Biosimilar Contract Manufacturing Market Size and Trends – 2025 - 2032
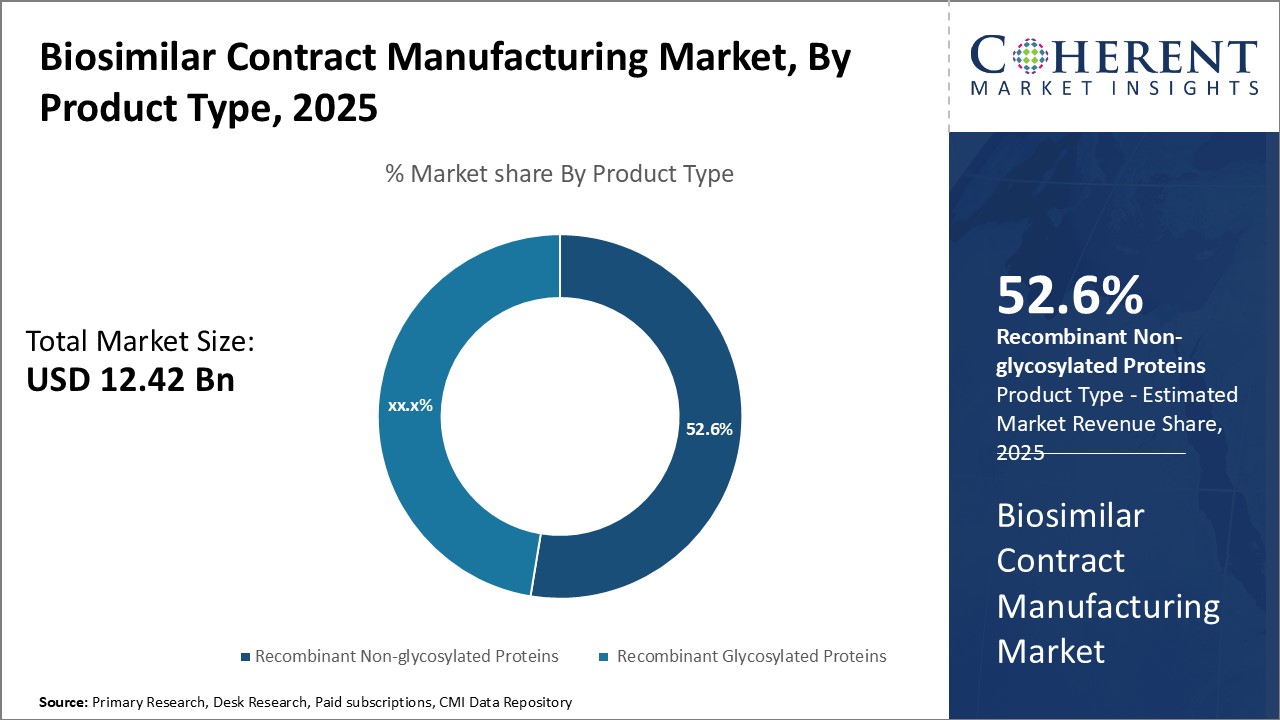
The Global Biosimilar Contract Manufacturing Market is estimated to be valued at USD 12.42 Bn in 2025 and is expected to reach USD 35.35 Bn by 2032, exhibiting a compound annual growth rate (CAGR) of 16.1% from 2025 to 2032.
Key Takeaways of the Biosimilar Contract Manufacturing Market:
- The recombinant non-glycosylated proteins segment is expected to contribute the highest share of the market with a share of 52.6% in 2025.
- The oncology segment is expected to contribute the highest share of the market with 31.6% in 2025.
- The mammalian manufacturing segment is expected to contribute the highest share of the market with 58.6% in 2025.
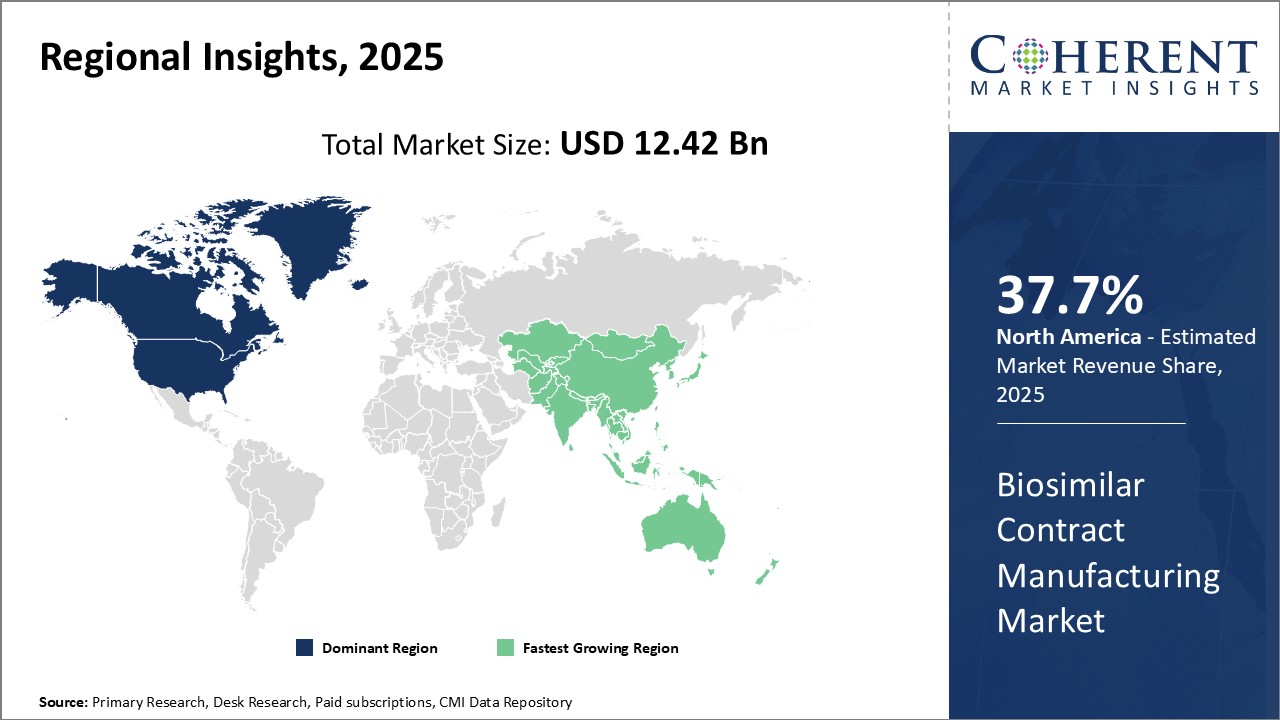
- North America is expected to top the market with 37.7% share, followed by Asia Pacific with 28.2% share in 2025.
Market Overview:
The market growth is attributed to the rising demand for biosimilar drugs due to the increasing prevalence of chronic diseases. In addition, the expiration of patents for biologic drugs and rising demand for affordable healthcare are expected to provide market players with lucrative opportunities. However, high costs associated with biosimilar development and manufacturing may hamper the market growth.
Product Type Insights - Growing Therapeutic Applications Drive the Demand for Recombinant Non-glycosylated Proteins
In terms of product type, the recombinant non-glycosylated proteins segment is expected to contribute the highest share of the market with a share of 52.6% in 2025 owing to their extensive therapeutic applications. Recombinant non-glycosylated proteins are produced outside an organism's native system and lack of sugar chains, making them simpler and less expensive to manufacture than glycosylated variants. Their straightforward production process allows for higher yields and lower costs, increasing accessibility for patients.
Application Insights - Rising Cancer Incidence Catalyzes Oncology Application Growth
In terms of application, the oncology segment is expected to contribute the highest share of the market with 31.6% in 2025, driven predominantly by rising cancer rates worldwide. Cancer diagnoses have climbed in step with risk factors like tobacco use, overweight/obesity, and environmental pollutants becoming more prevalent globally. The global rise in cancer diagnoses can be attributed to the growing prevalence of lifestyle-related risk factors, such as tobacco use, which remains a leading cause of cancer, alongside the rising rates of overweight and obesity due to unhealthy diets and sedentary lifestyles. Environmental pollutants, including exposure to carcinogenic substances in air, water, and soil, further exacerbate the risk of developing various types of cancers.
Type Insights - Mammalian Systems Dominate Due to Cancer Capabilities for Complex Protein Synthesis
In terms of type, the mammalian manufacturing segment is expected to contribute the highest share of the market with 58.6% in 2025 owing to its unique abilities compared to microbial systems. Most biosimilars rely on complex, human-like proteins that possess post-translational modifications such as glycosylation, phosphorylation, and disulfide bonding imparting 3D structure, activity, and stability. Mammalian cell lines offer a eukaryotic environment resembling human physiology able to produce properly folded, functional proteins.
Regional Insights
Need a Different Region or Segment? Download Free Sample
North America Biosimilar Contract Manufacturing Market Trends
North America dominates the biosimilar contract manufacturing market with an estimated share of 37.7% in 2025. This can be attributed to factors such as the strong presence of contract manufacturing organizations CMOs and the presence of well-established biopharmaceutical companies. Countries, such as the U.S. and Canada, have significantly invested in developing their biopharmaceutical ecosystem over the years through supportive regulations and funding for R&D. Several major biologics manufacturers in the region, such as Pfizer, also contribute to the market growth through their partnerships with CMOs for outsourcing biosimilar manufacturing needs.
Asia Pacific Biosimilar Contract Manufacturing Market Trends
The Asia Pacific region exhibits the fastest growth with an estimated share of 28.2% in 2025 and is expected to emerge as an important player driven by rising investments from governments and private sector to bolster local biosimilar drug development and manufacturing capabilities. Countries such as China, India, and South Korea offer strategic advantages in terms of skilled workforce, infrastructure development through initiatives such as special economic zones, and favorable business environment which have helped attract biopharmaceutical companies seeking to expand operations overseas.
Biosimilar Contract Manufacturing Market Outlook for Key Countries
U.S. Biosimilar Contract Manufacturing Market Trends
The U.S. biosimilar contract manufacturing market is poised for significant growth in the coming years. The U.S. government's initiatives to lower healthcare costs, including policies promoting the adoption of biosimilars, have fueled demand. The increasing acceptance of biosimilars in the U.S., coupled with the expiration of numerous biologic patents, has encouraged pharmaceutical companies to outsource manufacturing to contract manufacturers. In May 2024, Boehringer Ingelheim partnered with Quallent Pharmaceuticals to expand access to its citrate-free adalimumab-adbm, a biosimilar to Humira, in the U.S. This collaboration aims to increase availability for patients with chronic inflammatory diseases, reduce healthcare costs, and promote biosimilar adoption in the U.S. biosimilar contract manufacturing market.
Canada Biosimilar Contract Manufacturing Market Trends
Canada biosimilar contract manufacturing industry is growing steadily due to rising new product launches & approvals. For instance, in January 2022, Health Canada approved SIMLANDI, a biosimilar to Humira developed by Alvotech, a global biopharmaceutical company and marketed by JAMP Pharma, a Canada-based pharmaceutical company. It is the first high-strength version of this medicine available in Canada, similar to the popular formulations used in the U.S.
Germany Biosimilar Contract Manufacturing Market Trends
Germany's strong biopharmaceutical infrastructure supports biosimilar development. The government’s investment in healthcare innovation, including a USD 3.15 billion funding initiative announced in January 2024, is expected to boost biosimilar production capabilities, as reported by the Federal Ministry of Education and Research (BMBF).
India Biosimilar Contract Manufacturing Market Trends
India is a key player in the biosimilar manufacturing sector, leveraging cost advantages. The "Make in India" initiative, launched in March 2023, aims to enhance local manufacturing capabilities and attract global partnerships in biosimilars, as stated by the government of India.
Market Players, Key Devlopment, and Competitive Intelligence

Get actionable strategies to beat competition: Download Free Sample
Key Developments:
- On May 2024, Boehringer Ingelheim Biopharmaceuticals GmbH, a global leader in biopharmaceuticals known for its innovative medicines and therapies, announced a strategic partnership with Quallent Pharmaceuticals, a pharmaceutical company specializing in global distribution and healthcare solutions
- On May, 2024, Boehringer Ingelheim, a global leader in pharmaceuticals, announced a partnership with Quallent Pharmaceuticals to enhance access to its citrate-free biosimilar adalimumab-adbm, a treatment for chronic inflammatory diseases. This collaboration aims to increase availability and affordability of the biosimilar in the U.S., supporting Boehringer Ingelheim’s commitment to improving healthcare access and lowering costs for patients and the healthcare system.
- On September, 2023, Abbott, a global healthcare leader, announced a strategic agreement with mAbxience Holdings S.L., a Spain-based biotech company, to commercialize several biosimilars in emerging markets. Abbott will register and distribute these biosimilars, focusing on oncology, women’s health, and respiratory diseases, in key regions across Latin America, Southeast Asia, the Middle East, and Africa. The first biosimilars are expected to launch as early as 2025. This collaboration expands Abbott's portfolio and aims to improve access to affordable treatments in underserved regions.
- In June 2023, Samsung Biologics, a leading contract manufacturing organization (CMO) based in South Korea, and global pharmaceutical company, Pfizer, announced a strategic partnership on June 8, 2023, to enhance the manufacturing of Pfizer’s biosimilars portfolio. The agreement focuses on large-scale production for biosimilars in oncology, inflammation, and immunology.
Top Strategies Followed by Global Biosimilar Contract Manufacturing Market Players
- Established Players: Leading companies in the global biosimilar contract manufacturing market focus heavily on research and development to drive innovation. They invest extensively in R&D to develop high-performance biosimilar drugs with improved efficacy.
- For instance, in March 2024, Merck & Co. allocated approximately 50.8% of its total revenue, amounting to USD 30.5 billion, towards R&D, reflecting a significant commitment to advancing biologics and biosimilars
- Mid-Level Players: Mid-sized companies aim to deliver quality and affordable biologics and biosimilars. They target price-sensitive customer segments by offering cost-competitive solutions. These players invest in automation and digital technologies to optimize production processes.
- In October 2024, Teva Pharmaceuticals and mAbxience have signed a global licensing agreement to develop an anti-PD-1 oncology biosimilar.
- Small-Scale Players: Small biosimilar manufacturers focus on specialized market areas left untapped by larger competitors. They develop niche products targeting rare diseases, specific therapeutic areas, or regional markets.
- For instance, Biocon focuses on developing biosimilars for diabetes and oncology, addressing the growing demand in these segments, particularly in emerging markets
Emerging Startups - Global Biosimilar Contract Manufacturing Industry Ecosystem
- Innovative Technologies: Several biosimilar startups are developing innovative technologies like smart biosensors and AI solutions. For example, ‘BioX’ uses AI to accelerate cell-line development. Its neural networks help achieve a 10x increase in recombinant protein yields. Another startup ‘Biotech Labs’ is commercializing an implantable biosensor that monitors insulin levels in real-time via a smartphone app. Such advanced technologies have potential to significantly improve production efficiencies and patient outcomes.
- Sustainable Solutions: Some emerging companies focus on sustainability. ‘GreenBio’ utilizes agricultural waste as raw material for monoclonal antibody production. It has successfully produced two biosimilars with over 90% recycled materials content. ‘EcoMeds’ is commercializing a biodegradable polymer alternative to glass vials using plant cellulose.
Market Report Scope
Biosimilar Contract Manufacturing Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2024 | Market Size in 2025: | USD 12.42 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2025 To 2032 |
| Forecast Period 2025 to 2032 CAGR: | 16.1% | 2032 Value Projection: | USD 35.35 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Samsung Biologics, Biocon, Amgen, Pfizer, Boehringer Ingelheim, Lonza, Catalent, Wuxi Biologics, AbbVie, Merck KGaA, Rentschler Biopharma, Almac Group, Fujifilm Diosynth Biotechnologies, Evonik Industries, and Avid Bioservices |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Market Dynamics
Discover market dynamics shaping the industry: Download Free Sample
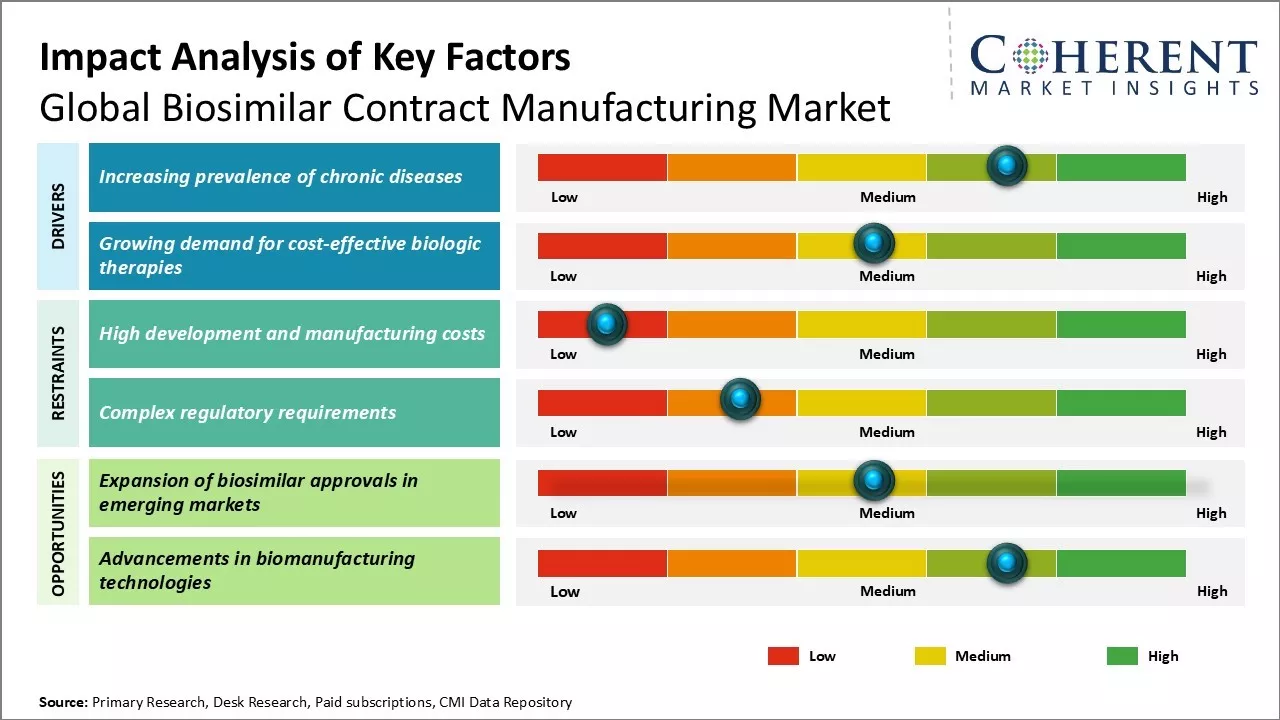
Global Biosimilar Contract Manufacturing Market Growth Factors - Increasing prevalence of chronic diseases
The rising prevalence of chronic diseases globally is driving the demand for biologics and biosimilars, as these conditions often require long-term or lifelong treatments that can be financially burdensome without insurance or public health support. For instance, In May 2023, data published by United Nations News highlighted a warning from the World Health Organization (WHO) that non-communicable diseases (NCDs) such as cardiovascular diseases, cancer, and diabetes account for three-quarters of global deaths annually. This figure is projected to rise to 86% of the estimated 90 million deaths by 2050.
Global Biosimilar Contract Manufacturing Market Challenge - High development and manufacturing costs
One of the major challenges faced by players in the global biosimilar contract manufacturing market is the high development and manufacturing costs associated with biosimilars. Developing a biosimilar is an expensive and complex process as it requires advanced technologies for analytical comparisons, comprehensive clinical testing, and clinical development programs. Strict regulatory guidelines also necessitate extensive analytical, preclinical, and clinical studies to demonstrate biosimilarity which substantially increases R&D investments.
Global Biosimilar Contract Manufacturing Market Opportunity - Expansion of biosimilar approvals in emerging markets
One of the major opportunities for players in the global biosimilar contract manufacturing market is the expansion of biosimilar approvals and uptake in emerging markets. Emerging countries in Asia Pacific, Latin America, the Middle East, and Africa provide an important growth avenue as they contribute significantly to the global healthcare spending. Regulatory frameworks supporting biosimilar development and commercialization are being established in many emerging nations. For instance, countries like China, India, Brazil, and South Africa have demonstrated progressive biosimilar regulations and a conducive environment for local manufacturing.
Analyst Opinion (Expert Opinion)
- The global biosimilar contract manufacturing market possesses strong momentum driven by the increasing development of biosimilars and the need to reduce production costs. As patents of major biologics expire, the demand for lower-cost biosimilars is set to surge in the coming years. This growing demand represents a major opportunity for contract manufacturers to gain new business. North America is expected to dominate the market owing to supportive regulatory guidelines and early acceptance of biosimilars. However, Asian markets like China and India are emerging as fastest growing regions due to increasing biologics manufacturing capabilities and reducing costs in these countries.
- While market trends remain very positive, capacity constraints, and heavy investments required for setting up facilities pose challenges to contract manufacturers. Additionally, the complex nature of biosimilar development and regulatory approvals remains a major restraint. Any manufacturing or quality issues can delay projects and impact revenues. Furthermore, intellectual property disputes between innovator companies and biosimilar developers may hamper market growth. Overall, the lucrative prospects of the biosimilars market, coupled with the need to share risks, provide a compelling case for biopharma companies to engage CMOs through the product lifecycle.
Market Segmentation
- Product Type Insights (Revenue, USD Bn, 2020 - 2032)
-
- Recombinant Non-glycosylated Proteins
- Recombinant Glycosylated Proteins
- Application Insights (Revenue, USD Bn, 2020 - 2032)
-
- Oncology
- Diabetes
- Infectious Diseases
- Chronic and Autoimmune Disorders
- Blood Disorders
- Growth Hormonal Deficiency
- Type Insights (Revenue, USD Bn, 2020 - 2032)
-
- Mammalian Manufacturing
- Microbial Manufacturing
- Service Type Insights (Revenue, USD Bn, 2020 - 2032)
-
- Upstream Processing
- Downstream Processing
- Biosimilarity testing
- Bioassay (in vitro/in vivo)
- Process Development
- Fill & Finish
- End User Insights (Revenue, USD Bn, 2020 - 2032)
-
- Biopharmaceutical Companies
- Contract Manufacturing Organizations (CMOs)
- Others
- Regional Insights (Revenue, USD Bn, 2020 - 2032)
- North America
- U.S.
- Canada
- Latin America
- Brazil
- Argentina
- Mexico
- Rest of Latin America
- Europe
- Germany
- U.K.
- Spain
- France
- Italy
- Russia
- Rest of Europe
- Asia Pacific
- China
- India
- Japan
- Australia
- South Korea
- ASEAN
- Rest of Asia Pacific
- Middle East
- GCC Countries
- Israel
- Rest of Middle East
- Africa
- South Africa
- North Africa
- Central Africa
- North America
- Key Players Insights
- Biocon
- Amgen
- Pfizer
- Boehringer Ingelheim
- Lonza
- Catalent
- Wuxi Biologics
- AbbVie
- Merck KGaA
- Rentschler Biopharma
- Almac Group
- Fujifilm Diosynth Biotechnologies
- Evonik Industries
- Avid Bioservices
Share
Share
About Author
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Missing comfort of reading report in your local language? Find your preferred language :
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
EXISTING CLIENTELE
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients