Blood Clot Retrieval Devices Market Size and Trends
The global blood clot retrieval devices market is estimated to be valued at USD 1.87 Bn in 2025 and is expected to reach USD 5.13 Bn by 2032, exhibiting a compound annual growth rate (CAGR) of 15.5% from 2025 to 2032.
Discover market dynamics shaping the industry: Download Free Sample
Factors such as the increasing incidence of strokes worldwide primarily driven by rising geriatric population, growing awareness about treatment options for strokes, and technological advancements in blood clot retrieval devices resulting in improved patient outcomes have been driving the demand for blood clot retrieval devices. Additionally, various studies that have proven the efficacy of mechanical thrombectomy over standard medical management alone in improving revascularization and clinical outcomes for acute ischemic stroke patients are further supporting the growth of this market. However, high costs associated with blood clot retrieval devices and lack of reimbursement in some underdeveloped regions are some of the factors expected to hamper the market growth to some extent during the forecast period
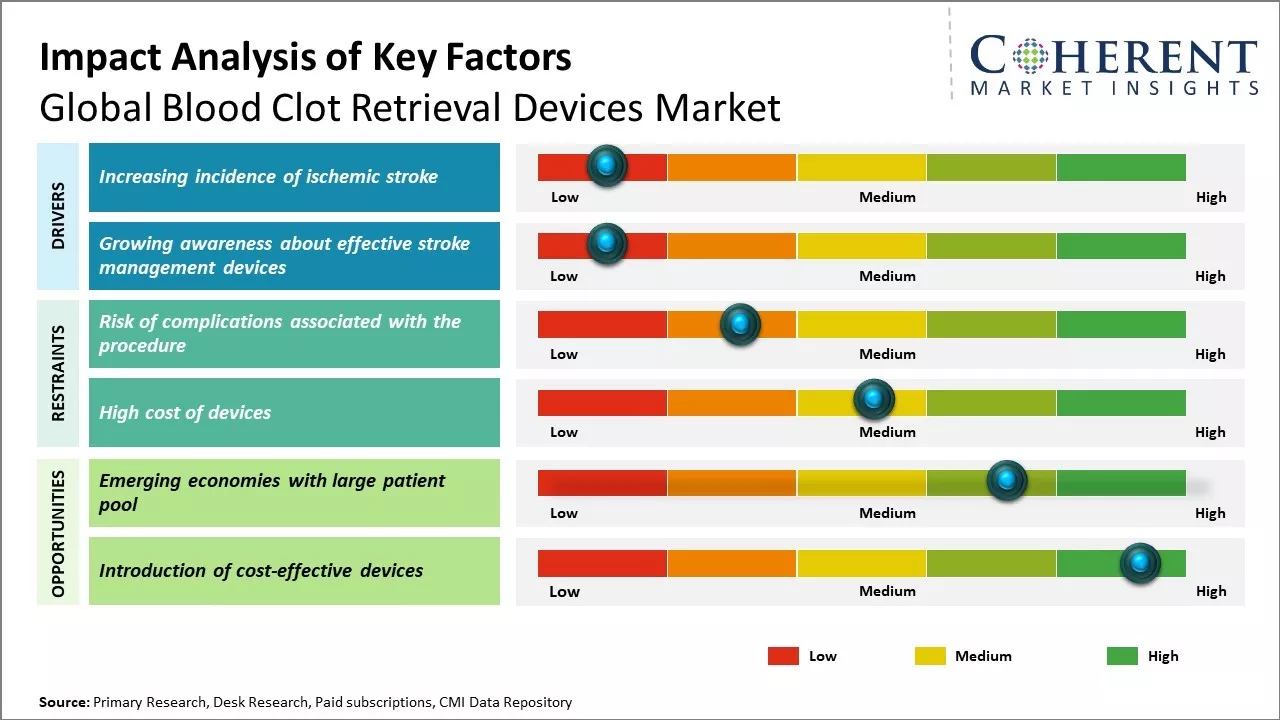
Increasing incidence of ischemic stroke
The global blood clot retrieval devices market is expected to see substantial growth driven by the rising incidence rates of acute ischemic stroke around the world. Stroke is one of the leading causes of disability as well as the fifth most common cause of death worldwide. According to some estimates, over 17 million people suffer from stroke annually and more than 6 million people die each year due to this condition. The chances of stroke also increase with age with over three-quarters of strokes happening in people aged over 65. As the worldwide population is aging at a very fast rate, the number of elderly groups is rising dramatically in several regions. For example, the United Nations predicts that over next three decades, the number of people over the age of 60 will more than double globally. This demographic shift will lead to a subsequent rise in the risk of developing age-related diseases like stroke. Early revascularization through mechanical thrombectomy using blood clot retrieval devices has significantly improved stroke outcomes in patients suffering from large vessel occlusion strokes. Thus, with stroke projected to remain a huge burden globally in coming years, the demand for advanced thrombectomy devices is expected to surge. For instance, in February 2022, significant changes in stroke incidence during the previous 29 years have been observed in the U.S., according to new data presented at the American Stroke Association's International Stroke Conference in 2022. Based on findings: The prevalence of stroke in those 75 years of age and older has significantly declined. On the other hand, stroke rates are rising among individuals under 49, especially in the Midwest and South. Between 1990 and 2019, the number of stroke cases in the U.S. increased by 60% overall, with ischemic strokes accounting for two-thirds of all instances.
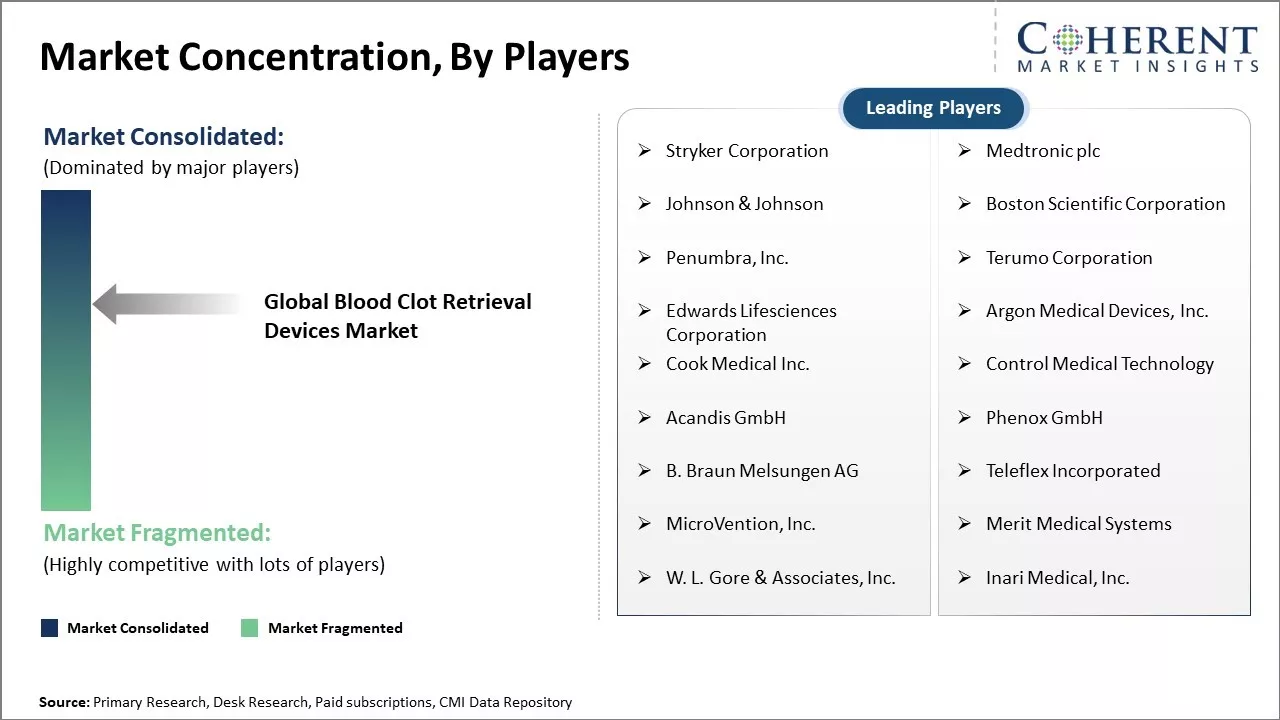
Market Concentration and Competitive Landscape
Get actionable strategies to beat competition: Download Free Sample
Growing awareness about effective stroke management devices
The increased awareness about effective stroke management devices among both healthcare professionals and patients has been a key driver for the growth of the global blood clot retrieval devices market. More people are now recognizing that treating stroke as a medical emergency and ensuring rapid flow of blood to the brain in a timely manner is very critical in improving patient outcomes post stroke. This awareness has been fueled by various educational campaigns conducted by public health organizations to ensure communities understand the importance of ACT (Acting FAST, calling 911, and Targeting care at a Comprehensive Stroke Center). For example, the American Stroke Association's "Time Lost is Brain Lost" campaign emphasized that stroke is a serious medical emergency and every minute counts when it comes to treating a stroke patient. Such initiatives have encouraged more people to seek urgent medical attention in case of a suspected stroke. As a result of this growing understanding of need for quick stroke treatment, there is significant demand for state-of-the-art blood clot retrieval devices that can help clinicians efficiently remove clots from blocked arteries in the brain. These advanced devices such as stent retrievers and aspiration devices have revolutionized the endovascular treatment landscape by improving reperfusion rates and subsequently leading to better clinical outcomes for stroke patients. For example, according to the Society of Vascular and Interventional Neurology, the percentage of eligible stroke patients receiving endovascular treatments within 6 hours has doubled between 2014 and 2020. This reflects increased adoption of mechanical thrombectomy surgeries using clot retrieval tools.
Key Takeaways from Analyst
The global blood clot retrieval devices market is expected to witness significant growth over the forecast period driven by the rising incidence of strokes globally. The growing geriatric population who are more prone to strokes will further drive the demand for blood clot retrieval devices. Additionally, technological advancements in blood clot retrieval devices allowing for improved effectiveness and safety during stroke treatment procedures are expected to provide opportunities for market growth.
North America currently dominates the global market owing to growing awareness about stroke treatment and management options. Supportive reimbursement policies for stroke treatment in the U.S. and availability of advanced healthcare infrastructure are further fueling the market growth in the region. Europe is also expected to account for a considerable market share due to the rising number of neurointerventional labs and initiatives by healthcare organizations to spread awareness among people.
However, high costs associated with blood clot retrieval devices and procedures could hamper market growth to some extent. Limited healthcare infrastructure and access in developing regions of Asia Pacific and Latin America may also restrain market expansion. Nonetheless, increasing healthcare expenditures, expanding private insurance coverage and improving economic conditions are likely to boost the adoption of these devices in middle-income countries over the forecast period.
Market Challenges: Risk of complications associated with the procedure
The risk of complications arising from blood clot retrieval procedures is a major concern restraining the growth of the global blood clot retrieval devices market. Blood clot removal is an intricate procedure involving advancement of wires and catheters through delicate blood vessels to the location of the clot. During this process, there is always a chance of damaging the vessels or dislodging secondary clots. Complications arising from the procedure such as vessel perforation, dissection or rupture can be life-threatening. This makes both physicians and patients apprehensive about using such devices. The risks are considerably higher in certain groups of patients such as the elderly who usually have fragile vessels and those with comorbid conditions like diabetes which weaken the vessel walls. Procedural complications often lead to severe disabilities or mortality. As per World Health Organization data from 2021, complications arising from catheter-based cardiovascular procedures caused approximately 250,000 deaths globally. Such troubling statistics have created a risk-averse mindset among the patient population regarding intravascular treatments. This subsequently restrains the growth potential for blood clot retrieval device manufacturers.
Market Opportunities: Emerging economies with large patient pool
Emerging economies with large patient populations present significant growth opportunities for the global blood clot retrieval devices market. Countries like India, China, Brazil, and other developing nations in Asia Pacific and Latin America are experiencing rapid urbanization and rising incomes. This has led to lifestyle changes like increased stress levels, sedentary jobs and unhealthy diets which are fueling the incidents of cardiovascular diseases and strokes in these regions. As per the World Health Organization's data, cardiovascular diseases accounted for over 15 million premature deaths globally in 2019 alone. Nearly 85% of these deaths occurred in low and middle-income countries. Strokes and other cerebrovascular diseases also claimed 6.2 million lives in the same year. The burden of such illnesses will continue to rise enormously in developing economies over the next decade due to their aging populations and risk factors like smoking, diabetes and obesity becoming more prevalent. This massive patient base needing procedures like mechanical thrombectomy to remove deadly blood clots from brain arteries creates a huge demand driver for specialized medical devices. For companies operating in the blood clot retrieval devices sector, these emerging territories offer tremendous scope for volumes growth. International players are increasingly focusing on expanding their reach into quicker adopting Asian and Latin American healthcare systems through distributor partnerships and setting up local manufacturing sites. This allows addressing unique needs of these markets in a cost effective manner while also catering to rising demand.
Discover high revenue pocket segments and roadmap to it: Download Free Sample
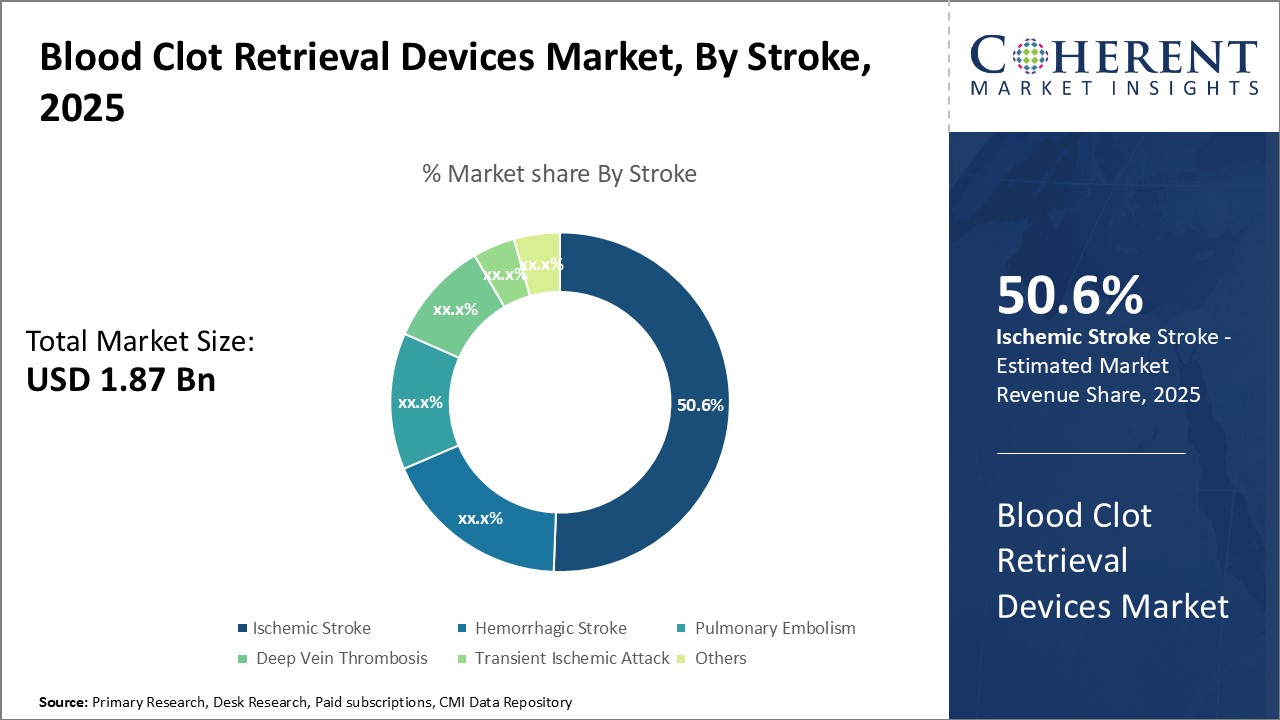
By Stroke - Prevalence of risk factors drives the growth of the Ischemic Stroke segment
In terms of stroke, the Ischemic Stroke segment is expected to contribute 50.6% of the market share in 2025 owing to its high prevalence across the globe. Ischemic strokes occur due to blockage or narrowing of arteries carrying blood to the brain. The primary risk factors for ischemic stroke include hypertension, high cholesterol, diabetes, smoking, lack of physical activity, poor diet, excess alcohol consumption, and cardiac issues such as atrial fibrillation. As per studies, uncontrolled hypertension is the single largest risk factor for ischemic strokes worldwide. The rising incidences of risk factors such as obesity, sedentary lifestyle, and improper diet are major contributors to increased hypertension and diabetes cases globally. For instance, North America and Europe have witnessed significant rise in prevalence of diabetes and obesity in past decade. Similarly, smoking is still highly prevalent in certain regions of Asia Pacific and Latin America. Unless aggressive awareness and preventive measures are taken, the burden of these risk factors and subsequent ischemic strokes cases are expected to rise further. Growing geriatric population is another important demand driver for this segment. Age is the strongest risk factor for strokes and chances of getting stroke doubles every decade after 55 years of age. With worldwide average life expectancy increasing steadily, the older population demographic prone to strokes is growing at an exponential rate. Government initiatives focusing on public education regarding warning signs, risk factors and importance of timely treatment for strokes are creating awareness. This is encouraging more patients to seek medical help immediately in case of a potential stroke. Widespread access to advanced stroke treatment technologies in developed nations and growing affordability in developing countries is additionally aiding early diagnosis and management of ischemic strokes.
By Device - Prevalence of risk behaviors fuel the growth of the mechanical embolus removal devices segment
In terms of device, mechanical embolus removal devices is expected to contribute 30.5% of the market share in 2025 owing to their higher adoption rate compared to other device types. Mechanical embolus removal devices are retrieval stents used to capture and remove blood clots from cerebral arteries of patients experiencing ischemic strokes. Changing risk behaviors among population such as rising smoking prevalence, binge drinking, and uncontrolled diabetes are resulting in higher cases of deep vein thrombosis, pulmonary embolism, and subsequent demand for clot removal. Cigarette smoke contains chemicals that make the blood stickier and more likely to form clots. Excess alcohol intake dehydrates the body and reduces the ability of blood to flow smoothly through veins. Poor blood sugar management increases clotting risks as well. Procedural advantages of mechanical embolus removal devices such as ability to restore blood flow in just one pass and minimum vessel trauma during clot capture are increasing physician preference for these devices. Advancements including modified stent designs for improved navigation in narrow vessels and use of radiopaque and magnetic materials for better visualization under imaging are further aiding their adoption. Rising affordability of endovascular procedures and availability of device reimbursements in certain regions is positively impacting the uptake of mechanical embolus removal devices. Successful clinical trials and approvals demonstrating safety and effectiveness of these devices for clot removal are additionally supporting the market growth.
By Application - Prevalence of cardiac conditions drive growth in the coronary arteries application segment
In terms of application, the coronary arteries segment is expected to contribute 40.5% of the market share in 2025 owing to high caseload of associated cardiac conditions globally. Coronary arteries supply blood to the heart muscle. Blockages or clots in these arteries can lead to heart attacks. Diseases such as coronary artery disease (CAD), peripheral artery disease, and atherosclerosis that cause plaque buildup or narrowing of coronary arteries are highly prevalent worldwide. Risk factors for these conditions include aging, family history, smoking, diabetes, hypertension, obesity, high cholesterol, and sedentary lifestyle. Developed economies have witnessed steep rise in CAD incidence rates in the aging population with aforementioned risk profiles over past few decades. Procedural benefits of minimally invasive endovascular therapies employing clot retrieval devices such as lower complication rates compared to open heart surgeries and faster recovery times are driving patients and physicians’ preference for these treatments. Improved reimbursement policies for cardiovascular procedures are supported their uptake. Growing focus on preventing heart attacks and related deaths through government led coronary health awareness campaigns aimed at public and targeting modifiable risk factors is resulting in increased diagnostic rates of coronary artery blockages. This is facilitating the demand for clot removal devices in coronary applications.
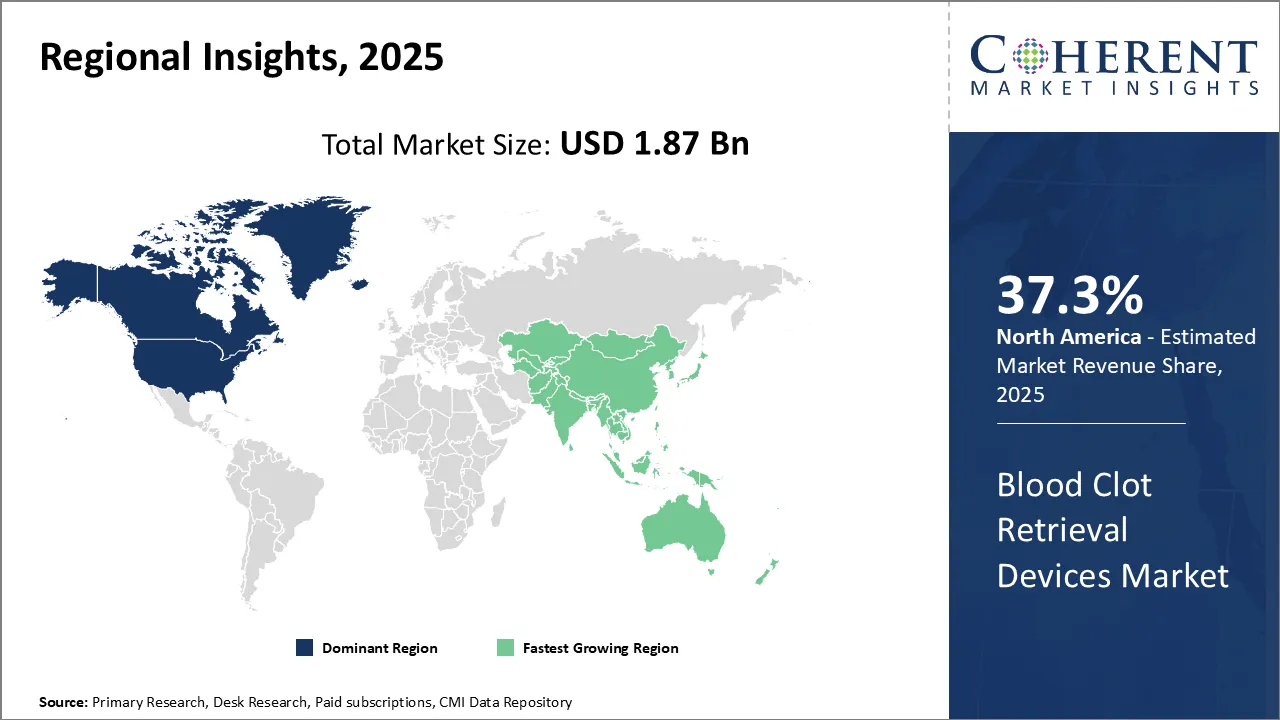
Regional Insights
Need a Different Region or Segment? Download Free Sample
North America has dominated the global blood clot retrieval devices market. The region is expected to account for 37.3% of the market share in 2025 and is expected to continue its leadership position during the forecast period. The strong dominance of the region can be attributed to factors like the growing prevalence of cardiovascular diseases, rising healthcare expenditure, presence of leading medical device manufacturers, and awareness about technologically advanced devices. Countries like the U.S. and Canada have a high adoption rate of novel technologies in healthcare, which has significantly contributed to the large market size of blood clot retrieval devices in North America.
The export and import activities of blood clot retrieval devices are also quite high within North American countries and other regions. For example, the trade of thrombectomy devices between the U.S. and European countries is a major factor supporting the regional market. Furthermore, reimbursement coverage for mechanical thrombectomy procedures has boosted the demand and affordability of blood clot retrieval devices in North America.
The Asia Pacific region has emerged as the fastest growing market for blood clot retrieval devices globally. The growth can be accredited to improving healthcare infrastructure, rising healthcare expenditure, increasing incidence of cardiovascular diseases, growing medical tourism, and presence of cost-effective treatment options. Countries like China, India, Japan, and South Korea are the major contributors to the Asia Pacific market. These countries are witnessing rapid economic development which has positively impacted the regional blood clot retrieval devices market. Rising disposable incomes, focus on quality care at affordable prices, and availability of latest technologies at lower prices than developed markets are some key factors driving the APAC market. The trade dynamics and pricing models of blood clot retrieval devices also favor growth in the Asia Pacific region.
Market Report Scope
Blood Clot Retrieval Devices Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2024 | Market Size in 2025: | USD 1.87 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2025 To 2032 |
| Forecast Period 2025 to 2032 CAGR: | 15.5% | 2032 Value Projection: | USD 5.13 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Stryker Corporation, Medtronic plc, Johnson & Johnson, Boston Scientific Corporation, Penumbra, Inc., Terumo Corporation, Edwards Lifesciences Corporation, Argon Medical Devices, Inc., Cook Medical Inc., Control Medical Technology, Acandis GmbH, Phenox GmbH, B. Braun Melsungen AG, Teleflex Incorporated, MicroVention, Inc., Merit Medical Systems, W. L. Gore & Associates, Inc., and Inari Medical, Inc. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Market Segmentation
- Stroke Insights (Revenue, USD Bn, 2020 - 2032)
- Ischemic Stroke
- Hemorrhagic Stroke
- Pulmonary Embolism
- Deep Vein Thrombosis
- Transient Ischemic Attack
- Others
- Device Insights (Revenue, USD Bn, 2020 - 2032)
- Mechanical Embolus Removal Devices
- Penumbra Blood Clot Retrieval Devices
- Stent Retrievers
- Aspiration Device
- Ultrasound Assisted Devices
- Application Insights (Revenue, USD Bn, 2020 - 2032)
- Coronary Arteries
- Peripheral Arteries
- Cerebral Arteries
- End User Insights (Revenue, USD Bn, 2020 - 2032)
- Hospitals
- Diagnostic Centers
- Specialty Clinics
- Ambulatory Surgical Centers
- Others
- Regional Insights (Revenue, USD Bn, 2020 - 2032)
- North America
- U.S.
- Canada
- Latin America
- Brazil
- Argentina
- Mexico
- Rest of Latin America
- Europe
- Germany
- U.K.
- Spain
- France
- Italy
- Russia
- Rest of Europe
- Asia Pacific
- China
- India
- Japan
- Australia
- South Korea
- ASEAN
- Rest of Asia Pacific
- Middle East
- GCC Countries
- Israel
- Rest of Middle East
- Africa
- South Africa
- North Africa
- Central Africa
- North America
- Key Players Insights
- Stryker Corporation
- Medtronic plc
- Johnson & Johnson
- Boston Scientific Corporation
- Penumbra, Inc.
- Terumo Corporation
- Edwards Lifesciences Corporation
- Argon Medical Devices, Inc.
- Cook Medical Inc.
- Control Medical Technology
- Acandis GmbH
- Phenox GmbH
- B. Braun Melsungen AG
- Teleflex Incorporated
- MicroVention, Inc.
- Merit Medical Systems
- W. L. Gore & Associates, Inc.
- Inari Medical, Inc.
Share
Share
About Author
Komal Dighe is a Management Consultant with over 8 years of experience in market research and consulting. She excels in managing and delivering high-quality insights and solutions in Health-tech Consulting reports. Her expertise encompasses conducting both primary and secondary research, effectively addressing client requirements, and excelling in market estimation and forecast. Her comprehensive approach ensures that clients receive thorough and accurate analyses, enabling them to make informed decisions and capitalize on market opportunities.
Missing comfort of reading report in your local language? Find your preferred language :
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
EXISTING CLIENTELE
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients
