and increasing awareness.
- Availability of alternative drug entities for fever and pain reduction can hamper the market growth. Moreover, stringent regulations pertaining to approval of IV drugs may act as a restraining factor. Increasing demand for fast-acting analgesics in hospital settings can counteract this effect. Manufacturers should focus on Asia Pacific region by strengthening their distribution networks. Contract manufacturing and collaboration with local players can help achieve higher penetration in emerging countries. Providing customizable solutions tailored for diverse geographies would also create opportunities for sustained long-term growth.
Market Report Scope
Paracetamol IV Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2024 | Market Size in 2025: | USD 867.5 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2025 To 2032 |
| Forecast Period 2025 to 2032 CAGR: | 3.5% | 2032 Value Projection: | USD 1,104.5 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Johnson & Johnson, Fresenius Kabi AG, Baxter International Inc., Pfizer Inc., B. Braun Melsungen AG, Hikma Pharmaceuticals PLC, Amgen Inc., Sandoz (a Novartis d IV ision), Teva Pharmaceutical Industries Ltd., Mylan N.V., Eton Pharmaceuticals, Inc., Aurobindo Pharma Ltd., Sun Pharmaceutical Industries Ltd., Apotex Inc., and Mallinckrodt Pharmaceuticals |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Market Dynamics
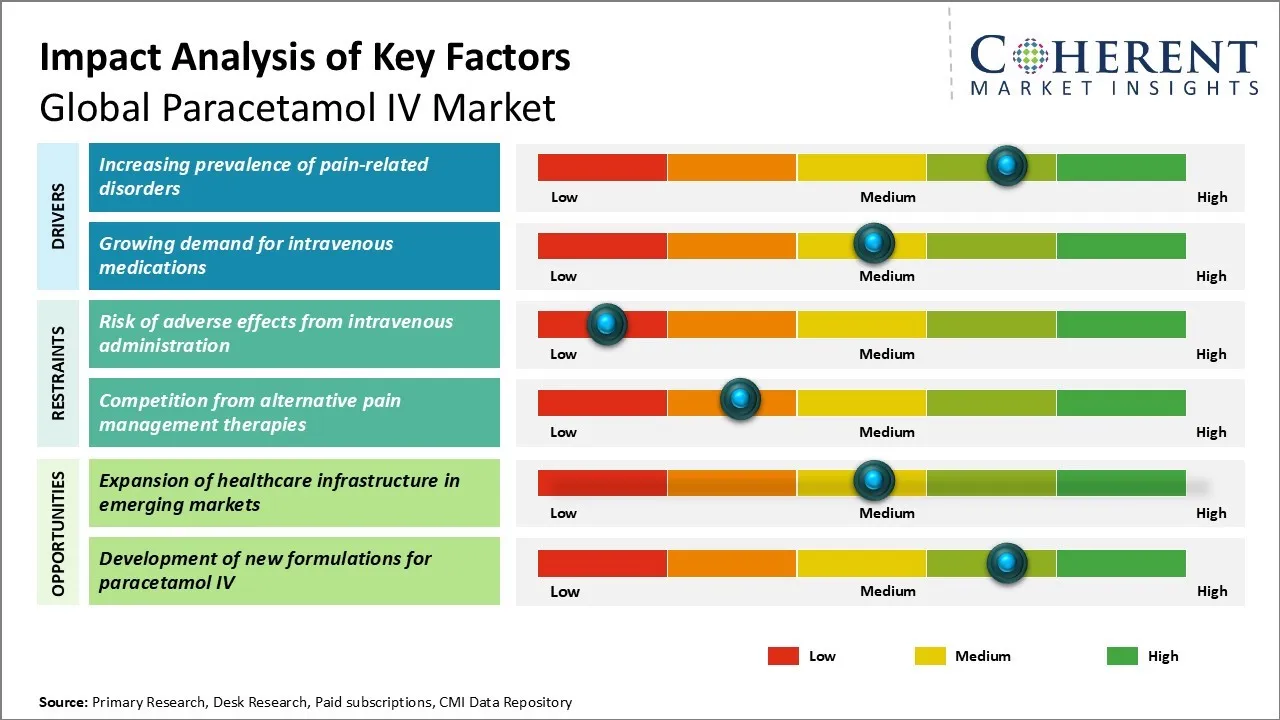
Market Driver - Increasing prevalence of pain-related disorders
Pain-related disorders like osteoarthritis, lower back pain, and migraines are increasingly prevalent worldwide, with rising incidences due to lifestyle factors. Intravenous paracetamol is gaining popularity as it provides faster pain relief within 10-15 minutes as compared to oral formulations. For instance, in May 2022, a study published in Frontiers in Pharmacology evaluated the effectiveness of intravenous (IV) paracetamol in managing postoperative pain and reducing opioid use following lumbar disc surgery. The meta-analysis revealed that IV paracetamol significantly decreased pain scores at multiple intervals.
Market Challenge - Risk of adverse effects from intravenous administration
Major challenge faced by global paracetamol IV market includes risk of adverse effects, such as anaphylaxis and hepatotoxicity, associated with intravenous administration. Despite being a widely used over-the-counter drug, these risks have led healthcare professionals and regulatory bodies to approach paracetamol IV cautiously. Ongoing clinical research is necessary to establish standardized dosage guidelines and evaluate long-term safety. Manufacturers must invest in educating healthcare providers about the benefits of paracetamol IV and conduct post-marketing surveillance to build confidence in its use.
Market Opportunity - Expansion of healthcare infrastructure in emerging markets
Expansion of healthcare infrastructure in emerging markets like China, India, and Brazil presents significant growth opportunities for global paracetamol IV market. Increasing investments in hospitals, medical facilities, and health insurance coverage are improving access to intravenous therapies. Rising living standards and disposable incomes allow consumers to afford advanced treatments. Paracetamol IV is an attractive option for postoperative care, addressing issues like nausea from oral administration. Leading manufacturers are likely to enter these markets through partnerships and affordable pricing, driving demand in the future.
Market Segmentation
- Type Insights (Revenue, USD Mn, 2020 - 2032)
-
- Single Dose
- Multi-Dose
- Application Insights (Revenue, USD Mn, 2020 - 2032)
-
- Pain Management
- Fever Reduction
- End User Insights (Revenue, USD Mn, 2020 - 2032)
-
- Hospitals
- Clinics
- Homecare Settings
- Others
- Regional Insights (Revenue, USD Mn, 2020 - 2032)
-
- North America
- U.S.
- Canada
- Latin America
- Brazil
- Argentina
- Mexico
- Rest of Latin America
- Europe
- Germany
- U.K.
- Spain
- France
- Italy
- Russia
- Rest of Europe
- Asia Pacific
- China
- India
- Japan
- Australia
- South Korea
- ASEAN
- Rest of Asia Pacific
- Middle East
- GCC Countries
- Israel
- Rest of Middle East
- Africa
- South Africa
- North Africa
- Central Africa
- North America
- Key Players Insights
- Johnson & Johnson
- Fresenius Kabi AG
- Baxter International Inc.
- Pfizer Inc.
- B.Braun Melsungen AG
- Hikma Pharmaceuticals PLC
- Amgen Inc.
- Sandoz (a Novartis division)
- Teva Pharmaceutical Industries Ltd.
- Mylan N.V.
- Eton Pharmaceuticals, Inc.
- Aurobindo Pharma Ltd.
- Sun Pharmaceutical Industries Ltd.
- Apotex Inc.
- Mallinckrodt Pharmaceuticals
Share
Share
About Author
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Missing comfort of reading report in your local language? Find your preferred language :
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
EXISTING CLIENTELE
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients
