Bacteriostatic Water For Injection Market Size and Trends
Global bacteriostatic water for injection market is estimated to be valued at USD 1.32 Billion in 2025 and is expected to reach USD 2.29 Billion by 2032, exhibiting a compound annual growth rate (CAGR) of 8% from 2025 to 2032. Global bacteriostatic water for injection growth is driven by factors like rising prevalence of chronic diseases and increasing number of surgeries being performed.
Discover market dynamics shaping the industry: Download Free Sample
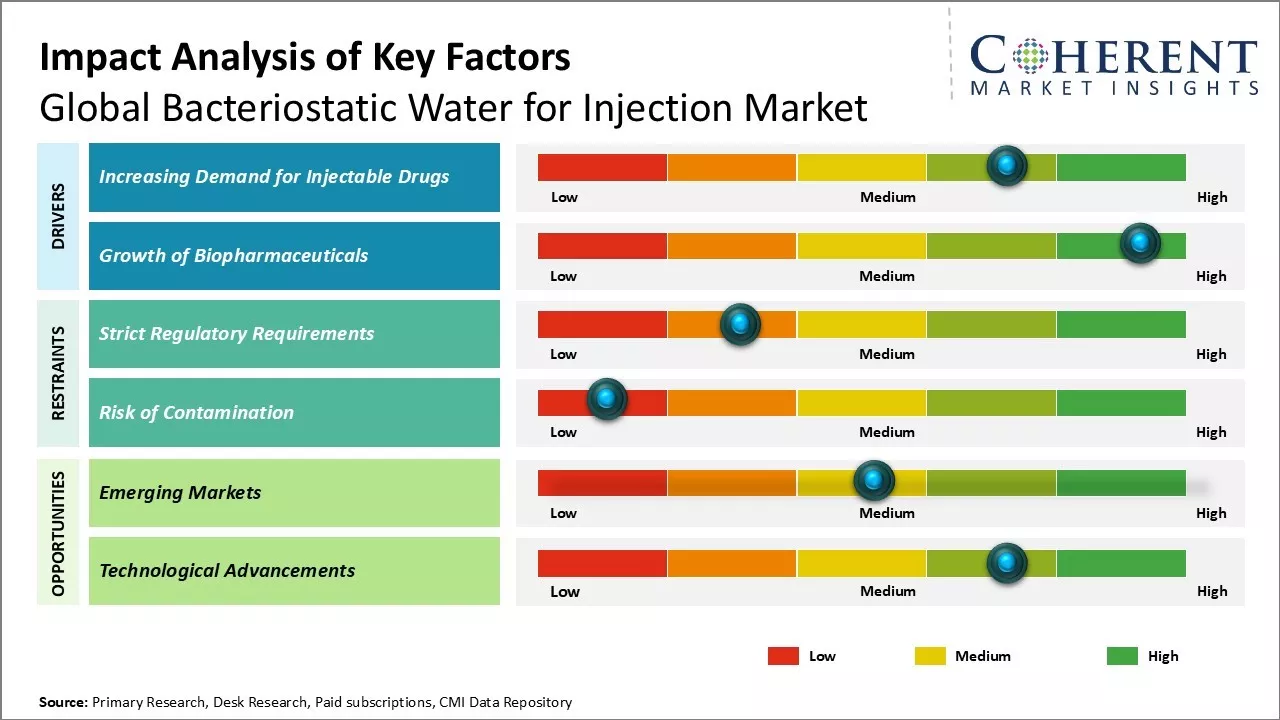
Market Driver - Increasing demand for injectable drugs
Rising demand for injectable drugs due to their rapid absorption and onset of action can drive the market growth. Administering drugs intravenously allows for direct introduction into the bloodstream, thus, bypassing the hepatic first-pass metabolic process that helps in attaining higher bioavailability.
Rising one-time dosage medications, which are very convenient for patients opposed to oral treatments requiring multiple daily pills, also boosts preference for injectable drugs. Self-injectable devices have become very sophisticated, thus, allowing for convenient home administration without requiring frequent hospital or clinic visits. This has especially benefited chronic disease patients in need of long-term management. Geriatric population prone to various co-morbidities boosts demand for injectable drugs to counter poor absorption related to aging physiology and difficulties in swallowing large oral pills.
Increasing complexity of drug molecules and monoclonal antibodies has raised safety concerns about toxic contaminants if administered orally. Sterile packaging and filling of injectable drugs ensures exclusion of pyrogens, microbial contaminants and other impurities. This makes injections the only viable option for these advanced medications, which are being developed intensively for severe diseases. The injectable route of delivery also allows inclusion of controlled release formulations through depot injections to achieve steadier drug levels over weeks or months with less frequent dosing. For instance, in May 2022, the U.S. Food and Drug Administration granted approval to Eli Lilly for Mounjaro (tirzepatide) injection, a novel once-weekly treatment that functions as a GIP (glucose-dependent insulinotropic polypeptide) and GLP-1 (glucagon-like peptide-1) receptor agonist. This medication is designed to enhance glycemic control in adults diagnosed with type 2 diabetes.
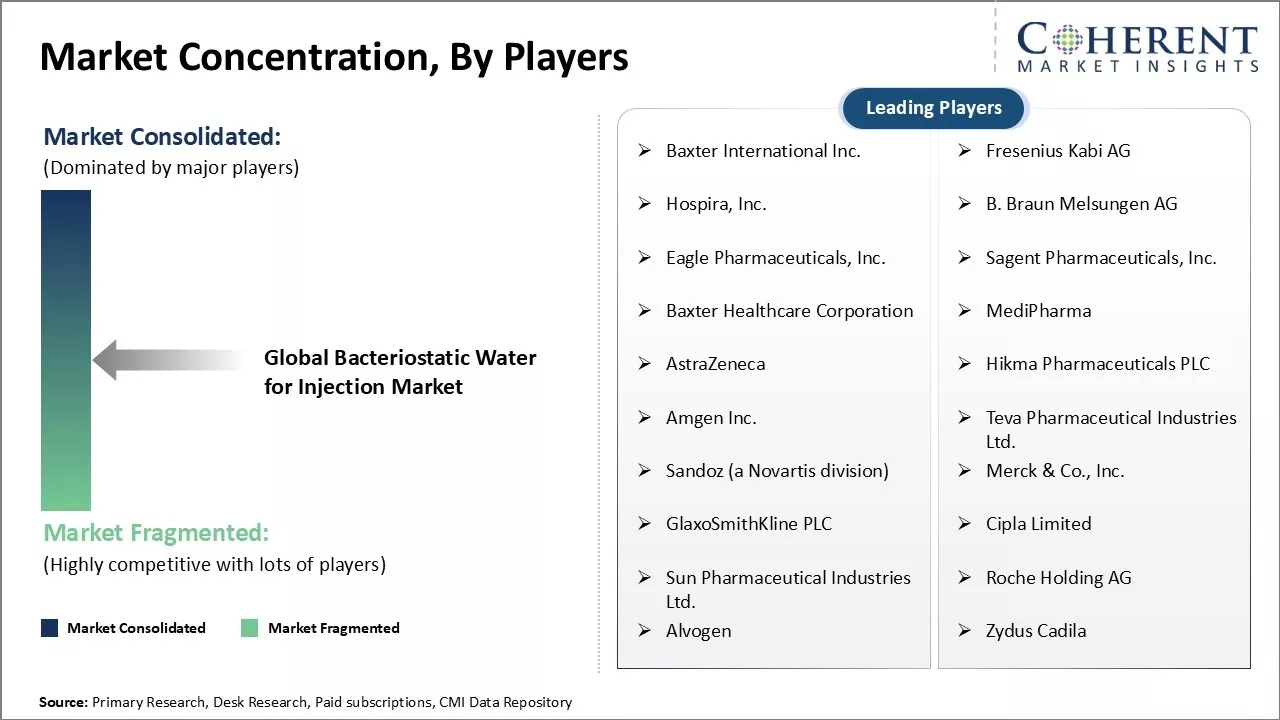
Market Concentration and Competitive Landscape
Get actionable strategies to beat competition: Download Free Sample
Growth of biopharmaceuticals
Growing biopharmaceuticals industry can drive the market growth. Continuous focus on research and development of sophisticated biologic entities to target complex disease pathways has resulted in approval and launch of novel biologics. Engineered for high specificity and efficacy, these biologic drugs have transformed treatment landscapes but includes high costs of development and manufacturing challenges. Most approved biologic drugs require intravenous or subcutaneous injection, owing to their large molecular structure and sensitivity to GI conditions if orally administered.
Constant advancements are being made to produce next-generation monoclonal antibodies, fusion proteins, vaccines and gene and cell therapies. Their production involves fermentation and purification in closed sterile environments. After initial studies confirm safety and efficacy, scaling up of manufacturing becomes crucial to meet growing demands. This involves bulk sterile filling of biologic drug products into vials, prefilled syringes or cartridges from which final dosage forms are prepared for parenteral administration. Bacteriostatic water acts as a sterile solvent used widely during the steps of dissolving, diluting or reconstituting lyophilized biopharmaceutical drug powders post manufacturing.
Rising investments from large pharmaceutical companies as well as smaller biotech startups can lead to increase in number of biologic drugs in clinical pipelines and undergoing FDA/EMA review. With successful approvals, these candidate molecules will require commercial scale sterile filling into final containers from where these will be dispensed for hospital and clinical use. Expanding market landscape of sophisticated biologic therapies can boost demand for bacteriostatic water used during their preparation, handling and administration processes.
Key Takeaways from Analyst:
Global bacteriostatic water for injection market growth is driven by rising prevalence of chronic diseases and increasing number of surgeries conducted worldwide. Growing demand for sterile injections to treat various medical conditions can also drive the market growth. North America currently dominates the market due to well-established healthcare infrastructure and rising surgical procedures in the region. However, Asia Pacific is likely to emerge as the fastest growing market due to expanding healthcare sector, rising medical tourism and increasing spending on healthcare in China and India.
Japan, China and South Korea are also showing sign of recovery after severe impact of COVID-19, thus, propelling opportunities for bacteriostatic water manufacturers in Asia Pacific. Europe can offer lucrative opportunities during the forecast period, owing to new product launches and adoption of minimally invasive surgeries. However, high costs associated with healthcare services can hamper the market growth especially in low and middle-income countries. Stringent regulatory framework and compliance factors can also hamper the market growth. Emergence of alternative infusion solution can also hamper the bacteriostatic water for injection market growth.
Market Challenge - Strict regulatory requirements
Global bacteriostatic water for injection market growth can be hampered due to strict regulatory requirements imposed by various regulatory bodies around the world. As bacteriostatic water is used for injections and is introduced into the human body, there are high safety and quality standards that need to be complied with. The manufacturers need to adhere to current good manufacturing practices (CGMP) and undergo rigorous quality checks and inspections. Any product failures or contamination issues can lead to severe financial and legal consequences. Regulations differ across countries and regions, requiring additional efforts for global market expansion. The regulatory approvals process is often long and costly. Maintaining full compliance with the evolving standards also significantly increases production costs for the manufacturers.
Market Opportunity- Emerging markets
Emerging markets in various regions present a major growth opportunity for global bacteriostatic water for injection market players. With expanding healthcare infrastructure and increasing access to medical services, there has been huge demand for injection drugs and related sterile water in developing economies. Countries across Asia Pacific, Latin America, Eastern Europe, Middle East and Africa offer a large patient pool and represent major untapped potential. Manufacturers should focus on obtaining the necessary regulatory approvals and setting up local production and distribution capabilities in priority emerging markets. This will help them address the demand more efficiently and gain an early foothold before new competitors. Tapping into emerging economies also provides an opportunity to diversify business risks away from saturated markets in developed nations.
Discover high revenue pocket segments and roadmap to it: Download Free Sample
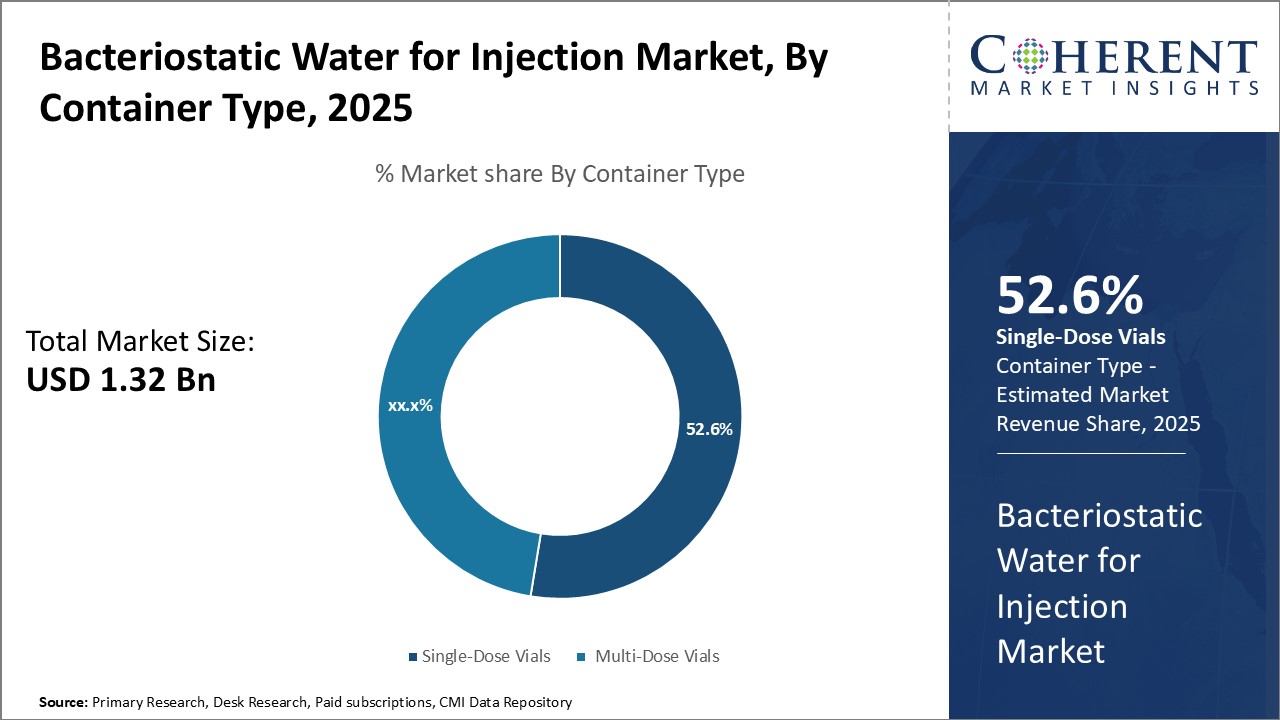
By Container Type -Convenience and safety boosts adoption of single-dose vials
In terms of container type, single-dose vials segment is estimated to contribute the highest market share of 52.6% in 2025, owing to their convenience and safety advantages over alternative packaging types. Being pre-filled and ready-to-use, single-dose vials eliminate the need for multi-step reconstitution processes, reducing medical personnel workload and risk of contamination. This makes them highly preferable for time-critical medical procedures where sterility is paramount. As each vial can only be used for a single patient, these also eliminate potential cross-contamination issues associated with multi-dose vials that are accessed multiple times.
Single-dose vials are particularly suited to meet the stringent sterility requirements of pharmaceutical manufacturing, where bacteriostatic water is commonly used as a diluent. Their self-contained packaging ensures sterility is maintained throughout the product filling and transportation stages. This provides manufacturers valuable assurance that diluted drug solutions prepared using single-dose vials meet necessary purity thresholds. As pharmaceutical production continues scaling up to meet growing global demand, there will be increase in usage of streamlined packaging like single-dose vials that simplify downstream product handling.
The individually-sealed packaging of single-dose vials also appeals to healthcare providers concerned with minimizing infection risks. In medical use and diagnostic settings, their single-patient design helps curb the spread of nosocomial diseases by ruling out potential cross-contamination between patients. This makes single-dose vials a staple across various clinical contexts, from operating rooms to emergency care units dealing with massive patient throughputs each day. Their user-friendly format further promotes adherence to sterile technique protocols.
By Application - Growing pharma outsourcing boosts production segment
In terms of application, pharmaceutical manufacturing segment is estimated to contribute the highest market share of 43.6% in 2025, as growing pharmaceutical production can boost demand for bacteriostatic water.
To reduce fixed costs and focus on core drug development activities, many innovator companies are shifting non-core manufacturing operations to specialized contract development and manufacturing organizations (CDMOs). This outsourcing wave increases CDMOs' share of the total pharma production volume.
Being GMP-compliant components essential for drug reconstitution and manufacturing quality control processes, bacteriostatic water sales directly benefit from expanded CDMO service offerings. Their use helps CDMOs reliably meet clients' stringent product standards.
Non-therapeutic nature of bacteriostatic water allows its international trade without onerous regulatory clearances. This enables CDMOs to consolidate global supply chains and distribute manufacturing across multiple sites to optimize resources. Centralized procurement of common production materials like bacteriostatic water also lowers costs for CDMOs.
As biologics and complex drug pipelines keep pharmaceutical R&D buzzing, there will be growth in pharma outsourcing. Bacteriostatic water manufacturers are well-positioned to capitalize on this trend, supporting larger API and formulated drug production volumes outsourced to CDMOs worldwide.
By End User- Strict hygiene standards fuel hospital adoption
In terms of end user, hospital & clinics segment is estimated to contribute the highest market share of 42.6% in 2025, due to hospitals' immense patient volumes and stringent asepsis protocols surrounding invasive procedures and vulnerable patient cohorts.
In healthcare facilities, risk of hospital-acquired or nosocomial infections poses a serious challenge. According to the WHO, over 30% of inpatients in developed countries acquire at least one healthcare-associated infection during their stay. This pressures hospitals to institute multilayered control measures addressing all aspects of care delivery.
As a critical component in prepping multi-dose injectables and flushing/hydrating infusion lines, bacteriostatic water helps maintain IV therapy sterility. Its antimicrobial properties provide an additional safeguard against contamination compared to regular water. This becomes invaluable when handling immunocompromised patients for whom even minor infections can turn life-threatening.
Hospitals also prioritize optimized efficiency to improve patient turnover and operating margins. Bacteriostatic water packaged for on-the-go access, like in single-patient sachets or vials, fits well with hectic clinical workflows. This streamlines preparation of medications, fluids and diagnostic reagents at the point of care.
Hospitals' focus on preventing healthcare-acquired infections and delivering cost-effective quality care can boost demand for bacteriostatic water to support a variety of frontline medical activities.
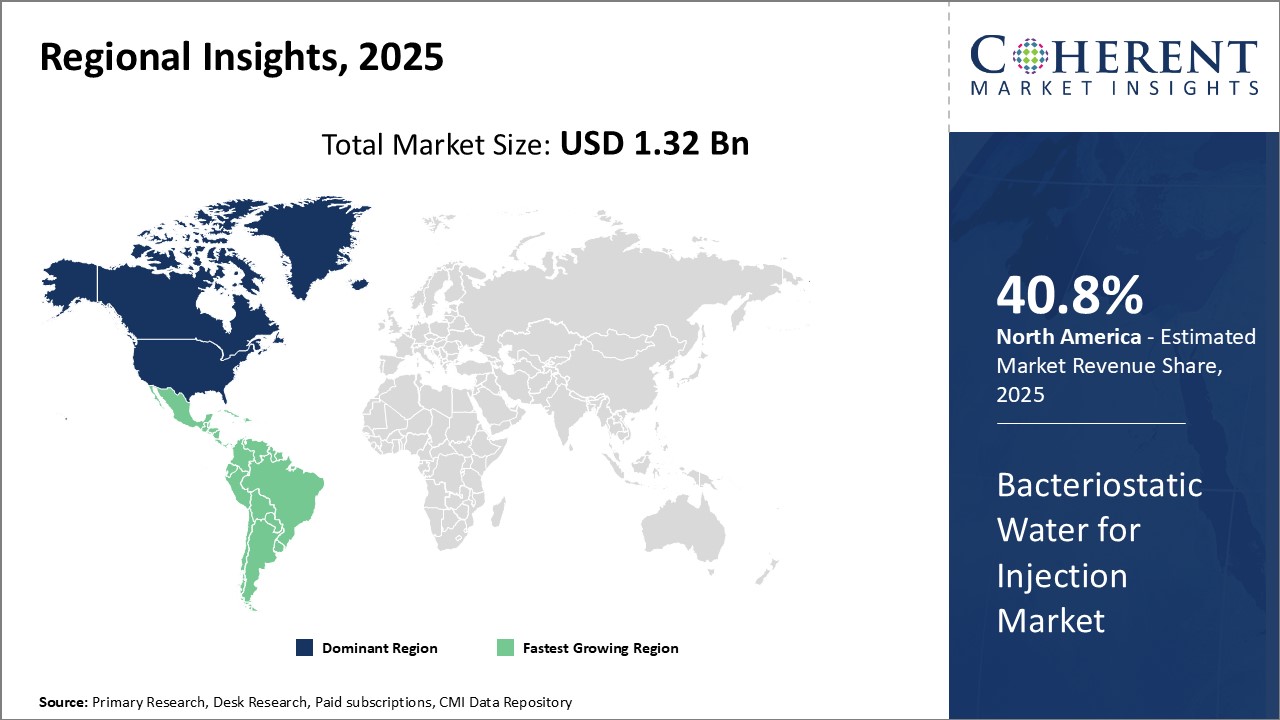
Regional Insights
Need a Different Region or Segment? Download Free Sample
North America region currently dominates the global bacteriostatic water for injection market with an estimated market share of 40.8% in 2025. With the presence of major pharmaceutical companies and availability of advanced healthcare infrastructure, the U.S. accounts for the largest share within the region. The country is also a major exporter of bacteriostatic water for injection to other parts of the world. Stringent manufacturing standards and quality regulations ensure the reliability of products manufactured within the region.
The Latin America region is witnessing rapid growth in the bacteriostatic water for injection market, driven by several factors. Increasing healthcare expenditures and the expansion of healthcare infrastructure are enhancing access to medical services and pharmaceuticals. Moreover, the rising prevalence of chronic diseases and the growing demand for injectable medications are fueling the need for safe and effective diluents like bacteriostatic water. Additionally, regulatory improvements and a rise in biopharmaceutical manufacturing in the region are contributing to market growth. These factors collectively position Latin America as a dynamic and rapidly expanding market for bacteriostatic water for injection.
Market Report Scope
Bacteriostatic Water for Injection Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2024 | Market Size in 2025: | USD 1.32 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2025 To 2032 |
| Forecast Period 2025 to 2032 CAGR: | 8.2% | 2032 Value Projection: | USD 2.29 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Baxter International Inc., Fresenius Kabi AG, Hospira, Inc., B. Braun Melsungen AG, Eagle Pharmaceuticals, Inc., Sagent Pharmaceuticals, Inc., Baxter Healthcare Corporation, MediPharma, AstraZeneca, Hikma Pharmaceuticals PLC, Amgen Inc., Teva Pharmaceutical Industries Ltd., Sandoz (a Novartis division), Merck & Co., Inc., GlaxoSmithKline PLC, Cipla Limited, Sun Pharmaceutical Industries Ltd., Roche Holding AG, Alvogen, Zydus Cadila |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Bacteriostatic Water for Injection Industry News
- On June 2024, Zydus Life Sciences modified its Biotax 1g injection by including 10ml sterile water in Nepal after the country's drug regulator raised concerns over the initial 5ml supply. This change was made to ensure proper dosing for both intramuscular and intravenous use. Zydus emphasized that the adjustment was for convenience and did not affect the product's quality or safety.
- On June 2024, On June 13, 2024, Asahi Kasei launched a new membrane system designed for producing Water for Injection (WFI), a type of sterile water used in preparing injections. This system uses Microza hollow-fiber membranes as an alternative to traditional distillation processes, significantly reducing CO2 emissions and production costs by minimizing the need for steam generation. The system, known for its high filtration performance, offers advantages such as higher water quality, simplified processes, and an impressive endotoxin removal rate of over 99.999%, compared to 99.9% with distillation.
- In May 2021 Hospira, Inc., a Pfizer company, recalled lot DN9185 of Sterile Water for Injection, USP, 100 mL Single Dose Glass Fliptop Vials due to a confirmed report of visible particulate in a vial. While the risk to patients is low, potential adverse events could include anaphylaxis, fever, vein irritation, and other serious conditions.
- In July 2021, Nexus Pharmaceuticals introduced potassium chloride in water for injection, available in single-dose IV bags with concentrations of 10mEq/100mL, 10mEq/50mL, and 20mEq/50mL. This injectable form of potassium chloride is used to treat potassium deficiency when oral supplementation is not feasible
*Definition: Global bacteriostatic water for injection market consists of manufacturing and sale of bacteriostatic water meant for injection purposes. Bacteriostatic water contains antimicrobial preservatives which inhibit the growth of microorganisms, helping increase the shelf-life of compounded sterile preparations. It is widely used to dilute or reconstitute medications as well as for rinsing ophthalmic medications and medical equipment.
Market Segmentation
- Container Type Insights (Revenue, USD Bn, 2020 - 2032)
-
- Single-Dose Vials
- Multi-Dose Vials
- Application Insights (Revenue, USD Bn, 2020 - 2032)
-
- Pharmaceutical Manufacturing
- Medical Use
- Diagnostic Applications
- Others
- End User Insights (Revenue, USD Bn, 2020 - 2032)
-
- Hospital & Clinics
- Diagnostic Labs
- Research Labs
- Others
- Regional Insights (Revenue, USD Bn 2020 - 2032)
-
- North America
- U.S.
- Canada
- Latin America
- Brazil
- Argentina
- Mexico
- Rest of Latin America
- Europe
- Germany
- U.K.
- Spain
- France
- Italy
- Russia
- Rest of Europe
- Asia Pacific
- China
- India
- Japan
- Australia
- South Korea
- ASEAN
- Rest of Asia Pacific
- Middle East
- GCC Countries
- Israel
- Rest of Middle East
- Africa
- South Africa
- North Africa
- Central Africa
- North America
- Key Players Insights
- Baxter International Inc.
- Fresenius Kabi AG
- Hospira, Inc.
- Braun Melsungen AG
- Eagle Pharmaceuticals, Inc.
- Sagent Pharmaceuticals, Inc.
- Baxter Healthcare Corporation
- MediPharma
- AstraZeneca
- Hikma Pharmaceuticals PLC
- Amgen Inc.
- Teva Pharmaceutical Industries Ltd.
- Sandoz (a Novartis division)
- Merck & Co., Inc.
- GlaxoSmithKline PLC
- Cipla Limited
- Sun Pharmaceutical Industries Ltd.
- Roche Holding AG
- Alvogen
- Zydus Cadila
Share
Share
About Author
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Missing comfort of reading report in your local language? Find your preferred language :
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
EXISTING CLIENTELE
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients
