Global chaple disease therapeutics market size is expected to reach US$ 160.2 Mn by 2032 from US$ 145.3 Mn in 2025, exhibiting a compound annual growth rate (CAGR) of 1.4% during the forecast period.
Research and development (R&D) activities by the biotechnological and pharmaceutical companies providing the facilities for treatment and increase in demand for therapeutics for the treatment of the disorder are expected to boost the market growth.
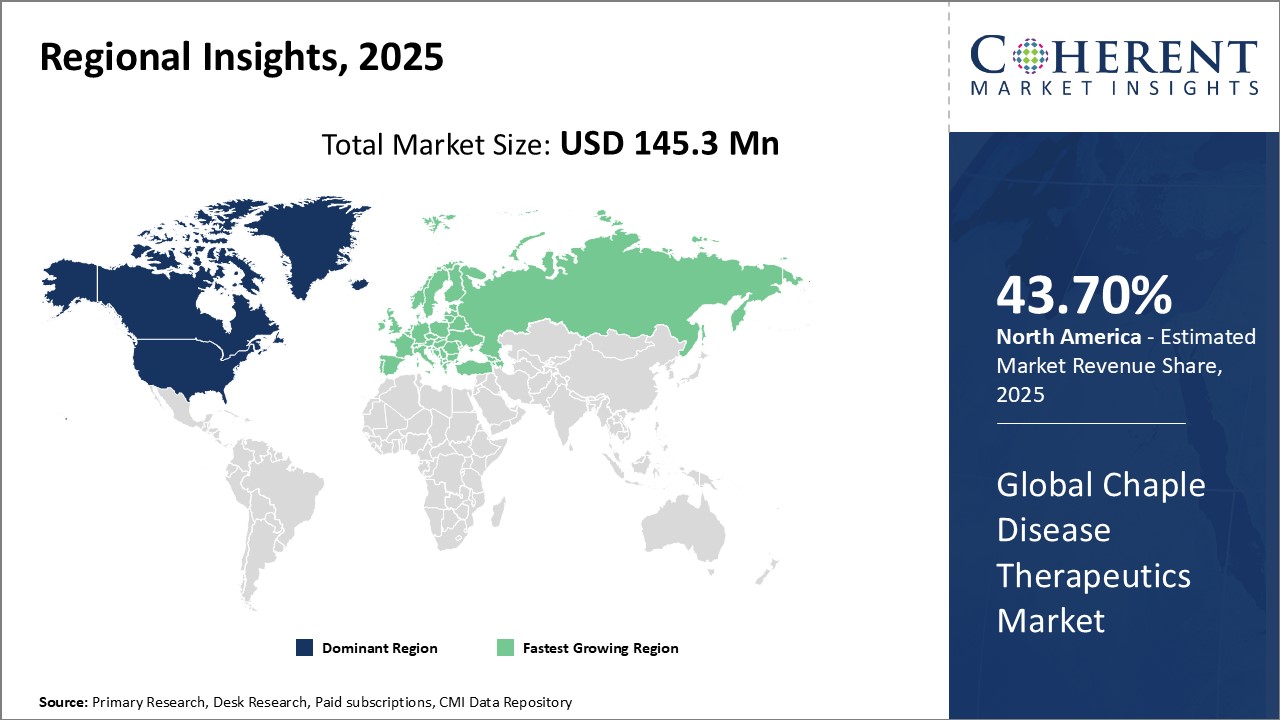
Global Chaple Disease Therapeutics Market- Regional Insights
- North America is expected to be the largest market for chaple disease therapeutics market during the forecast period, accounting for over 43.7% of the market share in 2025. The North American region dominates the global chaple disease therapeutics market with the presence of several top pharmaceutical companies and a large healthcare expenditure.
- Asia Pacific market is expected to be the second-largest market for chaple disease therapeutics market, accounting for over 25.2% of the market share in 2025. The Asia Pacific region, especially emerging countries like China and India, is witnessing the fastest growth in the Chaple disease therapeutics market. This is due to increasing research and development (R&D) activities, partnerships, and agreements among key market players.
- Europe market is expected to be the fastest-growing market for chaple disease therapeutics market, with a share of 19% during the forecast period. The growth of the market in Europe is emerging as a hotspot at a rapid pace due to favorable policy support, infrastructure developments, and evolving customer preferences.
Figure 1. Global Chaple Disease Therapeutics Market Share (%), By Region, 2025
To learn more about this report, Download Free Sample
Analyst View:
Global chaple disease therapeutics market is projected to experience significant growth in the near future, due to increasing research and development activities. The market lacks effective treatment options and existing therapeutics only provide symptomatic relief. This presents significant opportunities for pharmaceutical companies to develop novel therapeutic targets and disease modifying drugs. Asia Pacific is anticipated to be the fastest growing region due to the large patient pool and improving healthcare infrastructure in developing countries such as India and China.
Global Chaple Disease Therapeutics Market - Drivers
- Increasing partnerships among key market players: Increasing adoption of inorganic growth strategies such as partnerships is expected to drive the market growth over the forecast period. For instance, on August 23, 2025, Regeneron Pharmaceuticals Inc., a global pharmaceutical company, announced a partnership with Orsini Specialty Pharmacy (Orsini), an independent specialty pharmacy focused on rare diseases and gene therapies. Under the terms of partnership, Orsini Specialty Pharmacy (Orsini) will be the exclusive specialty pharmacy partner for VEOPOZ™ (pozelimab-bbfg). A monoclonal antibody, VEOPOZ is the first and only treatment for those living with chaple disease, an ultra-rare and life-threatening hereditary disease caused by an over activation of the complement system. VEOPOZ is approved for the treatment of adult and pediatric patients 1 year of age and older with CD55-deficient protein-losing enteropathy (PLE), also known as CHAPLE disease.
- Improving healthcare infrastructure: Improving healthcare infrastructure across the world is set to be a major driver of growth in the global chaple disease therapeutics market over the coming years. Chaple disease is a genetic disorder that impairs the body's ability to break down fats and other lipids, and can lead to serious health issues if left untreated. As more countries focus on expanding access to advanced healthcare and building out their medical systems, more people around the world will be able to receive effective treatment and management of chaple disease. Better access to diagnosis and long-term care will be important for driving long-term patient adherence and compliance to therapies as well. This helps minimize complications and produce the best health outcomes. As public health systems mature in more locations around the globe, they will strengthen the foundations for sustainable growth of the chaple disease therapeutics industry in the near future.
Global Chaple Disease Therapeutics Market- Opportunities
- Emerging markets in Asia Pacific and Latin America: Emerging markets in Asia Pacific and Latin America offer immense potential for growth in global chaple disease therapeutics. Chaple disease is prevalent in developing nations and according to World Health Organization (WHO), countries like India, Brazil, China, and Mexico account for a major share of the global disease burden. These emerging economies are witnessing rapid economic and infrastructural development which is improving access to healthcare. Simultaneously, growing health awareness and expanded insurance coverage are enabling more patients to seek treatment. Pharmaceutical companies can capitalize on these favorable dynamics. Governments in the region have indicated their commitment to tackling non-communicable diseases and several have launched public health programs focused on early detection and management of conditions like chaple disease. For instance, in 2020, the National Medical Mission under India's Ayushman Bharat initiative aims to set up 1.5 million health and wellness centers by 2023, which will boost screening efforts National Institution for Transforming India (NITI Aayog)
- Rising healthcare expenditure: The significant factor influencing the growth rate of the global chaple disease therapeutics market is the growing healthcare expenditure, which helps in improving its infrastructure. For instance, according to the International Health Care System of the U.S., in June 2020, U.S. government organizations aim to improve the healthcare infrastructure by increasing funding, setting legislation, and national strategies, and cofounding and setting basic requirements and regulations for the Medicaid program. Similarly, in November 2022, according to the Canadian Institute for Health Information, the total healthcare expenditure in Canada was US$ 331 Bn in 2022, or US$ 8,563 per Canadian, while health expenditure represented 12.2% of Canada's gross domestic product (GDP) in 2022, following a high of 13.8% in 2020.
Global Chaple Disease Therapeutics Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2024 | Market Size in 2025: | USD 145.3 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2025 To 2032 |
| Forecast Period 2025 to 2032 CAGR: | 1.4% | 2032 Value Projection: | USD 160.2 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Alexion Pharmaceuticals Inc., Akari Therapeutics, Apellis Pharmaceuticals, Amgen Inc., CinnaGen Co., Novartis AG, F. Hoffmann-La Roche Ltd, Regeneron Pharmaceuticals Inc., and Alnylam Pharmaceuticals, Inc. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Global Chaple Disease Therapeutics Market- Trends
- Focus on gene therapy and regenerative therapy: Focus on gene therapy and regenerative therapy is one of the major emerging trends influencing the global chaple disease therapeutics market. Chaple disease is a rare inherited disorder which damages cells and tissues over time. Conventional treatments such as analgesics, corticosteroids, and others only manage the symptoms but do not cure the disease. With advancements in genetic research and cell therapy technologies, scientists are developing potentially curative therapies that target the underlying genetic defects causing Chaple disease.
Global Chaple Disease Therapeutics Market - Restraints
- Long approval timelines: The lengthy approval timelines required for new therapeutic drugs is significantly hampering the growth of the global chaple disease therapeutics market. Obtaining regulatory approvals from entities like the U.S. Food and Drug Administration (FDA) and European Medicines Agency (EMA) is a rigorous, time-consuming and costly process. On an average, it takes 8-12 years for a potential new treatment to navigate through the stages of pre-clinical and clinical trials, and regulatory review before it can be available for patients. This prolonged timeline poses challenges for biopharmaceutical companies as well as patients. For drug developers, the vast time commitment and investment that is required to bring a treatment to market affects their return on investment and deters them from pursuing new therapies. It also delays patient access to potentially life-changing or curative innovations. This is especially concerning in the case of rare or orphan diseases like chaple disease where the patient pool itself is small. Any delays in therapy availability directly impact the lives and outcomes of these vulnerable patient populations who have limited or no treatment options otherwise.
Counterbalance – Thus, key market players need to conduct thorough research and compile all required documentation ahead of time and can ensure all materials submitted for approval meet necessary guidelines and are as complete as possible
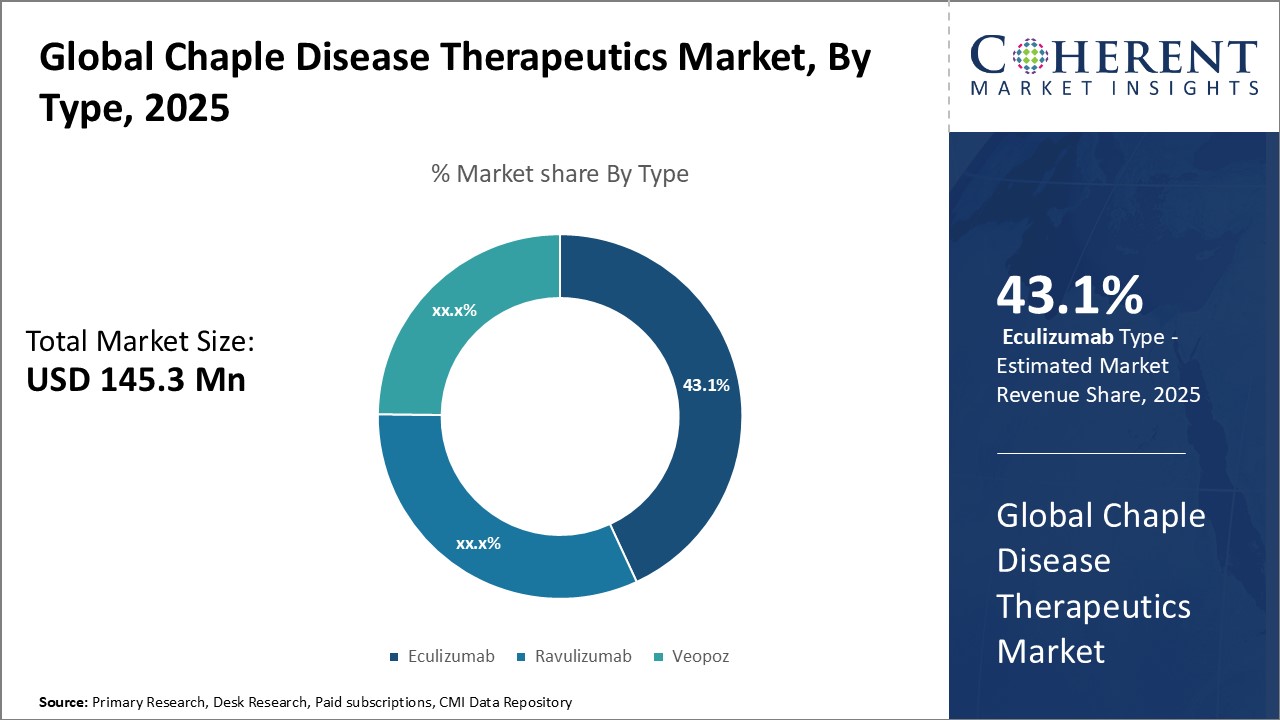
Figure 2. Global Chaple Disease Therapeutics Market Share (%), By Type, 2025
To learn more about this report, Download Free Sample
Top Companies in Global Chaple Disease Therapeutics Market
- Alexion Pharmaceuticals Inc.
- Akari Therapeutics
- Apellis Pharmaceuticals
- Amgen Inc.
- CinnaGen Co.
- Novartis AG
- Hoffmann-La Roche Ltd
- Regeneron Pharmaceuticals Inc.
- Alnylam Pharmaceuticals, Inc.
Definition: CHAPLE—which stands for complement hyperactivation, angiopathic thrombosis, and protein-losing enteropathy is an inherited immune disease that causes the complement system (the part of your immune system that defends the body against injury and foreign invaders like bacteria and viruses) to become overactive. It is caused by mutations of the complement regulator CD55 gene, which can lead to the complement system attacking the body’s own cells.
Share
Share
About Author
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Missing comfort of reading report in your local language? Find your preferred language :
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
Select a License Type
EXISTING CLIENTELE
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients




