Immune Cell Engineering Market is estimated to be valued at USD 4.23 Bn in 2025 and is expected to reach USD 17.51 Bn in 2032, exhibiting a compound annual growth rate (CAGR) of 22.5% from 2025 to 2032.
Immune cell engineering involves modifying immune cells such as T cells, NK cells, and dendritic cells for research and therapeutic applications. Key applications include the development of immunotherapies like CAR T-cell therapy, studying immune responses, and drug discovery. The growth of this market is driven by rising investments in cell-based research and advancements in gene-editing technologies.
The immune cell engineering market is segmented by cell type, product, disease indication, application, end user, and region. By cell type, the T cell engineering segment accounted for the largest share in 2025. T cells play a central role in cell-mediated immunity and are increasingly engineered for adoptive cell transfer immunotherapies like CAR T-cell therapy to treat cancer. The rising number of CAR T-cell therapy clinical trials and approvals is driving the growth of engineered T cells.
Global Immune Cell Engineering Market Regional Insights:
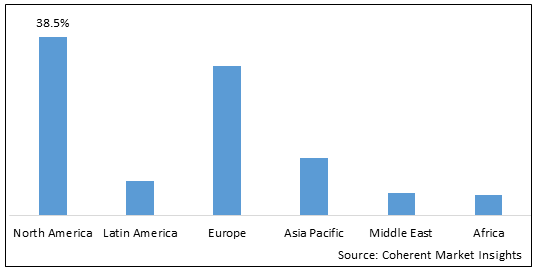
- North America is expected to be the largest market for global immune cell engineering market during the forecast period, accounting for over 38.5% of the market share in 2025. The growth of the market in North America is attributed to the high adoption of immunotherapies, presence of leading biopharma companies, and increasing cancer research.
- The Europe market is expected to be the second-largest market for immune cell engineering market, accounting for over 32.1% of the market share in 2025. The growth of the market in is attributed to the rising investments by government bodies to support cell-based research.
- The Asia Pacific market is expected to be the fastest-growing market for immune cell engineering market, with a CAGR of over 22.6% during the forecast period. The growth of the market in Asia Pacific is attributed to an increasing focus on developing cell therapies and government funding to support biotechnology sector.
Analyst View: The global immune cell engineering market presents lucrative opportunities for companies involved as demand for advanced cell therapies continues to grow. Adoptive cell transfer therapies that utilize engineered T cells or chimeric antigen receptor (CAR) T cells have shown promising clinical trial results in treating cancers. Major drivers of the market include increasing investment in immunotherapies by big pharmaceutical companies and expanding clinical trials for immune cell therapies. However, the development of these personalized therapies is highly complex and custom manufacturing still remains a challenge. Process development and regulatory compliance add to the overall costs. Given the complexity, scaling up production capacity is a major restraint to realizing the market potential. High therapy costs also limit broader patient access currently. North America dominates the global immune cell engineering market currently due to the presence of global leaders and major ongoing clinical research. However, Asia Pacific is expected to see the fastest growth over the coming years. The continuous expansion of healthcare infrastructure as well as rising research funding in China, Japan, and India will boost the regional market. Collaborations between global drug makers and local Asian pharma companies will help drive technology transfer and capacity growth. Overall, while commercialization challenges remain, the immune cell engineering field holds promise for effective treatment of cancer as well as other diseases. Growing clinical evidence and partnerships across the ecosystem are likely to support market evolution over the long term if production and cost barriers are systematically addressed.
Figure 1. Global Immune Cell Engineering Market Share (%), by Region, 2025
To learn more about this report, Download Free Sample
Global Immune Cell Engineering Market Drivers:
- Rising Incidence of Chronic Diseases: The rising prevalence of chronic diseases like cancer, autoimmune disorders, and cardiovascular diseases around the world is a key factor driving the growth of the immune cell engineering market. Diseases like cancer severely affect the immune system, and cell-based immunotherapies are being increasingly adopted as an effective therapeutic strategy. For instance, engineered T cell therapies like CAR T-cell therapy have shown promising results in certain hematological cancers. The success of such advanced immunotherapies is driving investments in immune cell engineering research to develop treatments for various chronic diseases.
- Technological Advancements in Cell Engineering: Continuous advances in gene editing, cell isolation, and expansion technologies are significantly supporting the growth of the immune cell engineering industry. The development of efficient gene editing tools like CRISPR/Cas9 has enabled precise and safer modifications in immune cells. Other technologies, like microfluidics and high-throughput assays, are enhancing cell engineering workflows. Growing understanding of immune cell signaling pathways and interactions is also enabling the engineering of enhanced immune cell therapies. These ongoing technological innovations are expected to expand the scope and applications of engineered immune cells.
- Growth in Cell Therapy Clinical Trials: There has been a remarkable surge in the number of clinical trials evaluating novel engineered immune cell therapies over the last decade. Several companies and academic institutions are increasingly investigating the safety and efficacy of various modified immune cell therapies. For instance, the number of CAR T-cell therapy trials rose from just 1 in 2009 to over 300 in 2018. The encouraging response rates from some of these trials are driving increased activity in this space. The exponential growth of global cell therapy clinical trials signifies the rising adoption of immune cell engineering.
- Increasing Investments in Cell-based Research: Public and private entities are steadily ramping up their investments in cell engineering research and infrastructure development to support advancements in the field. Government bodies like the NIH and charities like the Cancer Research Institute have expanded funding for academic research on engineered immune cells and immunotherapy. Leading biopharma players have also enhanced capital allocation for internal immunotherapy programs and related strategic acquisitions. These rising investments in research and development (R&D) and infrastructure to aid continued innovation in engineered immune cells are fueling market growth.
Global Immune Cell Engineering Market Opportunities:
- Expanding Therapeutic Applications: While cancer immunotherapy has been the primary application, engineered immune cells are likely to find increased usage for treating various other diseases such as cardiovascular diseases in the coming years. Ongoing research is evaluating their potential application in neurodegenerative, autoimmune, cardiovascular, and infectious diseases. Cell therapies are also being explored to induce tolerance in patients undergoing transplants or with autoimmune disorders. The capability to modify cell specificity and enhance targeted immune function will enable tailored immune cell therapies for diverse unmet needs.
- Developments in Stem Cell Engineering: Advances in induced pluripotent stem cell technology and direct reprogramming of somatic cells are expanding the horizons of immune cell engineering. Instead of relying on donor blood, stem cells can act as a renewable source for producing large customizable T cell and NK cell therapies. Research on modulating differentiation and function of IPCs and HSCs to obtain enhanced engineered immune cells is anticipated to expand therapeutic access. Efforts to scale stem cell manufacturing will support developments in this area.
- Synergistic Combination Therapies: Engineered immune cells are being evaluated in combination with complementary approaches like immunomodulator, targeted small molecules, and antibody therapies. Combined multimodal therapies that can target mechanisms of cancer resistance have significant potential. Research is also ongoing on dual-engineered cells that can integrate multiple therapeutic properties. The development of synergistic combination approaches with engineered T cells is an emerging opportunity to improve patient outcomes.
- Growing Adoption of Automation: The integration of laboratory automation and closed cell processing systems in immune cell engineering workflows can make therapies safer, more efficient, and more consistent. Automated modular GMP-compliant manufacturing platforms will be crucial for delivering individualized cell therapies. Companies are increasingly adopting tools like automated cell sorters, bioreactors, viral vector production suites, and AI-enabled processes for engineered immune cell applications. The push towards automation and standardized solutions will propel market growth.
Global Immune Cell Engineering Market Trends:
- Shift Towards Point-of-Care Manufacturing: Decentralized point-of-care manufacturing models for engineered cell therapies are evolving to make treatments faster and more personalized. Onsite hospital biofactories with automated manufacturing capability can customize cell therapies using a patient’s cells and deliver it rapidly. Several companies are developing turnkey point-of-care cell engineering platforms that require minimal expertise. Enabling wider accessibility through decentralized manufacturing close to the patient is an important trend.
- Rising Industry-Academia Collaborations: Robust partnerships between industry and academic institutes are on the rise to combine capabilities and fuel innovation in immune cell engineering. Pharmaceutical players are collaborating with universities with proprietary technologies or research expertise to develop next-gen immunotherapies. Such open innovation networks allow leveraging of synergistic capabilities. Increasing licensing deals and strategic partnerships among biopharma firms, technology providers, and academic groups will remain a key trend.
- Growing Focus on Off-The-Shelf Therapies: There is a rising focus on developing universal off-the-shelf engineered T cell products that can circumvent the costs and delays of personalization. Companies are working on proprietary techniques to generate allogeneic therapies using healthy donor cells that will not face rejection issues. Advances in cell banking, genetic modifications, and use of memory T cell subsets are aiding such efforts. The progress towards off-the-shelf engineered cell therapies is expected to expand treatment access.
- Emergence of Synthetic Biology Approaches: The field of engineered immune cells is increasingly incorporating principles of synthetic biology to create smarter, multifunctional therapeutic cells. Gene circuits, logic gating, and modular receptor scaffolds are being leveraged to design complex cell behaviors and sensing capabilities. Companies like Poseida Therapeutics and T-CURX are building extensive capabilities in synthetic biology and next-gen cell reprogramming. The application of such innovative platforms will enable more advanced and nuanced immune cell therapies.
Global Immune Cell Engineering Market Restraints:
- High Costs of Cell and Gene Therapies: Engineered cell therapies are presently very expensive, with certain approved autologous CAR T-cell therapies priced over USD 300,000. The costs are mainly driven by lengthy personalized manufacturing, viral vectors, complex production processes, limited scale, and extended hospitalization stays. This poses a significant barrier to widespread adoption. While prices are expected to decrease with growth in scale and competition, high costs will remain a key market challenge.
- Complex Manufacturing Processes: The production of engineered immune cell immunotherapies involves complex, multi-step processes and stringent protocols. Critical aspects include the isolation of desired cell subsets, genetic modification, and expansion of cells to obtain sufficient numbers, stringent quality checks, and cold chain logistics. Automated solutions for simplifying and standardizing such elaborate engineering and manufacturing workflows remain limited currently. The complex processes constrain rapid, large-scale production.
- Side Effects of Immune Cell Therapies: Engineered cell immunotherapies, especially those involving genetic manipulation, have been associated with potentially fatal side effects like cytokine release syndrome and neurotoxicity. Their long-term effects remain unknown. There are also risks linked to insertional oncogenesis from viral vectors. Concerns regarding unpredictable adverse effects can restrict their adoption among patients and hinder regulatory approvals. More research is vital to enhance the precision and safety of engineered immune cells.
Immune Cell Engineering Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2024 | Market Size in 2025: | USD 4.23 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2025 To 2032 |
| Forecast Period 2025 to 2032 CAGR: | 22.5% | 2032 Value Projection: | USD 17.51 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Thermo Fisher Scientific Inc., Merck KGaA, Danaher Corporation, Lonza, Miltenyi Biotec, Takara Bio Inc., Sartoroius CellGenix GmbH, FUJIFILM Irvine Scientific, Agilent Technologies, Inc., Bio-Techne, Bio-Rad Laboratories, Inc., Becton, Dickinson and Company, Charles River Laboratories, Creative Bioarray, Sartorius AG, Novogene Co, Ltd., AGC Biologics, Catalent, Inc., WuXi AppTec, Discovery Life Sciences |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Recent Developments:
New product launches:
- On August 10, 2023, Astellas Pharma, Inc., a pharmaceutical company and Poseida Therapeutics, a clinical stage company entered into a strategic investment agreement to further support the development of cell therapies for cancer. Astellas will be investing a total of US$ 50 million and a one-time US$ 25 million payment for a right of exclusive negotiation and first refusal to license one of Poseida’s clinical-stage programs, P-MUC1C-ALLO1, which is an allogeneic chimeric antigen T cell (CAR-T) cell therapy in development for multiple solid tumor indications.
- On July 19, 2023, Integrated Biosciences, a biotechnology company, announced a drug discovery platform that enables precise control of the integrated stress response (ISR), a biological pathway that is activated by cells in response to a wide variety of pathological and aging-associated conditions, to use synthetic biology, and in particular ontogenetic, to control age-related cellular signaling pathways. This technology allows for novel drug discovery efforts, allowing us to query specific aspects of cellular biology that produce faster, on-target drug screens with built-in mechanism of action validation.
- In December 2022, Ferring B.V., a biopharmaceutical company, announced that the U.S. Food and Drug Administration had approved Adstiladrin (nadofaragene firadenovec-vncg), a non-replicating (cannot multiply in human cells) adenoviral vector based gene therapy indicated for the treatment of adult patients with high-risk Bacillus Calmette-Guérin (BCG)-unresponsive non-muscle-invasive bladder cancer (NMIBC) with carcinoma in situ (CIS) with or without papillary tumors.
- In April 2022, Thermo Fisher Scientific, Inc., a biotechnology company, launched the new Gibco CTS DynaCellect Electroporation System for more consistent and efficient delivery of molecules into hard-to-transfect cells. This improved cell engineering workflow efficiency in cell line development and cell therapy manufacturing.
- In December 2021, Novartis, a pharmaceutical company, announced the launch of T-Charge, its next-generation CAR-T platform, which can be used as the foundation for various new investigational CAR-T cell therapies in the company’s pipeline. The T-Charge platform helps in developing CAR-T cell therapy products with greater proliferative potential and fewer exhausted T cells.
- In March 2021, Beckman Coulter, a biotechnology company, launched the CytoFLEX SRT cell sorter to automate and standardize sorting during cell engineering processes for reproducibility.
Acquisition and partnerships:
- On September 18, 2023, Lupagen Inc., a pioneer in cell and gene therapy delivery, announced a new development and supply agreement partnership with Fresenius Kabi, a global leader in injectable medicines, infusion technology, and clinical nutrition, to advance bedside cell and gene therapy. Under the partnership, Fresenius Kabi will provide expertise from its transfusion medicine unit to optimize Lupagen’s platform for delivering cell and gene therapy treatments at the bedside of patients suffering from many conditions including cancer and others.
- On April 11, 2023, Discovery Life Sciences, a manufacturer of biomarkers, acquired ReachBio Research Labs (ReachBio), a company specializing in cell-based assays, predictive hematotoxicity, and drug screening services. This acquisition will allow Discovery to scale its consolidated drug discovery and preclinical development offerings to give customers access to a broader range of innovative and specialized cell biology services.
- In December 2022, Gilead Sciences, Inc., a pharmaceutical company, announced a collaboration with Arcellx, Inc., a biotechnology company, to co-develop and co-commercialize Arcellx, Inc.’s lead late-stage product candidate named CART-ddBCMA for the treatment of patients with relapsed or refractory multiple myeloma
- In February 2021, Charles River Laboratories acquired Cognate Bio Services to expand its cell and gene therapy capabilities and fully integrate CDMO offerings.
- In November 2020, Catalent Inc., a biotechnology company, acquired Bone Therapeutics' subsidiary Skeletal Cell Therapy Support to expand its capabilities in cell therapies and strengthen its expertise in GMP manufacturing of MSCs.
- In March 2020, FUJIFILM Corporation acquired Biogen's manufacturing facility in Denmark to expand its capacity to produce gene therapies and cell therapies.
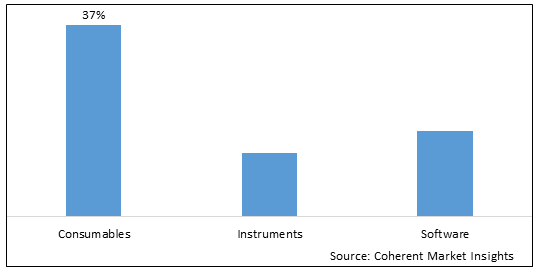
Figure 2. Global Immune Cell Engineering Market Share (%), by Product, 2025
To learn more about this report, Download Free Sample
Top companies in Immune Cell Engineering Market:
- Thermo Fisher Scientific Inc.
- Merck KGaA
- Danaher Corporation
- Lonza
- Miltenyi Biotec
- Takara Bio Inc.
- Sartoroius CellGenix GmbH
- FUJIFILM Irvine Scientific
- Agilent Technologies, Inc.
- Bio-Techne
- Bio-Rad Laboratories, Inc.
- Becton, Dickinson and Company
- Charles River Laboratories
- Creative Bioarray
- Sartorius AG
- Novogene Co, Ltd.
- AGC Biologics
- Catalent, Inc
- WuXi AppTec
- Discovery Life Sciences
*Definition: Immune cell engineering is the process of modifying and manipulating immune cells such as T cells, B cells, NK cells, and dendritic cells for research, therapeutic, and bio production applications. It involves techniques like gene editing, vector transduction, and electroporation to introduce foreign genes, alter receptors, or enhance function. Engineered immune cells are used for cancer immunotherapy, studying immune responses, drug discovery, and cell therapy manufacturing. The immune cell engineering field is driven by emerging gene editing tools like CRISPR and a growing understanding of immunotherapy.
Few Other Promising Reports in Pharmaceutical Industry
Drug Discovery Outsourcing Market
Share
Share
About Author
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Missing comfort of reading report in your local language? Find your preferred language :
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
Select a License Type
EXISTING CLIENTELE
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients




