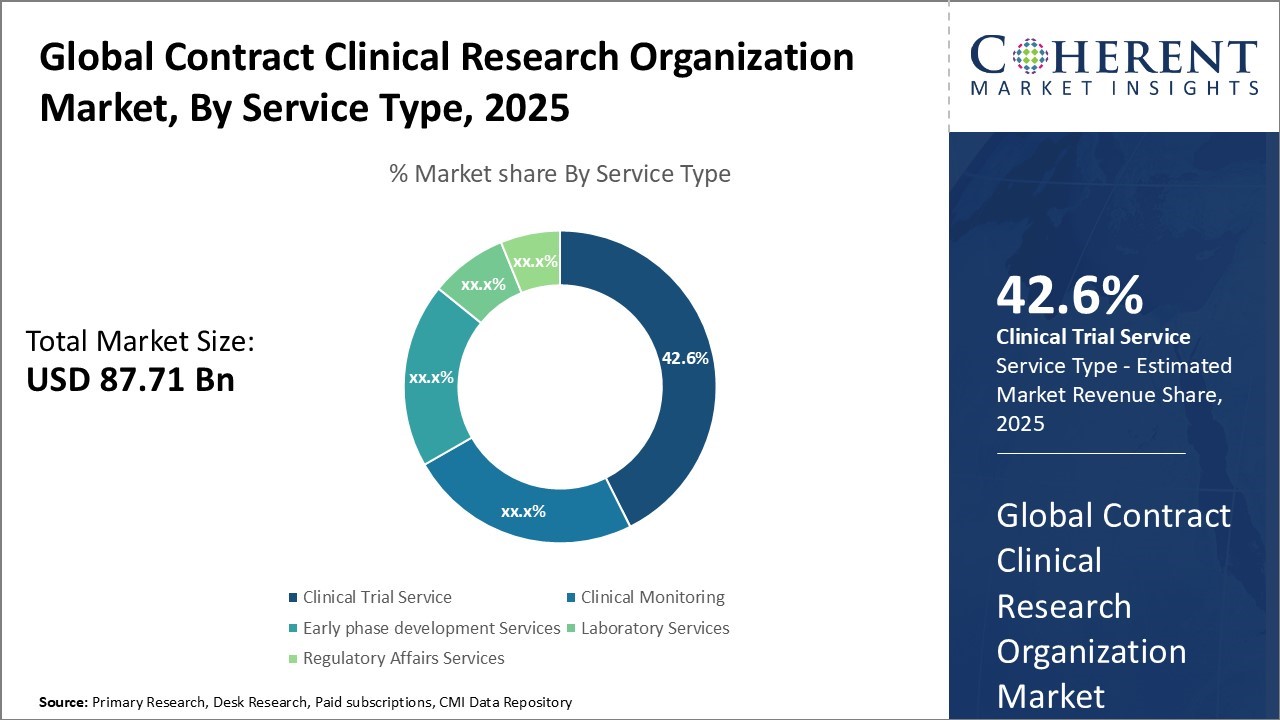
Global Contract Clinical Research Organization Market Size and Forecast
The Global Contract Clinical Research Organization Market is estimated to be valued at USD 87.71 Bn in 2025 and is expected to reach USD 165.56 Bn by 2032, exhibiting a compound annual growth rate (CAGR) of 9.5% from 2025 to 2032.
Key Takeaways of the Global Contract Clinical Research Organization Market
- The clinical trial service segment is expected to contribute the highest share of the market with 42.6% in 2025.
- The oncology segment is expected to contribute the highest share of the market with 37.6% in 2025.
- The pharmaceutical companies segment is expected to contribute the highest share of the market with 40.8% in 2025.
- North America is expected to top the market with 37.8% of the market share in 2025.
- Asia Pacific, holding 29.2% of the market share in 2025, is estimated to exhibit the fastest growth rate during the forecast period.
To learn more about this report, Download Free Sample
Market Overview
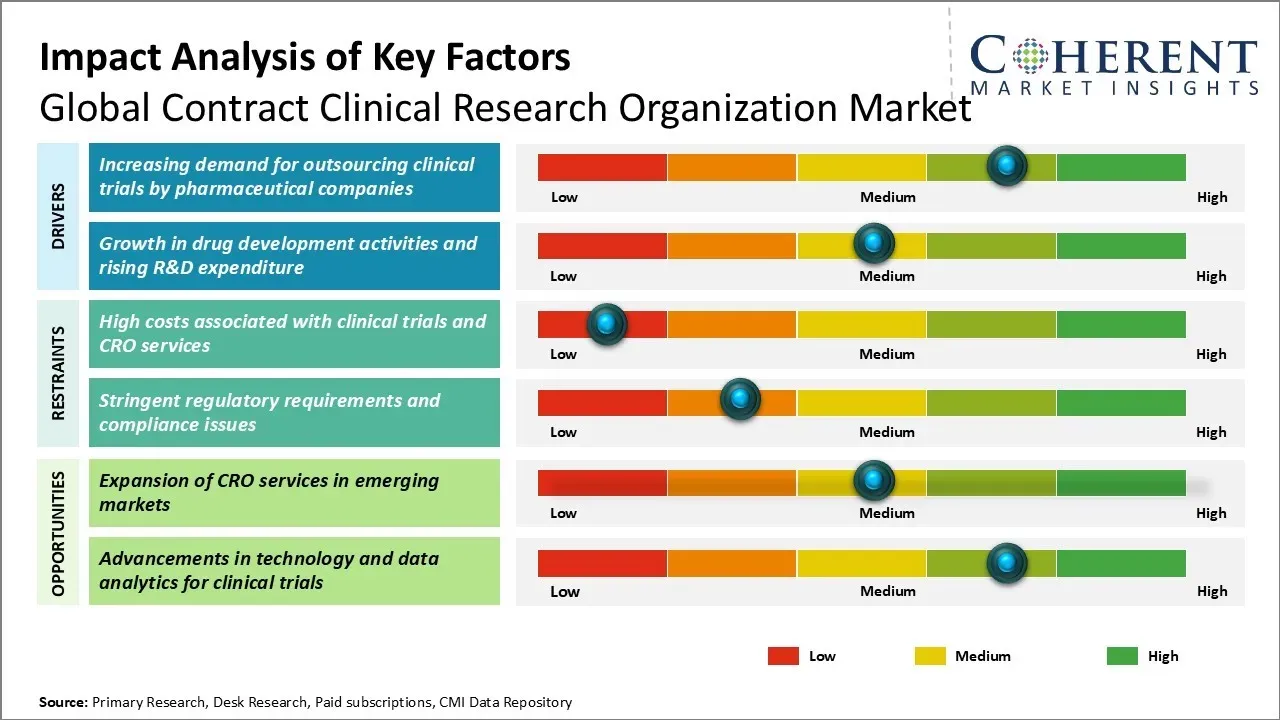
There is an increase in research and development activities in the pharmaceutical and biotechnology industry. Moreover, outsourcing of clinical trials and research activities help companies reduce costs and improve efficiency. Research organizations provide high quality research services to various sponsors which is also driving the market growth. However, lack of skilled workforce and strict regulatory requirements associated with clinical trials may hamper the market growth during the forecast period.
Impact of Artificial Intelligence (AI) on the Global Contract Clinical Research Organization Market
The Contract Clinical Research Organization (CRO) market is witnessing transformative changes driven by the adoption of Artificial Intelligence (AI). As pharmaceutical and biotechnology companies increasingly outsource clinical trials, CROs are leveraging AI to streamline processes, enhance data accuracy, and accelerate drug development timelines. This integration of AI is reshaping the industry, delivering greater efficiency and improved patient outcomes.
A prime example is ICON plc, a global CRO, which has embedded AI-powered technologies across its clinical trial operations. ICON’s strategic initiative involves harnessing machine learning algorithms to analyze vast clinical datasets rapidly, enabling faster identification of patient cohorts and adverse events. This AI integration helps ICON reduce trial timelines and enhance the robustness of trial designs, giving clients a competitive edge in drug development.
Current Events and their Impact on the Global Contract Clinical Research Organization Market
|
Current Events |
Description and its impact |
|
Geopolitical Tensions and Regulatory Changes
|
|
|
Economic and Investment Trends
|
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Global Contract Clinical Research Organization Market Insights, By Service Type
In terms of service type, the clinical trial service segment is expected to contribute the highest share of the market with 42.6% in 2025 owing to the complexities involved in clinical trial design and conduct. Clinical trial service provides end-to-end support for clinical development programs including protocol design, site selection and management, patient recruitment, data management, statistical analysis, medical monitoring, and regulatory support. The success of clinical trials hinges critically on robust trial design and seamless execution. For instance, a complex Phase III cardiovascular trial might require a CRO to manage hundreds of sites globally, recruit thousands of patients with specific inclusion criteria, and handle vast amounts of safety and efficacy data, which is highly challenging for an in-house team to coordinate efficiently.
Global Contract Clinical Research Organization Market Insights, By Therapeutic Area
In terms of therapeutic area, the oncology segment is expected to contribute the highest share of the market with 37.6% in 2025 owing to the immense unmet medical needs. Cancer prevalence is rising globally due to aging population and changing lifestyle factors. However, only a few highly toxic drugs have been approved each year to treat various cancers. There is tremendous urgency to accelerate the discovery and development of novel targeted therapies, immunotherapies, combination regimens, and personalized medicine approaches.
Global Contract Clinical Research Organization Market Insights, By End User
In terms of end user, the pharmaceutical companies segment is expected to contribute the highest share of the market with 40.8% in 2025 owing to the increasing trend of outsourcing non-core functions. Rising costs of drug development have challenged in-house R&D budgets and timelines of pharmaceutical firms. At the same time, intense market competition necessitates the rapid delivery of innovative medicines. This has prompted pharmaceutical firms to increasingly rely on CROs to perform non-core critical path services like clinical operations, biostatistics, and regulatory consultancy. For instance, a large pharmaceutical company developing multiple drugs concurrently might outsource all their Phase I and II clinical operations to a CRO to manage resources efficiently, reduce fixed costs, and leverage the CRO's specialized infrastructure and global reach.
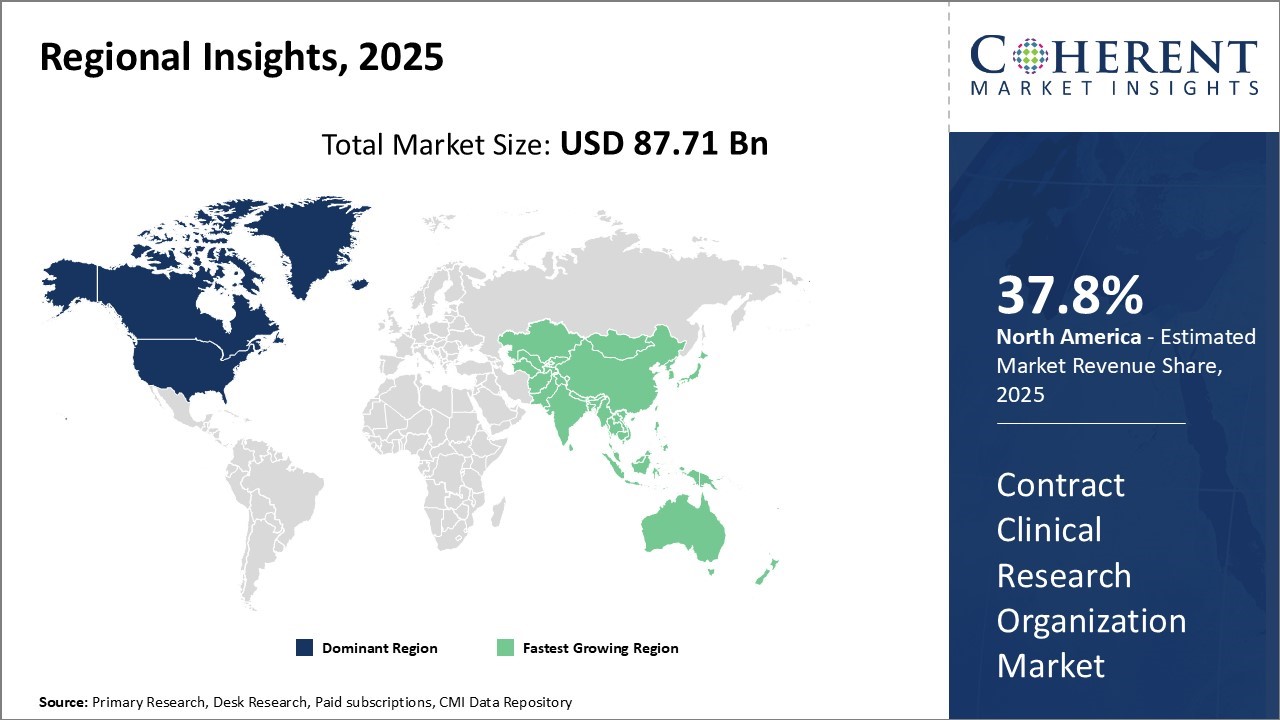
Regional Insights
To learn more about this report, Download Free Sample
North America Contract Clinical Research Organization Market Trends
North America's leadership in the contract clinical research organization market, with an estimated 37.8% share projected for 2025, is driven by the strong presence of established CROs and a supportive regulatory framework. Additionally, high healthcare expenditures and the focus of pharmaceutical companies to outsource non-core activities have further strengthened North America's position.
For instance, according to clinicaltrials.gov, as of January 2024, a total of 477,346 clinical trials are registered across the globe, out of which 30% (145,575) trials are registered in the U.S.
Asia Pacific Contract Clinical Research Organization Market Trends
Asia Pacific region is expected to exhibit the fastest growth with 29.2% in 2025 and is expected to witness significant opportunities over the coming years. This can be credited to the region's low-cost advantages and the ongoing expansion of the pharmaceutical industry in countries such as China and India. Further, government initiatives to develop healthcare infrastructure have amplified clinical research activities. For instance, beyond India's regulatory advancements, China's accelerated pace of domestic drug innovation and substantial increase in biopharmaceutical R&D expenditure are notably driving CRO demand. As of early 2025, Chinese pharmaceutical companies are increasingly developing novel drugs for global markets, necessitating comprehensive clinical trial services from both local and international CROs with strong regional expertise.
Contract Clinical Research Organization Market Outlook for Key Countries
U.S. Contract Clinical Research Organization Market Trends
Rising chronic diseases are driving pharmaceutical companies to outsource clinical trials, fueling contract clinical research organization market growth. In January 2024, according to the American Cancer Society's 2024 update, approximately 2 million new cancer cases are projected to be diagnosed in the U.S. in 2024, equating to around 5,480 new cases daily. This substantial cancer burden underscores the urgent need for expedited regulatory approvals and clinical trial support for cancer therapies.
Canada Contract Clinical Research Organization Market Trends
Canada drives growth in the contract clinical research organization market due to its advanced healthcare infrastructure, rising prevalence of chronic diseases, and diverse patient demographics. For instance, in August 2024, the Canadian Institutes of Health Research (CIHR) announced it was providing CAD 43 million to support 14 clinical trials that target health threats that include TB, sepsis, stroke and COVID‑19. Such funding supports CROs engaged in managing and implementing trials.
China Contract Clinical Research Organization Market Trends
China contract clinical research organization market is experiencing rapid growth, driven by the country’s expanding pharmaceutical industry and a strong focus on enhancing domestic R&D capabilities. In November 2024, Merck signed a USD 3.3 billion deal with China-based, LaNova Medicines, to license an experimental cancer drug, highlighting China’s growing R&D capabilities. The country’s advanced clinical trial infrastructure, competitive costs, and diverse patient pool further drive its contract clinical research organization market growth.
India Contract Clinical Research Organization Market Trends
India continues to lead as a preferred destination for clinical trials owing to its large patient pools and cost advantages. India is emerging as a key player in the contract clinical research organization market, offering cost-effective clinical trial services. For instance, in September 2024, the government framed standard operating procedures (SOPs) for clinical research organizations within the country to maintain safe clinical trials. These are the New Drugs and Clinical Trials (Amendment) Rules, 2024, and these new regulations begin to take effect on April 1 2025. The new regulations will now be applied to all previous Government and private clinical research centres, along with those organizations conducting bioavailability or bioequivalence studies.
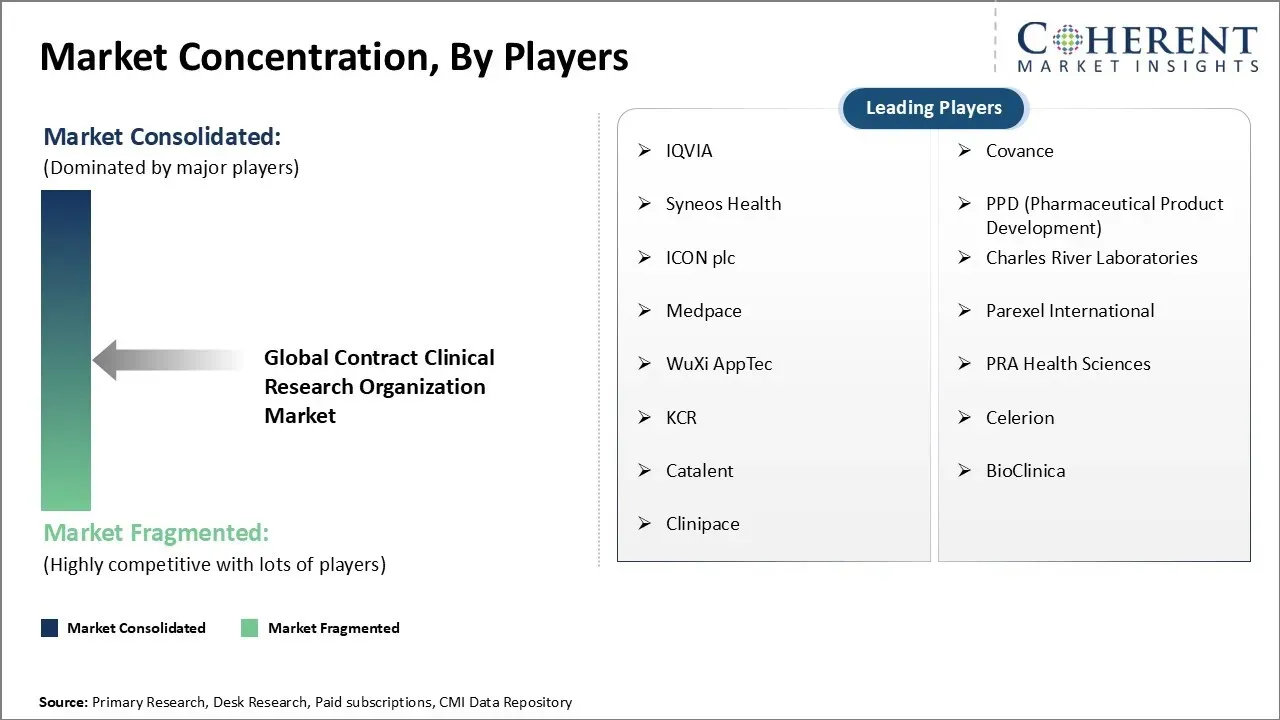
Market Players, Key Development, and Competitive Intelligence
To learn more about this report, Download Free Sample
Global Contract Clinical Research Organization Market Growth Factors - Increasing Demand for Outsourcing Clinical Trials by Pharmaceutical Companies
Pharmaceutical companies increasingly outsource clinical trials to CROs due to the complexity of drug development, which involves extensive research, global site coordination, and large-scale volunteer recruitment across various trial phases. For example, in late 2024 and early 2025, there has been a significant increase in the outsourcing of oncology trials, which now account for 35% of all oncology trial starts, driven by novel modalities like antibody-drug conjugates and cell and gene therapies.
Global Contract Clinical Research Organization Market Opportunity - Expansion of CRO Services in Emerging Markets
The expansion of CRO services in emerging markets presents a major opportunity for the growth of the global contract clinical research organization market. Countries such as China, India, Brazil, and others are emerging as top destinations for the outsourcing of clinical trials due to advantages such as large patient pools, low costs, and expanding medical expertise. These markets offer attractive opportunities for global CROs to expand their geographic footprint and offer clinical research services at competitive costs.
For example, India's vast and diverse patient population, coupled with lower operational costs, continues to attract global trials, with government initiatives further streamlining regulatory processes to boost clinical research. Similarly, China's rapidly expanding pharmaceutical industry and large patient pool are driving substantial CRO investment, as demonstrated by the increasing demand for clinical trial services in the region.
Key Developments
- In June 2024, QPS Holdings, LLC, a global contract research organization (CRO), announced new clinical research operations leadership. This move aims to align its global medical affairs, regulatory affairs, data services, and clinical operations with its Phase I clinics, enhancing agility and efficiency to meet the evolving demands of drug development.
- In February 2024, Ergomed Group, a leading provider of clinical research and pharmacovigilance services, expanded its U.S. footprint by opening a new office in Massachusetts, U.S.
- In February 2024, Charles River Laboratories, a premier provider of preclinical and clinical laboratory services, partnered with Wheeler Bio, a biomanufacturing company, to accelerate the transition from therapeutic discovery to manufacturing for its clients.
- In January 2024, WuXi Biologics, a global leader in biologics R&D and manufacturing, announced a collaboration with BioNTech SE, a biotechnology company, to identify investigational monoclonal antibodies for next-generation therapeutic development.
Market Report Scope
Contract Clinical Research Organization Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2024 | Market Size in 2025: | USD 87.71 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2025 To 2032 |
| Forecast Period 2025 to 2032 CAGR: | 9.5% | 2032 Value Projection: | USD 165.56 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
IQVIA, Covance, Syneos Health, PPD (Pharmaceutical Product Development), ICON plc, Charles River Laboratories, Medpace, Parexel International, WuXi AppTec, PRA Health Sciences, KCR, Celerion, Catalent, BioClinica, and Clinipace |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Analyst Opinion (Expert Opinion)
- Key drivers of growth in the Contract Research Organization (CRO) market include increasing R&D expenditure by large pharmaceutical companies and their aim to outsource non-core functions. Additionally, the need to conduct clinical trials efficiently and cost-effectively will drive more sponsors to utilize CROs.
- However, risks surrounding data privacy and evolving regulatory changes pose significant challenges. Strict data privacy laws being implemented worldwide can escalate compliance costs. Furthermore, delays in site initiation and subject enrollment can negatively impact project timelines and budgets.
- North America will continue dominating the market due to the strong presence of major pharmaceutical companies and biotech startups actively outsourcing clinical trials. Asia Pacific is expected to exhibit the fastest growth, primarily owing to lower trial costs and increasing clinical research expertise in countries like China and India.
- Prominent players in the market are strengthening their capabilities and global footprint to secure more outsourced projects. Many CROs are offering new services, such as clinical trial modeling and solutions for post-marketing studies. The adoption of technologies like e-clinical solutions, artificial intelligence, and Blockchain by CROs is anticipated to improve the efficiency and quality of clinical trials going forward.
Market Segmentation
- Service Type Insights (Revenue, USD Bn, 2020 - 2032)
- Clinical Trial Service
- Phase III
- Phase II
- Phase I
- Phase IV
- Clinical Monitoring
- Site Monitoring
- Data Monitoring
- Early phase development Services
- Chemistry, Manufacturing and Controls Services
- Preclinical Services
- Laboratory Services
- Analytical Testing Services
- Bioanalytical Testing Services
- Regulatory Affairs Services
- Clinical Trial Service
- Therapeutic Area Insights (Revenue, USD Bn, 2020 - 2032)
- Oncology
- Cardiovascular Diseases
- Infectious Diseases
- Neurology
- Other Therapeutic Areas
- End User Insights (Revenue, USD Bn, 2020 - 2032)
- Pharmaceutical Companies
- Biotechnology Companies
- Medical Device Companies
- Research & Academic Institutions
- Regional Insights (Revenue, USD Bn, 2020 - 2032)
- North America
- U.S.
- Canada
- Latin America
- Brazil
- Argentina
- Mexico
- Rest of Latin America
- Europe
- Germany
- U.K.
- Spain
- France
- Italy
- Russia
- Rest of Europe
- Asia Pacific
- China
- India
- Japan
- Australia
- South Korea
- ASEAN
- Rest of Asia Pacific
- Middle East
- GCC Countries
- Israel
- Rest of Middle East
- Africa
- South Africa
- North Africa
- Central Africa
- North America
- Key Players Insights
- IQVIA
- Covance
- Syneos Health
- PPD (Pharmaceutical Product Development)
- ICON plc
- Charles River Laboratories
- Medpace
- Parexel International
- WuXi AppTec
- PRA Health Sciences
- KCR
- Celerion
- Catalent
- BioClinica
- Clinipace
Sources
Stakeholders
- Clinical Research Directors
- Contract Clinical Operations Managers
- Pharmaceutical Sponsorship Leads
- Regulatory Affairs Specialists
- Others
Databases
- Global Clinical Trial Database
- ClinicalTrials.gov
- Pharmaprojects
- Others
Magazines
- Applied Clinical Trials
- Clinical Researcher
- PharmaVOICE
- OutsourcingPharma
- Others
Journals
- Journal of Clinical Research Best Practices
- Therapeutic Innovation & Regulatory Science
- Contemporary Clinical Trials
- Others
Newspapers
- PharmaTimes
- BioPharm International
- The Pharma Letter
- Fierce Biotech
- Others
Associations
- Association of Clinical Research Organizations (ACRO)
- Society for Clinical Research Sites (SCRS)
- Drug Information Association (DIA)
- European CRO Federation
- Others
Public Domain sources
- World Health Organization (WHO)
- U.S. Food and Drug Administration (FDA)
- European Medicines Agency (EMA)
- Others
Proprietary Elements
- CMI Data Analytics Tool
- Proprietary CMI Existing Repository of information for last 8 years
Share
Share
About Author
Manisha Vibhute is a consultant with over 5 years of experience in market research and consulting. With a strong understanding of market dynamics, Manisha assists clients in developing effective market access strategies. She helps medical device companies navigate pricing, reimbursement, and regulatory pathways to ensure successful product launches.
Missing comfort of reading report in your local language? Find your preferred language :
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
EXISTING CLIENTELE
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients